Algorithm for Calculating the Optimization of the Parameters for the Electroextraction of Metal Based on the Example of Zinc
Author: Kashapov N.F., Kashapov L.N., Chebakova V.Yu.
Journal: Журнал Сибирского федерального университета. Серия: Техника и технологии @technologies-sfu
Section: Математическое моделирование. Численный эксперимент
Article in issue: 1 т.19, 2026.
Free access
A numerical algorithm combining the fourth-order Runge–Kutta scheme with the Hooke–Jeeves direct-search optimiser is proposed to solve an inverse kinetic problem that arises during zinc powder electrodeposition from alkaline electrolytes. The model describes two competing cathodic chains – zinc reduction and parasitic hydrogen evolution – through a coupled Cauchy system with positivity constraints. Unknown rate constants are estimated by minimising the squared deviation between calculated and experimental zinc yields, which reduces the amount of laboratory testing required. An applied result of the calculations also provides the ability to predict the yield of the deposited metal powder. Verification against published data at current densities of 125–500 A/m² shows relative errors below 5 % after 60 min of electrolysis and confirms that nucleation effects dominate only at the earliest stages. The approach therefore offers an efficient tool for parametrising and optimising electroextraction processes for zinc and other metals. At the same time, the velocity constants that are found can be used for calculations based on larger-dimensional models when calculating the spatial characteristics of an electrical system.
Electroextraction optimisation, Hooke–Jeeves method, Inverse reaction kinetics, Zinc powder deposition, Runge–Kutta simulation
Short address: https://sciup.org/146283256
IDR: 146283256 | UDC: 544.6
Алгоритм оптимизации параметров для электроэкстракции металла на примере цинка
Для решения обратной кинетической задачи, возникающей при электроосаждении цинкового порошка из щелочных электролитов, предлагается численный алгоритм, сочетающий схему Рунге-Кутты четвертого порядка с методом численной оптимизации, а именно методом прямого поиска Хука–Дживса. Модель описывает две конкурирующие катодные цепи – восстановление цинка и параллельное выделение водорода – с помощью системы, состоящей из задач Коши с наложенными ограничениями. Неизвестные константы скоростей оцениваются путем минимизации квадратичного отклонения между расчетным и экспериментальным выходами цинка, что сокращает объем необходимых лабораторных исследований. Прикладным результатом расчетов также является возможность прогнозирования выхода осаждаемого металлического порошка. Сверка с опубликованными данными при плотностях тока 125–500 А/м2 показывает относительную погрешность менее 5 % после 60 мин электролиза и подтверждает, что эффекты зародышеобразования преобладают только на самых ранних стадиях. Таким образом, этот подход предлагает эффективный инструмент для параметризации и оптимизации процессов электроэкстракции цинка и других металлов. При этом найденные константы скоростей могут быть использованы для расчетов по моделям большей размерности при вычислении пространственных характеристик электротехнической системы.
Text of the scientific article Algorithm for Calculating the Optimization of the Parameters for the Electroextraction of Metal Based on the Example of Zinc
Annually the world generates a large amount of industrial waste containing heavy metals, especially due to ferrous and non-ferrous metallurgy and metalworking. These materials are usually classified as hazardous due to the possibility of the leaching heavy toxic elements during periods of long-term – 93 – storage. At the same time, due to the rather high value of non-ferrous metals, their rational use is attracting more and more interest.
One of the recycling methods being used is electroextraction of metal powder from alkaline electrolytes. The precipitation of metal powders in hydroelectrometallurgical processes, as well as the properties and the resulting cathode deposit, are all influenced by a set of parameters including ones such as: electrolyte concentration, temperature, the density of applied current, the material making up the electrode and electrolyte acidity. As a result, a large number of experiments are necessary to identify the optimal parameters necessary to determine the maximum performance level while maintaining specific degrees of energy consumption.
In the study [1], research was carried out on processes involved in the electrodeposition of Pd, Rh and PdRh in alcohol solutions and alcohol solutions under the presence of impurities in the form of aldehydes (formaldehyde, acetaldehyde). The density of the deposited coating and the size of the resulting nanoparticles were also estimated.
This paper [2] describes the following processes: electroextractions of cobalt, which leads to a decrease in manganese impurities from sulfate solutions of cobalt (II) and manganese (II).
In the article [3], a proposal is made for the optimization of the process for obtaining enriched boron from boron carbide scrap formed during the production of boron carbide and it is noted that electroextraction reduces the presence of impurities in the form of oxygen and carbon.
In the article [4], a proposal was made to use electroextraction to remove the residual copper content in cast iron and steel.
Laboratory studies [5] were carried out on the cathodic process of lead electroextraction from trilonate electrolyte obtained after the purification of solutions after the leaching of lead cakes from zinc production.
Due to the widespread use of zinc to facilitate the protection of metal from corrosion, as well as in the production of alloys and paints, it has seen wide usage as a cementing metal. The use of zinc in hybrid batteries is also an interesting area of exploitation. In this regard, in [14], a new method for creating zinc nitride hybrids for practical application as highly efficient multifunctional electrocatalysts for the process of electrocatalysis and energy storage in Zn-air batteries is proposed. Hybrid batteries based on processes where zinc deposition is present are also considered in [15, 16].
Within the context of the current study the electroextraction of zinc powder from alkaline electrolyte is also very relevant. In [17] the technology used for obtaining zinc through the processing of waste powder created by steel production is detailed, and in [18] an overview is given of work by several authors who studied the overall influence of various conditions present in the electroextraction of zinc powders from alkaline electrolytes and whose finding showed that there is no consensus on the real influence of electrolysis in terms of current output and power consumption. In [19] the current output data for the electrolysis of a sodium hydroxide solution in the presence of sodium tetrahydroxocinate obtained by leaching zinc-containing ores is presented.
When developing new technologies and optimizing ones which already exist mathematical modeling makes it possible to reduce the number of experiments by linking internal and external parameters, and thus in [6] a computer simulation of the Pd deposition carried out in ethanol is presented with calculations having been performed using the Vienna Ab-initio Simulation Package (VASP version 5.3.3).
Moving on to the precipitation of stabilized ibuprofen nanoparticles from the liquid phase, in [4] a spatial model is presented. Unfortunately however, the implementation of spatial models is quite complex and often requires large computational resources. At the same time, models made up of and containing kinetic equations, allow us to perform the initial analysis of the efficiency of the electrolyzer given the fact that Faraday’s law produces overreaching yield estimates, especially when in the presence of parallel branches of step reactions.
If the values of the velocity constants of near-electrode processes are known, then the problem of predicting the yield of the deposited substance is not particularly difficult and can be calculated numerically using Runged‒Kutta methods used to solve calculations with regard to systems consisting of Cauchy based calculations and tasks. An example of this in [7] is the usage of the second-order Runge-Kutta method to estimate the transient power regimes existing in a thermal reactor. In most of the cases that were analyzed in the authoring of this paper, the transient power values were comparable to the values calculated by other methods, while in all cases the relative deviations calculated were found to be below the order of 10–2.
In study [8] Runge-Kutta-Felberg method were used in studying the influence of homogeneous-heterogeneous reactions on the surface of a rotating disk subjected to solar heating while a hybrid nanofluid was flowing around it. Therefore if can be stated that the Runge-Kutta method, in combination with other methods, can be used as an integral part of formulating a numerical algorithm used in the calculation of more complex problems.
In the case when reaction rate constants are unknown, tasks involving inversion often occur, which can sometimes be reduced to problems involving optimization, for example in the case of chemical reactors [9–12] and in problems involving chemical kinetics in gases [13]. In this paper inverse problems are solved under conditions when the rate constants are unknown and the mass of the deposited metal powder is given at certain points in time. The formulation and solution to this problem has been carried out through the example of obtaining zinc powder by electrolysis of a sodium hydroxide solution containing sodium tetrahydroxocinate. In the solving of this problem, an algorithm was developed based on the Runge-Kutta method for solving system wide kinetic equations and the Hook-Jeeves direct search method for determining the rate constants of kinetic processes.
1. Task Parameter Determination Based on the Example of Zinc Electrodeposition
Based on the data in [19], we have shown that it is possible to predict the zinc yield using numerical optimization. When leaching an ore containing zinc, depending on the concentration of sodium oxide in the solution, the zinc in the electrolyte can contain both zinc oxide ZnO and the unified material [Zn(OH) 4 ]2–. It was shown in [20] that, in general, a 20 % concentration of sodium hydroxide solution NaOH was enough so that the molar ratio of hydroxyl group ions OH– to zinc was sufficient for their existence [Zn(OH) 4 ]2–, in such a case, if the concentration of hydroxyl group ions OH- is too small, then the compound [Zn(OH)4]2– becomes zinc oxide Zn O. Thus, the process of leaching zinc-containing ore is described as a general dissolution reaction of the following type 2NaOH + Zn + 2H 2 O → Na 2 [Zn(OH) 4 ] + H 2 .
In direct connection to this, there are different opinions regarding the flow of near-electrode reactions causing the release of zinc. Thus, in review [18], which was based on the work in [21], it is shown that during the electrodeposition of zinc [Zn(OH)4]2–, the compound is subject to modification into one which is hydrogenated Zn(OH) 2 , in this case zinc hydroxide is an insoluble base and, as a consequence, its presence should be noted in the resulting precipitate. However, in most studies this has not been observed.
The construction of a mathematical model is based on the following assumptions:
-
1) Na2[Zn(OH)4] in the electrolyte the ions become completely dissociated.
Na2[Zn(OH)4]^2Na+ +[Zn(OH)4]2' (1)
-
3) at the electrode, zinc is released during the following electrochemical step reactions: [Zn(OH)4]2- —^Zn2+ + 40H“
Zn2+ +2e—^Zn, (2)
where k 1, k 2 - is the constant rate of the corresponding reactions.
-
5) In work shown in [18, 19], the hydrogen yield is noted together with the zinc precipitate at the cathode, so it is necessary to add the reactions of hydrogen release at the cathode to the cathodic step reactions (2). This is describe in the form presented in [22, 23]:
H20—^—>H+ +0H“,
H++OH’-^H2O,
H++e—^H,
H + H—^H2, (3)
where k 3, k 4, k 5, k 6 shows the rate constants of the corresponding reactions.
It should be noted that the parallel emission of hydrogen leads firstly to alkalinization of the nearelectrode layer, which in turn can lead (under certain values of the hydrogen index), to the precipitation of hydroxides or oxalates in relation to the deposited metal, something which was possibly observed in [21]. Secondly the aforementioned emission leads to the introduction of adsorbed hydrogen atoms into the growing metal precipitate. These processes lead to a change in the properties and structure of the resulting precipitate (the so-called hydrogen brittleness of the metal). One of the tasks associated with the optimization of the zinc production process is to obtain the parameters for which the hydrogen yield is minimal. Therefore, despite the principle of the independence of parallel reactions, we have included both of them in the mathematical model although the parallel reactions are not directly related to each other:
Here E 0 – shows the standard electrode potential of the reaction, but in the case of parallel reactions, it may shift, therefore in [24] it was noted that for many systems, the potential of the electrode in the reaction of cathodic metal deposition due to polarization, can be more negative than the potential of the reversible hydrogen electrode.
Despite the fact that these competing reactions are not directly related to each other, the rates of the step reactions depend on the overvoltage ratio
, . | BE ] \ overvoltage
-
k = A exp--exp--— .
-
\ RT) \ RE )
Here A, B are constants from the Arrhenius equation, E signifies the activation energy , R is the universal gas constant , F is the Faraday constant , в is the surface inhomogeneity coefficient, T is the electrolyte temperature, E overvoltage is the overvoltage at the electrode depending on the concentrations of only those potential-determining components of the electrolyte that participate in electrode reactions at this electrode
RT I \
^overvoltage _"^^Vzn2+^+ ^^H^Zn/-
Here shows the n-charge values, the concentrations of ions, atoms and zinc, respectively.
Another factor affecting the rate of solid product deposition is the nature of the deposited coating, namely whether it is porous or a solid continuous layer. In the case of a porous film, it has been shown not to provide resistance to reagents suitable for the interface of the medium and does not affect the reaction rate; in the case of a non-porous film, the resistance of the boundary layer can be disregarded, when considering the main resistance of the layer of the released product [25]. Moreover, if the process is controlled by diffusion through the boundary layer, then the speed of the process , where
δ is the thickness of the product layer and S is the area of the working electrode. S- is the thickness of the product layer in the approximation of the solution of the Yander equation and can be considered proportional to time: and .
The structure of a metal deposit is determined by a number of factors, such as the composition of the solution, the presence of impurities in that solution, preliminary surface preparation, current density, etc. In research [26] the results of a study of the kinetics of leaching low-grade zinc silicate ore with sulfuric acid are presented and it is shown that the rate of the process is determined by the diffusion through the layer. In [27] the effect of additives on the structure of the selected layer Zn (porous) is shown and it is stated that at a sufficient current density, the deposition process is a reaction with a controlled transfer of mass. The paper also presents photos of the surface at different current densities during the electrolysis of a solution NaOH c at concentrations of 240 g/l and c 25 g/l at a temperature of 35C. The spongy and porous nature of the deposited zinc coating was also noted in [28]. Therefore, the deposition of a porous film is used to simulate zinc deposition.
Thus, the mathematical model consists of a system describing electrochemical stepwise cathode reactions and has been calculated as being thus:
Here D _
Zn(OH)
dCH2O _ ~^--^н2о
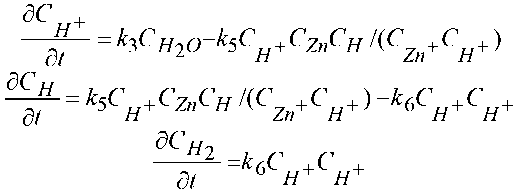
SC 2-
Zn(OHy dt
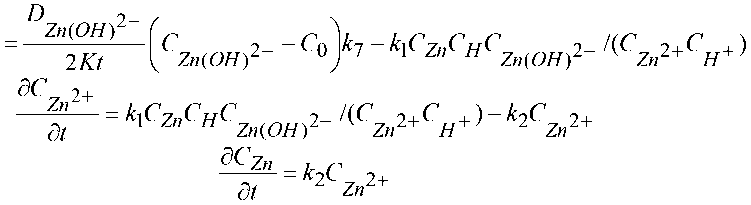
2- is the diffusion of sediment ions [Zn(OH) 4 ]2– through a porous film, C 0 is the
concentration of these ions within the volume of the electrolyte.
This notation of the kinetic equations described by the solution of Cauchy problems implies that the reaction rate constants are positive, i =1,…, 7, in the proportionality coefficient K, and the concentration values, j ={,,}.
To calculate the mass of deposited zinc sludge, calculations are done based on zinc concentration and film volume
MZn = CZnS^2KT x 65,38, (5)
where T is the final time of the calculations.
The process of water associationH++OH’-^H2Ois not taken into account in this model since the calculations and analysis of the rates of step reactions performed in [23] in predicting the cathodic yield of hydrogen during the electrolysis of an alkaline electrolyte showed a very small value in the constant that differed very little from the zero constant k 4.
When setting the initial conditions on the state of the system before applying voltage we operated under the belief that sodium hydroxide, as well as sodium tetrahydroxocinate, completely dissociate into ions. Thus, the hydroxyl group is present in the electrolyte both in the free state OH- and in the bound state [Zn(OH)4]2–, in this case, the bound state is determined by the concentration of zinc received from the ore. The concentration of positive ions is calculated from the ionic production of water. The concentration of hydrogen molecules and atoms and the released zinc is considered “zero” at the initial time marker. When choosing the initial approximations for the rates of constant nearelectrode processes, we took into account that in the case of two competing chains of near-cathode reactions, one of which proceeds with the release of a solid phase, more complex kinetic regularities are observed. Whereas when conducting calculations associated with the release of the gas phase, which is characterized by only one chain of step reactions and when the initial approximation of the reaction rates can be selected based on the ratio between the rates of step reactions, stoichiometric numbers and the rate of limiting reaction [22, 23].
From the experiments the known output data on the current of the released substances at certain points in time can be shown as being: l= 1,…, n .
The solution of the problem of finding the rate constants of near-cathode electrochemical processes and predicting the output is based on minimizing the functional:
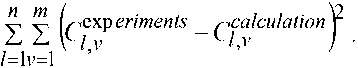
Here l it is responsible for the summation over time, v is responsible for the summation of substances whose concentrations are known. Ideally, it is desirable to have data for both hydrogen and the mass of the precipitated sediment, but as a rule, this is the mass of the deposited substance at several different time points and the functional takes the form of:
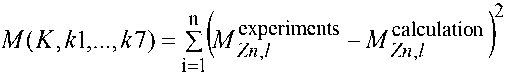
2. Method for Solution
In [22, 23], the calculations presented show the parameters of gas release during electrolysis for the case of a single near-electrode reaction of substance release in the gas phase, while there was no more than a 10 % deviation of the actual predicted values of the released gas concentration as opposed to the experimental data.
The algorithm that the authors have proposed, in contrast to well-known statistical methods, such as the construction of regression models and interpolation methods, such as splines, allows us to determine the predicted values of the mass of zinc using a smaller number of experiments. This relates to not only the form of functional dependence with fitting coefficients, but allows for the direct evaluation of: the contributions of electrochemical processes, the identification of limiting processes and obtaining boundary conditions for equations calculating the concentration of components of the electrolyte in volume, all directly based on the theory of electrochemical processes.
In the current work the application of the numerical optimization method to solving the inverse problem that arises when predicting the current yield of a substance in the solid phase when in the presence of two parallel near-cathode reactions that are not directly related to each other, and by using the example of zinc deposition with concomitant hydrogen release, is shown.
The proposed algorithm is based on the Hook-Jeeves method of direct search, supplemented by constraint checking, and the Runge-Kutta method applied to solving a kinetic system of equations describing stepwise electrochemical reactions.
The Hook-Jeeves method refers to direct search methods and relies directly on the value of the functional from which the minimum is searched. It occurs iteratively and includes two stages.
The first stage consists of an exploratory search around the base point (K, k1,…, ki,…, k7) by the increment vector, H(h, h1,…, h7) where hi is the step along the coordinate, ki, i =1,..,7. For the implementation of this stage, for each individual coordinate the values of the functional (6) are calculated at three points: the direct points g(K, k1,…, ki,…, k7) and the points separated from it by one step along the coordinate under study. Then the point at which the functional takes the minimum value is used as a new base point. These iterations continue until the reference point stops changing. At the same time, the calculation algorithm assumes that the point where the coordinate value takes a negative value or the values of any of the concentrations taking a negative value during the calculation, are then excluded from consideration.
For example, for the coordinate responsible for the proportionality coefficient K these are the points g( K , k 1 ,…, k i ,…, k 7 ), g 1 ( K , k 1 ,…, k i ,…, k 7 ), g 2 ( K , k 1 ,…, k i ,…, k 7 ), here K – h ≥ 0. To calculate the functional (6) at these points ( K , k 1,…, k i,…, k 7), M ( K+h , k 1,…, k i,…, k 7), M( K-h , k 1,…, k i,…, k 7), through the formula for converting the concentration to mass (5) it is necessary to know the mass of the metal deposit at the moments Tl, l= 1,…, n.. To calculate the mass of deposited sediment MJ* for moments T l , l= 1,…, n We need to solve a system of kinetic equations consisting of Cauchy problems (4). To solve this system, we used the fourth-order Runge-Kutta method with a time step of ∆t,
The system is counted up to the point in time T while preserving the zinc concentration values for all time points necessary for calculating functional (6) Tl , l =1,…, n , then, according to formula (5), we calculate the mass of the deposited sediment at these time points and calculate the functional itself (6). Further, for the new value of coordinate K we take that of the values K , K + h , and K – h provided that K – h ≥ 0 possesses a value of the functional (6) which accepts the minimum. The recalculations continue until the value of the coordinate K doesn’t stop changing, and then we move to the next coordinate.
After calculating the last coordinate, a new point g ’ is obtained, which can either coincide with the original point g ’ = g or differ from it g ’ ≠ g .
Then, if the original reference point coincides with the newly obtained point g ’ = g , then we reduce the values of the components of the vector of coordinate increments and continue the search around the reference point. Otherwise, in the worst case, when g ’ ≠ g , a sample search is then carried out. In it the values at which, again, the value of functional (6) is minimal are taken as the new basis point. The exit condition is a limit on the size of the components of the increment vector H .
When calculating the objective function, we used a numerical method from the Runge-Kutta method family at the fourth order of accuracy, i.e. its error rate is O( t 4), where t is the time step. To search for the objective function, the Hook-Jeeves method was used. Its accuracy is determined by setting a limit on the possible deviation of the calculated parameter value from the values of the parameters that determine the minimum. Thus, the error rate of the method is represented as max i (Δ t 4, ε i ).
3. The Results of Numerical Calculations
The proposed algorithm, which implements the mathematical formulation of zinc powder deposition, was verified on the full-scale experiment described in [19]. It investigated the effect of applied voltage on near-electrode processes of the electrodeposition of zinc in sodium hydroxide solutions on a stainless steel cathode. When preparing the electrolyte, the ore containing zinc oxide was calcined at 400 °C for 2 hours, then it was crushed to an approximate size of 150 µm and leached with a 20 % sodium hydroxide solution at 100 °C for 4 hours. Further the solution was injected with sodium sulfide, for precipitation of lead contained in the resulting solution. The prepared electrolyte is then ready for use after separating the liquid and solid phases. Zinc ion concentration Zn2+ in the prepared solution consists of 10 g/l. The working electrode was made of stainless steel and has an area of 1 cm2. The reference electrode was selected as a saturated calomel electrode based on the reaction between elemental mercury and mercury (I) chloride, and a graphite plate was used as a counter electrode.
We used the experimental conditions to deduce the initial conditions:
1,000 grams/l of solution gives 80 %
^NaOH\t=0 gives 20 %
Thus, ^NaOH |/-q = 250 the gram/l molar mass of sodium hydroxide is 39.997 g/mol, thus ^NaOH |/=о = 250 g/l: 39,997 g/mol≈6,25 mol/l. A molar mass of zinc of 65.38 g / mol and a concentration of 10 g/l gives approximately 0.153 and following the dissolution reaction:
2NaOH + Zn + Na2 [Zn(OH)4] ^ 2Na+ + [Zn(OH)4]2 + H2 .
Thus we will have
0,153 mol/l. At the same time 0,153 mol/l zinc binds only
2×0,153≈0,306 mol/l the remaining 6,25 mol/l –0,306 mol/l = 5,944 mol/l participates in the dissociation reaction NaOH ^Na+ + OH , as sodium hydroxide is a strong base, thus ^ он\=й ~ 5,944 mol/l. The initial concentration of the hydroxyl ion is not directly involved in the near-cathode step reactions identified by us, but the ionic product of water shows that under normal conditions С +1 н I
0,168 14
mol/l. Zinc is bound by the hydroxide group, and the values of the absolute concentrations are taken close to “zero”, taking into account the fact that the deposition of metals occurs around the nuclei, the electrochemical processes of the stage of formation of the nuclei (adatoms) differ from the stage of their growth, which we are actually interested in C^J^O, while the concentration of zinc atoms taken is
750^:18-^* 41,7mol/l
1 mol
equal to “zero”Cz.|«=°. From the concentration of water we get Qo|
, under normal conditions, water contains ^i^-^xio mol/l, next, we take ^ h|
О,
/=О
taking into account the necessary presence of the nuclei. Since in parallel reactions, one of which takes place with the deposition of the substance in the solid phase, there is a difficulty in determining the limiting reaction, and consequently, the values of the initial approximations for the values of the rate constants of all stage reactions. We take their values to be equal to one. Later, numerical calculations showed a good level of convergence of the calculations with changes in the approximations for the initial values of the rate constants of processes.
In [19], the experimental data on the yield of zinc is shown in%, and is calculated according to the following formula: 7Zn=—1----^-xl00%where m0 is the initial mass of the cathode in g, mi the
1.22x/x/ mass of the cathode with zinc deposited on it in i – at the point in time. The indicators were taken every 10 minutes, I – current strength in A, and t=10*i/ 60. Since the article [19] does not provide data on the hydrogen yield, we will construct the functional only from the known mass of deposited zinc.
The calculations were carried out using two methods and showed that the values of zinc concentrations in the calculation did not actually depend on the method of calculation and the construction of the numerical algorithm, that is, the differences in the values were insignificant. At the same time, the calculation time for the second method was shorter, although it consisted of two stages. The first method consisted in a direct calculation using the complete model described by the system (4) with the restrictions and co-relationship imposed on it (5). In the second method, calculations were performed in two stages. At the first stage, we obtained initial approximations for the rates of stepwise reactions of zinc production:
дС ?_
Zn^OHy
dt
SC 2+
Zn2+ dt
D э_
Zn(OHy с С^к кС
2Kt Zn^OHy 7 1 Zn^OHy
— к\( ? — каС
1 Zn(OH)2~ 2 Zn2+
SCZ„ dt
— к^С 2+
2 Zn2+
At the second stage, the obtained constants were refined using the system (4). Accordingly, this suggests that for more accurate calculations, it is necessary to know the volume of released hydrogen, for example, in [19] it is noted that the release of hydrogen depended on the rate of zinc expansion, which was determined by the values of the applied current. Table 1 shows a comparison of the calculated values for the mass of deposited zinc and the experimental data of [19], where functional (6) was constructed from the mass of zinc at time points T =10 minutes and T =60 minutes. Table 2 shows the ranges of the experimentally obtained mass Zn and the calculated values obtained at a current strength of 125A at two points (t=1 hour and t = 10 minutes)
The number of points with experimental values used to calculate the objective function is determined by the number of experiments performed. The influence of the points where data was taken does not have a decisive effect on the error rate, so in [23] this question was investigated
Table 1
|
Time, mines |
Relative deviation of calculated values from theoretical values (%) |
|||
|
125 A/m2 |
375 A/m2 |
250 A/m2 |
500 A/m2 |
|
|
20 |
35 |
27 |
40 |
26 |
|
30 |
16 |
21 |
26 |
18 |
|
40 |
12 |
11 |
17 |
8 |
|
50 |
3 |
3.5 |
8 |
0 |
|
60 |
0 |
0 |
0 |
0 |
|
70 |
2 |
3 |
6 |
3 |
|
100 |
25 |
2 |
18 |
8 |
Table 2
|
T, min |
Range of experimental data [19] |
Calculated values |
|
20 |
0.467–0.49 |
0.30115 |
|
40 |
0.976–1.03 |
0.8516 |
|
60 |
1.4945–1.6 |
1.564 |
|
70 |
1.761–1.921 |
1.97 |
|
100 |
2.49–2.6687 |
3.36 |
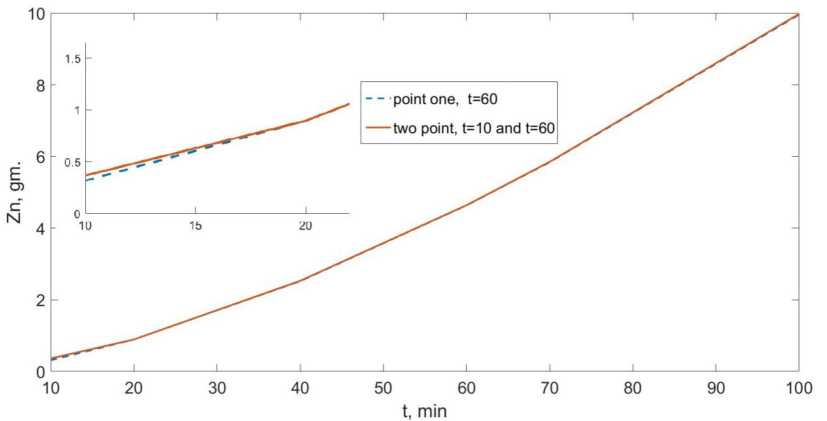
Fig. 1. Model of zinc yield during electrolysis of 20 wt% NaOH at 500 A/m² for the processes of substance release in the gas phase, when the approximation of invariance and uniformity of the electrode area were used. When a zinc deposit is deposited on the electrode, this process is shown in Figure (1). It shows a comparison of the calculated data at a current strength of 500A in two cases: calculations were given at one point t=1 hour, and at two points (t=1 hour and t=10 minutes)
The calculations showed the greatest discrepancy at the beginning of the electrolysis process. From this it can be concluded that the phase of nucleation has a marked effect at the initial stage and then with the growth of zinc crystals, its influence decreases. Thus, at the initial stage of growth of zinc crystals, hydrogen is released both on the electrode and on the surface of the zinc crystals, which makes the surface strongly inhomogeneous. So we conclude that modes at current density 500 A/ m2and 375 A/m2, according to [19], have a lower rate of coating the electrode area with zinc than 125 %. A/m2 and 250 A/m2 and, accordingly, a larger surface for the release of hydrogen, as well as a longer time for the release of hydrogen on the cathode material, since it is released more easily on steel than on metallic zinc. Also accordingly, more accurate information on the development of the initial stage could be obtained by taking into account data on the yield of hydrogen.
Conclusions
In this paper, we have numerically solved the inverse problem of finding the reaction rates of heterogeneous processes in the electroextraction of metal powders, taking into account the parallel release of hydrogen, using the example of zinc. To solve this problem, we propose using numerical algorithms for its solutions based on the Hook–Jeeves direct search method and the Runge-Katt method for solving systems consisting of Cauchy problems. An applied result of the calculations also provides the ability to predict the yield of the deposited metal powder. The calculation algorithm was verified on the basis of experimental data on a mass of deposited zinc and showed a high level of agreement with the experimental data. Calculations have shown that at the initial stage, the stage of the growth of nuclei has a great influence, accordingly on the difference in the rate of hydrogen formation on the surface of the steel from which the electrode is made and on the surface of the zinc crystal. At the same – 103 – time, numerical studies involving both parallel reactions have shown that the influence of the initially set concentration of zinc ions does not affect the accuracy of calculations. Thus, the stage of zinc ion formation is not a limiting one. In the future, it would be of interest for the development of the model, to study the phase of the appearance of nuclei (anatomists) and to include the most influential processes of nuclei formation in the mathematical model.
Since the rate constants depend on factors specific to the systems under direct investigation, such as the inhomogeneity and roughness of the electrode surface, the charged particle transfer coefficient and the electrolyte concentration, it seems impractical to verify the algorithm by comparing the process rates determined for other systems. At the same time, the velocity constants that are found can be used for calculations based on larger-dimensional models when calculating the spatial characteristics of an electrical system.
Conflict of Interest
The authors declare that they have no conflicts of interest.


