Ameliorative potential of Allium cepa or quercetin on the histology, immunohistochemistry and oxidative stress biomarkers parameters against potassium bromate-induced hepatotoxicity in adult male rats
Author: Agu Solomon T., Meshioye Deborah O., Akintola Ahmed A., Gbadegesin Michael A., Odunola Oyeronke A.
Journal: Журнал стресс-физиологии и биохимии @jspb
Article in issue: 3 т.18, 2022.
Free access
Background: Allium cepa has been linked to a variety of health advantages, although little is known about its hepatoprotective properties. Using biochemical, immunohistochemical, and histological markers, the current study was conducted to examine the efficacy of quercetin-rich aqueous extract of Allium cepa (AEAC) against potassium bromate-induced hepatotoxicity in Wistar rats and the probable mechanism. There are a total of 40 male Wistar rats used in this study and were equally divided into eight (8) groups. Group A: Control. Group B: potassium bromate, KBrO3 (dissolved in water), at 30 mg/kg. Group C: Quercetin as a standard (50 mg/kg). Groups D and E: AEAC, at 300 mg/kg and 150 mg/kg respectively. Groups F and G: KBrO3 plus AEAC. Group H: KBrO3 plus quercetin. On alternate days for 90 days, all administrations were done by intubation. Results: KBrO3 generated a significant difference in the levels of oxidative stress biomarkers and DNA fragmentation compared to the control (p 0.05). Histological and immunohistochemical examinations of liver samples revealed patterns that were comparable to those seen in enzyme biomarkers. In all cases, combining KBrO3 with AEAC or pure quercetin attenuated the effects of KBrO3. Conclusion: Therefore, potassium bromate induced hepatocellular toxicity via oxidative stress while treatment with the AEAC ameliorated its effects.
Allium cepa, hepatocellular toxicity, histology, immunohistochemistry, onion, rats
Short address: https://sciup.org/143179052
IDR: 143179052
Text of the scientific article Ameliorative potential of Allium cepa or quercetin on the histology, immunohistochemistry and oxidative stress biomarkers parameters against potassium bromate-induced hepatotoxicity in adult male rats
Potassium bromate (KBrO 3 ) is a by-product of ozonation water disinfection and may be found in both tap and bottled water (Liu and Mou, 2004). Furthermore, due to its oxidizing capabilities, KBrO 3 is commonly employed as a food ingredient in the bread-making process for wheat maturation and as a neutralizer in cold-wave hair treatments (Kurokawa et al ., 1990). Previous research has shown that potassium bromate's harmful effects are caused by the production of reactive oxygen species (McDorman et al ., 2005). Lipid peroxidation and genotoxicity are caused by reactive oxygen species (Jaloszynski et al ., 2007). The increase of 8-hydroxyguanosine (8-OH dG) production from DNA deoxyguanosine residues by free radicals has been attributed to KBrO 3 ’s genotoxicity (Ahlborn et al ., 2009). Furthermore, the International Agency for Research on Cancer has categorized KBrO 3 as a category 2B carcinogen (probably harmful to humans) (IARC, 2019). Several cases of human toxicity, including hearing and renal impairment, have been documented after incidental or intentional intake of a few grams of KBrO 3 (De Angelo et al ., 2001)
The liver's high sensitivity to chemical injury has been linked to several characteristics, including its anatomical closeness to the blood supply and its involvement in xenobiotic biotransformation. As a result, preventing hepatocellular damage is extremely important. The mechanisms of cellular changes linked with KBrO 3 exposure in liver tissues are poorly understood. Toxic drug exposure impairs liver function, which is a direct result of alterations in the organ's histological features (Singh et al ., 2001). Because ingestion of KBrO 3 , like many harmful substances, might be unintentional, a suitable preventive/therapeutic agent for KBrO 3 therapy must be developed that is both inexpensive and widely available.
In several experimental models, medicinal herbs and plants are effective in treating oxidative stress-related disorders in various organs (Hussain et al ., 2014).
The bulbs of red onions (Allium cepa) contain a variety of phytochemicals, the majority of which are hydrocarbons and their derivatives, such as cepaenes and thiosulfinates (Dorsch and Wanger, 1999; Goldman et al., 1996). There are additional pigments like anthocyanins, as well as flavonoids like kaempferol and quercetin (Dorant et al., 1994).
Quercetin (3, 3, 4, 5, 7- pentahydroxyflavone), is a flavonoid found in fruits, seeds, nuts, tea, and red wine (Hollman et al ., 1999). Because of the discoveries that it inhibited numerous tumor-related processes such as oxidative stress, cellular proliferation, and metastasis, quercetin is gaining in popularity (Lugli et al ., 2009).
The red onion bulb has also been proven to be beneficial in the treatment of a variety of human and animal ailments, including cancer and diabetes, as well as being a natural antioxidant, antibacterial, and antiallergic resource ( l-Meleigy et al ., 2010)
The red onion has been shown to have antibacterial, antiparasitic, and antimycotic properties in several in vitro investigations (Rose et al ., 2005). The goal of this study was to see how a quercetin-rich aqueous extract of Allium cepa (A AC) affected potassium bromate hepatotoxicity in male Wistar rats
MATERIAL AND METHODS
Chemicals
Potassium bromate used was purchased from Sigma-Aldrich, St. Louis, MO. All the other chemicals used were of the highest purity grade.
Plant Materials
Allium cepa (red onion bulb) was procured from Bodija, Ibadan vegetable market, and authenticated at the Department of Botany, University of Ibadan, Nigeria. A specimen of the plant was then deposited in the herbarium of the Department of Botany bearing voucher specimen identification number UIH 22631.
Extraction of quercetin-rich constituents of Allium cepa
This extraction method was a modification of Nwaehujor et al. (2014). The onion bulb was washed with clean tap water followed by distilled water. The outer scaly leaves of the bulbs were manually removed and the fleshy part of the onion was rewashed with distilled water. xactly 10 g of the onion bulb was cut into small parts and crushed in 500 ml of water using a blending machine. The mixture was filtered through a muslin cloth and then Whatman no. 1 filter paper. The filtrate was evaporated at 45 0C to dry to produce crude quercetin and the dried residue was kept in a sterile bottle at 40C until required for use.
Experimental animals and treatment
Male Wistar rats weighing 100-120 g were obtained f rom the animal facilities of the Faculty of Basic Medical Sciences.
In the Department of Biochemistry, University of Ibad an, they were housed in clean polypropylene cages and kept in a room maintained at 24-280C with controlled cycles of 12 hours light and 12 hours dark and relative air humidity of 40-60%.
They were permitted to acclimatize for seven (7) day s before the initiation of the treatment and provided regul ar commercial pellets (Vita Feeds, Ibadan) and water ad libitum.
The experimental protocol was carried out following the National Institute of Health's guidelines for the care and welfare of research animals (NIH, 1985), as approved by the university institutional animal ethical committee of the Federal University of Agriculture, Makurdi, Nigeria, with reference number UI/FVM/17/22.
The experiment was carried out on forty male Wistar rats that were randomly separated into eight (8) groups of five (5) rats each and were treated differently depending on their group.
-
• Group A: Rats were administered distilled water only
-
• Group B: Rats were given KBrO 3 (dissolved in distilled water) at 30 mg/kg body weight
-
• Group C: Rats were administered quercetin at a dose of 50 mg/kg
-
• Group D: Rats were given A AC at a dose of 300 mg/kg
-
• Group : Rats were given A AC at a dose of 150 mg/kg body weight
-
• Group F: Rats were administered 30 mg/kg KBrO 3 + 300 mg/kg of A AC
-
• Group G: Rats were given 30 mg/kg of KBrO 3 + 150 mg/kg of A AC
-
• Group H: Rats were given 30 mg/kg KBrO 3 + 50
mg/kg of quercetin.
For 90 days, all treatments were given by gavage once a day on alternate days. The rats were monitored for indicators of toxicity and mortality throughout this time. A computerized weighing scale was used to record their body weights every week.
Termination of the experiment and harvest of tissues
The rats were sacrificed by cervical dislocation once the treatments were completed, and blood was taken in a plain sample bottle for biochemical assays. The serum was obtained by centrifuging the clotted blood sample at 3000 rpm for 5 minutes and storing it at 4 0C until it was utilized for analysis. ach experimental rat's liver was also thoroughly removed with dissecting scissors and forceps, cleaned in a 1.15 percent KCl solution, blotted with filter paper, and weighed. The liver was sectioned for histological analysis, and the remaining piece was homogenized using a Teflon homogenizer in an ice-cold homogenizing solution (0.1M phosphate buffer, pH 7.4).
To extract the post mitochondrial fractions, the homogenates were centrifuged at 10,000 rpm for 15 minutes at 4°C in a cool centrifuge. The liver biomarkers, oxidative stress, and antioxidant activity were all determined using this fraction and serum.
Biochemical analyses
A spectrophotometer was used to examine the biochemical parameters: the reduced glutathione (GSH) level was evaluated using the liver homogenate according to the technique of Jollow et al (1974). According to the approach described by Wright et al ., lipid peroxidation was measured by measuring the quantities of Malondialdehyde formed as a result of lipid peroxidation (1981). According to Mohandas et al ., the activity of the enzyme glutathione peroxidase (GPx) was determined (1985). Catalase (CAT) activity was determined according to the method of Claiborne (1985). The level of SOD activity was determined by the method of Misra and Fridovich (1972). Glutathione S-transferase (GST) was determined by the method of Habig et al . (1974) . The level of H 2 O 2 was assessed by H 2 O 2 -mediated horseradish peroxidase-dependent oxidation of phenol red by the method of Pick and
Keisari (1981). Nitrite assay was done using Griess reagent with some modifications of the method of Green et al . (1982).
Determination of γ GT was done using commercial kits (Rekon diagnostics. Pvt. Ltd).
Randox Laboratories Limited kits (Code: TP245 370343 C8 WJ 2 x 500ml) were used for determining the total protein level. Randox Laboratories Limited kits (Cat NO.:AS101 2 x 100ml) were used to determine AST level. Randox Laboratories Limited kits (AL 100 1 x 100ml) were used to determine ALT level.
The percentage of DNA fragmentation was determined by the method of Gibb et al . (1997).
Histological preparation
Immunohistochemical investigations of COX-2, Bcl-2, Caspase-3, and p53 expression level
For immunohistochemical investigations, 4 microns paraffin sections were stained immunohistochemically for visualizing cysteine-aspartic protease (caspase-3), BCL-2, P53, and COX-2 positive cells, using the suitable antibody in each staining time (Hsu et al ., 1981). The intensity of caspase-3, P53, BCL-3, and COX-2 expression in liver sections was quantified by using NIH image j software.
Statistical analysis
All results are expressed as mean ± standard deviation (Mean ± SD). The data were analyzed by oneway analysis of variance (ANOVA) with Graph pad prism version 8.01.
RESULTS
AEAC and Quercetin ameliorated potassium bromate (KBrO 3 )-induced hepatocellular toxicity in Wistar rats
Administration of KBrO3 only at a dose of 30 mg/kg body weight caused a significant (p < 0.05) increase in the activity of alanine amino transaminase (group B) as compared with the control given distilled water only (group A), as in Figure 1A. On the other hand, treatments of rats with KBrO3 and A AC at the dose of 300 or 150 mg/kg (Groups F and G respectively) led to a significant reduction in the KBrO3 -induced elevated enzyme activity when group B is compared with group F or G. The effect of the A AC on KBrO3 toxicity as measured by ALT activity is comparable with that of quercetin (group H, Figure 1A). The activities of other enzymes markers of liver toxicity, aspartate amino transaminase (Figure 1B) and γ-glutamyl transferase (Figure 1C) follow patterns similar to that of alanine amino transaminase activity in the control and experimental animals. In all cases, the higher dose of A AC (300 mg/kg) seemed to be more effective in ameliorating the KBrO3 -induced activities of the enzymes (Figures 1 A-C). Histological analysis of the liver of the experimental rats corroborated the observation made with the enzyme indices of hepatotoxicity (Figure 2)
Enhancement of the cellular antioxidant defense by AEAC and quercetin in the liver tissue of rats treated with KBrO 3
Administration of KBrO 3 alone to rats caused significant (p<0.05) changes in the activities of major detoxifying enzymes like superoxide dismutase (SOD), catalase, glutathione S-transferase (GST), and glutathione peroxidase (GPx) and the level of cellular reduced glutathione (GSH) in the hepatic tissues when compared with the control given distilled water only (Figure 3). However, treatment of rats with KBrO 3 and A AC (at 300mg/kg or 150 mg/kg) or quercetin at 50 mg/kg (Group F, G, and H respectively) caused a restoration of the activities of the enzymic and non-enzymic antioxidant defense components to levels significantly different from the group treated with KBrO 3 (Group B) only and similar to or not significant (p>0.05) different from the negative control (Group A). It was observed that the groups of rats given A AC alone (Groups D and ) or quercetin alone (Group C) have a significant (p<0.05) increase in the level of GSH and activities of GST and GPx compared with the control,
Group A (Figure 3). This, therefore, confirmed the tissue antioxidant system enhancement effects of the extracts.
Mitigation of the KBrO 3 -induced inflammatory biomarkers (Malondialdehyde and Nitric Oxide) by AEAC and/or Quercetin
Administration of KBrO 3 (group B) caused a significant (p<0.05) increase in Malondialdehyde (MDA) levels when compared with the control group (group A), given distilled water as shown in Figure 4. The level of MDA was significantly (p<0.05) lower in the groups given KBrO 3 and A AC or quercetin compared with the group given KBrO 3 alone (Group F/G/H versus Group B). Assessment of the effects of KBrO 3 on hepatic Nitric Oxide (NO) levels in the treated rats revealed trends similar to the observations made with MDA levels above (Figure 4).
Changes in the KBrO 3 -induced Liver injury in rats
Treatment of rats with 30 mg/kg body weight KBrO 3 alone resulted in severe portal congestion, with moderate periportal vacuolar degeneration due to disruption of hepatic architecture, however, rats in groups administered distilled, quercetin, and A AC alone showed normal hepatocellular architecture. In the groups co-administered KBrO 3 + A AC at the graded doses, there was a significant reduction in the degree of the hepatic architecture disruption showing minimal portal congestion and mild periportal cellular infiltration.
Variation of KBrO 3 -induced DNA fragmentation in the liver cells of rats
There was significant (p<0.05) DNA fragmentation in the liver homogenate of rats treated with KBrO 3 (Group B) alone when compared with those given distilled water only (Group A) (as shown in Figure 5). Quercetin and A AC abrogated the effect of KBrO 3 in Groups F, G and H treated with KBrO 3 and A AC or quercetin (Figure 5).
Changes in the expression of apoptotic-related proteins in the rats
Cox-2, p53, and caspase-3 genes were expressed at very low levels in the hepatic tissues of the control rats given distilled water only (Figure 6 panel A). However, in the group of rats treated with KBrO 3 only (Group B), the corresponding tissue sections showed intense staining for cox-2, p53, and caspase-3 expressions (Figure 6 panel B). Treatment with A AC or quercetin ameliorated the KBrO 3 -induced expression of cox-2 and caspase-3 and p53 genes in the liver cells in the groups treated with KBrO 3 and A AC or quercetin (Groups F, G, and H) compared with those treated with KBrO 3 only (Group B). On the other hand, BCL-2 protein was mildly expressed in group A and also in the KBrO 3 -treated, group B. There was a significant increase in expression of the BCL-2 protein in the groups treated with KBrO 3 and A AC or quercetin.
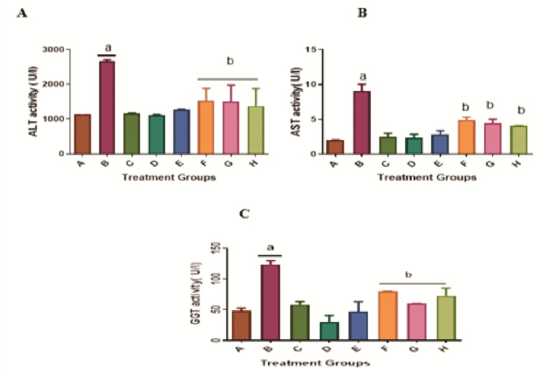
Figure 1: ffect of A AC on serum alanine, serum aspartate Aminotransferase, and γ-glutamyl transferase activities in male Wistar rats treated with KBrO 3 . Values are means + SD (n=5). a= significantly different compared with negative control, group A, and b= significantly different compared with positive control group B, treated with KBrO 3 only (p<0.05). Groups A Received distilled water, B: 30 mg/kg KBrO 3 , C: 50 mg/kg Quercetin, D: 300 mg/kg A AC, : 150 mg/kg A AC, F: KBrO 3 + 300 mg/kg A AC, G: KBrO 3 + 150 mg/kg A AC and H: KBrO 3 + Quercetin.
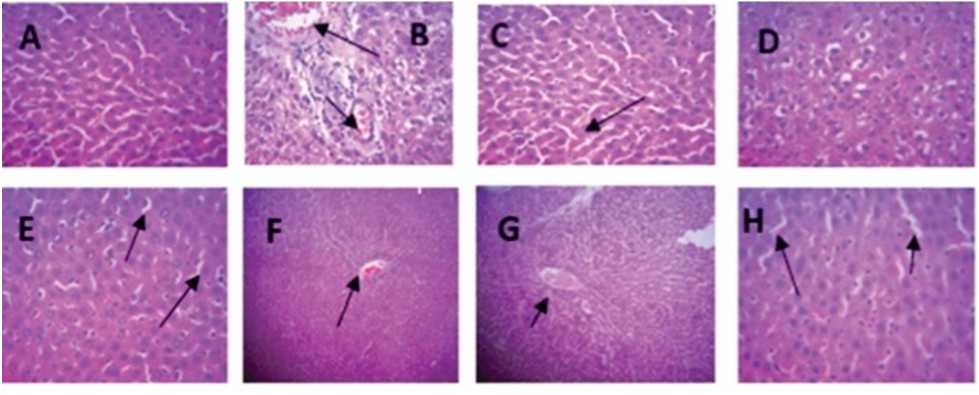
Figure 2: Selected photomicrographs of liver tissues sections (×100) in male Wistar rats treated with KBrO 3 and/or A AC: There are no lesions in the liver of the control group. (A) but severe portal congestion, with moderate periportal cellular infiltration in the group, treated with only (B). There is a mild to moderate periportal vacuolar degeneration of hepatocytes in the group treated with Quercetin only (C) while treatment with A AC at 300 and 150 mg/kg produced no visible lesion (D and ). Treatment with KBrO 3 and A AC at 300 mg/kg (F) shows mild diffuse vacuolar degeneration whereas mild diffuse sinusoidal congestion was observed in the livers of rats that received KBrO 3 and A AC at 150 mg/kg (G). There is mild central venous and sinusoidal congestion in the group treated with KBrO 3 and Quercetin (H).
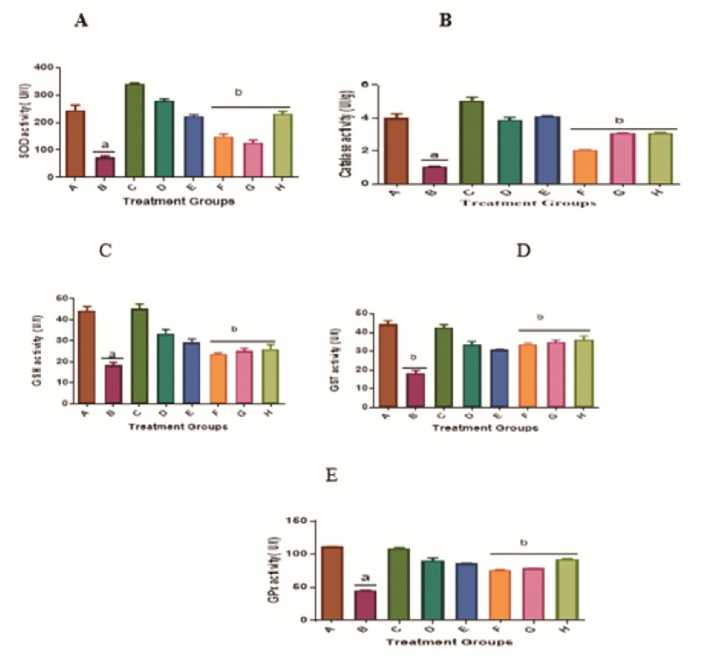
Figure 3: nhancement of the cellular antioxidant defense by A AC in the liver tissue of rats treated with KBrO 3 . Values are means + SD (n=5). a= significantly different compared with negative control, group A, and b= significantly different compared with positive control group B, treated with KBrO 3 only (p<0.05). Groups A received distilled water, B: 30 mg/kg KBrO 3 , C: 50 mg/kg Quercetin, D: 300 mg/kg A AC, : 150 mg/kg A AC, F: KBrO 3 + 300 mg/kg A AC, G: KBrO 3 + 150 mg/kg A AC and H: KBrO 3 + Quercetin.
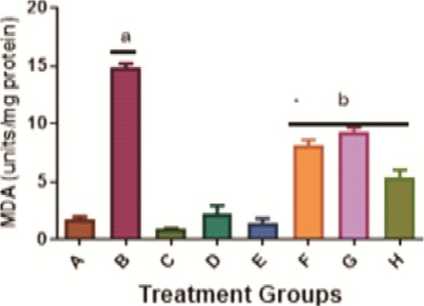
Figure 4: Mitigation of the KBrO 3 -induced inflammatory biomarkers (Malondialdehyde and Nitric Oxide) by A AC levels in the liver of male Wistar rats. Values are means + SD (n=5). a= significantly different compared with negative control, group A (p<0.05). b= significantly different compared with positive control group B, treated with KBrO 3 only (p<0.05). Groups A received distilled water, B: 30 mg/kg KBrO 3 , C: 50 mg/kg Quercetin, D: 300 mg/kg A AC, : 150 mg/kg A AC, F: KBrO 3 + 300 mg/kg A AC, G: KBrO 3 + 150 mg/kg A AC and H: KBrO 3 + Quercetin.
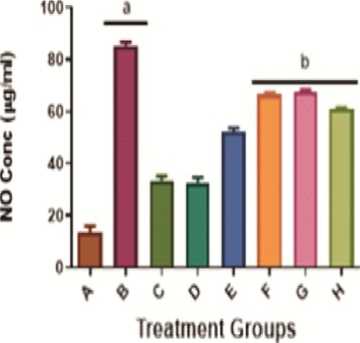
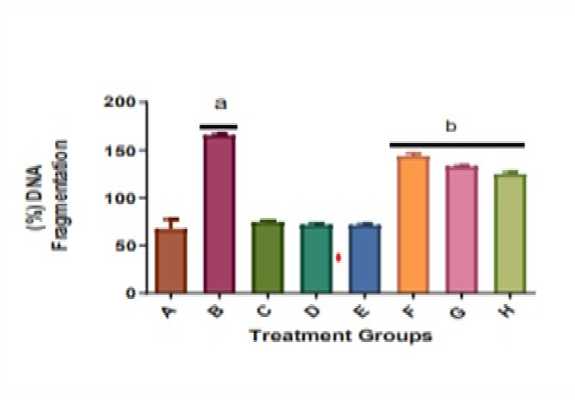
Figure 5: ffect of A AC on the proportion of DNA fragmentation in the liver of male Wistar rats treated with potassium bromate. Values are means + SD (n=5). a= significantly different compared with negative control, group A, and b= significantly different compared with the positive control group B, treated with KBrO 3 only (p<0.05). Groups A received distilled water, B: 30 mg/kg KBrO 3 , C: 50 mg/kg Quercetin, D: 300 mg/kg A AC, : 150 mg/kg A AC, F: KBrO 3 + 300 mg/kg A AC, G: KBrO 3 + 150 mg/kg A AC and H: KBrO 3 + Quercetin.
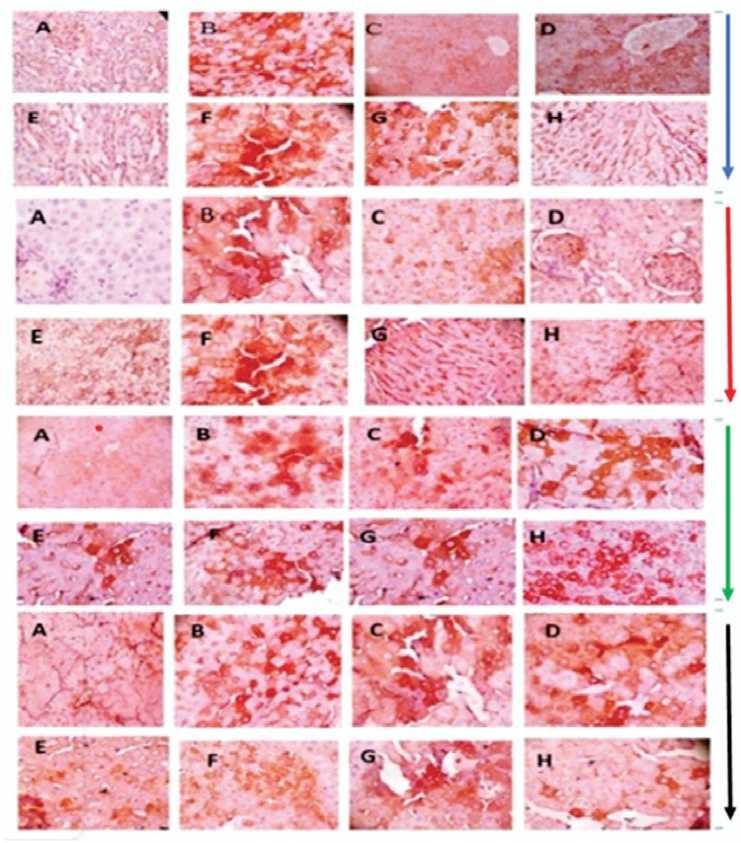
Figure 4: Representative photomicrographs of liver tissues comparing the expression levels of COX-2 (Blue arrow), P53 (Red arrow), Caspase-3 (Green arrow), and BCL-2 (Black arrow) proteins in male Wistar rats treated with potassium bromate and/or A AC. There is a negligible or mild expression of Cox-2, P53, Caspase-3, and BCL-2 in the negative control group (A), quercetin treated group (C) and A AC treated groups (D and ); severe expression in KBrO 3 treated group (B); There is a moderate expression in the group treated with KBrO 3 and A AC or quercetin (F, G, and H).
DISCUSSION
According to research, chemical toxicity is connected to an increase in reactive oxygen species (ROS) production, which causes oxidative stress in cells (Khan et al., 2001). Superoxide anions and their derivatives (hydroxyl radical), the most common ROS produced in living cells are extremely reactive, causing peroxidation of cell membrane lipids resulting in membrane damage and cellular content leakage. Lipid peroxidation and associated membrane damage have been linked to the etiology of a variety of diseases, including hepatocellular carcinoma.
In the current study, KBrO3 administration caused a significant (p>0.05) increase in the transaminases (ALT and AST) and γ-glutamyl transferase (GT) activities when compared to the control group. These findings are consistent with previous research that found an increase in rat alanine and aspartate amino transaminases in the rats treated with KBrO3 (Khan et al., 2012; Dimkpa et al., 2013). The alterations of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities are biochemical indicators of hepatocellular injuries (Atawodi et al., 2014). When compared to the group given just KBrO3, treatment of KBrO3 with A AC at doses of 300 mg/kg or 150 mg/kg resulted in lower ALT, AST, and GT activity, suggesting that the extract can counteract the toxic effects of KBrO3 on the liver.
The liver tissue photomicrographs are commonly used to evaluate the extent of tissue damage, by the microscopic study of their structural architectures. Thus, histopathological findings in tissue sections of animals are commonly used to validate the biochemical findings of a study (Al-Attar et al ., 2015). Whereas there were no apparent abnormalities in the liver architecture of rats in the groups that were administered distilled water and pure quercetin alone, the group treated with KBrO 3 alone had portal congestion and mild periportal vacuolar degeneration due to disturbance of hepatic architecture. The harmful effects of KBrO 3 that are present here are comparable to those reported by other researchers (Dimkpa et al ., 2013; Abuelgasin et al ., 2008; Akanji et al ., 2008). A combined treatment of A AC at doses of 300 mg/kg or 150 mg/kg, as well as quercetin with KBrO 3 , showed a diminution in the hepatic architectural disturbance, with low portal congestion and modest periportal cellular infiltration. This indicates that the A AC has an abrogative effect against KBrO 3 -mediated hepatotoxicity in rats. Previous studies have shown that plant extracts could prevent pathological changes in liver tissues of animals and every so often may reverse pathological changes already induced by toxicants to animal tissues (Adejor et al ., 2003; Alqasoumi, 2014).
Different exogenous anxieties, including KBrO 3 , can cause oxidative pressure. Past exploratory proof has exhibited that KBrO 3 incites oxidative pressure (Delker et al ., 2006).
Furthermore, when the rats were administered KBrO 3 alone, the levels of malondialdehyde (MDA) and nitric oxide (NO) were significantly (p>0.05) increased compared to the control group. This finding was in consonance with the report of Raouia et al ., (2021). MDA and NO levels were ameliorated by A AC treatment in a dose-dependent manner. Additionally, as compared to control, KBrO 3 administration depleted cellular reduced glutathione (GSH) levels.
The CAT, SOD, GST, GSH, and GPX represent an armoury of antioxidants usually produce by the body to neutralize or 'mop up' free radicals that can harm the cells. They, therefore, defend the cells against oxidative stress. The ability of the body to produce these antioxidants is controlled by genetic makeup and influenced by exposure to environmental factors such as chemicals as well as dietary intakes (Halliwel, 1999).
KBrO 3 treatment caused a significant (p>0.05) reduction in the activities of antioxidant enzymes: SOD, catalase, GST, and GPX in rats, whereas the administration of A AC along with KBrO 3 significantly (p>0.05) restored GSH and antioxidant enzyme activities (SOD, catalase, GST, and GPX) near normal in a dosedependent manner, implying that the A AC increased the production of cellular antioxidants. Consequently, the A AC replaced the GSH levels and the antioxidant enzymes that were depleted during the neutralization of oxidative stress caused by KBrO 3 . As a result, the extract may have acted as a hepatoprotective agent against KBrO 3 by restoring GSH levels, GST activity, and the glutathione redox cycle enzymes in hepatic tissues as shown. This is in keeping with Watanabe et al ., prior’s recommendation (2004).
The observation made with the A AC may be linked to the presence of reputable antioxidants such as quercetin in the extract. Quercetin has been shown to possess potent antioxidant properties (Read, 1995; Orsolic et al ., 2004). Moreover, the findings with the extract here, as a protective agents and antioxidants system restorer against KBrO 3 -induced toxicity and oxidative stress, are similar to the result obtained by administration of pure quercetin only.
In likewise manner, the administration of KBrO 3 caused a significant increase in the percentage of DNA fragmentation in the liver issues. The increased proportion of DNA fragmentation in the liver has been linked to oxidative DNA damages (Khan and Sultana, 2005). The findings that groups of animals treated with A AC or quercetin along with KBrO 3 recorded a lower percentage of DNA fragmentation than those treated with KBrO 3 alone support the fact that the extract contains bioactive compounds which play a vital role in the inhibition of DNA fragmentation or DNA repair or both by abrogating the injurious effect of the KBrO 3 on the hepatocytes.
The expression of apoptotic-related proteins is modulated by toxicants that affect cellular redox status such as KBrO 3 (Corsby et al ., 2000). Changes in the expression of apoptotic-associated proteins including caspase-3, BCL-2, and p53 might be linked to genome instability. The p53 gene, in particular, is a tumor suppressor gene that regulates the cell cycle, apoptosis, and DNA repair (Bowen et al ., 2006).
In this work, we found that the group treated with KBrO 3 only had greater P53, Caspase-3, COX-2, and lower BCL-2 expression than the control group. However, co-administration of KBrO 3 with A AC and/or quercetin suppressed the high production of P53 and Caspase-3 and raised the expression of the BCL-2 protein. It is now well known that apoptosis modulation is a part of several phases of oncogenesis, and that BCL-2, anti- and pro-apoptotic BCL-2 family members, as well as P53 and Caspase-3, may all play key roles. The impact of transcription factors controlling cellular proliferation is similar to the apoptotic/anti-apoptotic system. COX-2 inhibition appears to be a promising target for the prevention of a variety of proliferative disorders, including cancer (Konurek et al ., 2005). COX-2 is controlled by the transcription factor NFkB, which creates a favorable environment for tumor growth. Following exposure to KBrO 3 , the expression of COX-2 in liver tissues increased in the current investigation. Inhibition of KBrO 3 -induced COX-2 expression in the hepatic tissues was noticed following treatment of KBrO 3 with graded dosages of A AC or quercetin.
As a result of the preceding, it may be inferred that A AC administration prevents KBrO 3 -induced hepatocellular damage in rats. Hepatic architectural damage caused by KBrO 3 exposure was significantly reduced by the extract. In addition, in treated rats, the A AC controls the production of transcription factors and proteins involved in the cell cycle and apoptosis pathways and increases antioxidant defenses against KBrO 3 -induced distortion. The A AC's capacity to limit the indicators of inflammation, oxidative stress, and apoptotic regulation in Wistar rats suggests it might be a promising option for chemoprevention of hepatocellular damage.
ACKNOWLEDGEMENTS
We acknowledge the support of the technical staff in our department, Mr. ric Saboh.
CONFLICTS OF INTEREST
The author declare that they have no potential conflicts of interest.
References Ameliorative potential of Allium cepa or quercetin on the histology, immunohistochemistry and oxidative stress biomarkers parameters against potassium bromate-induced hepatotoxicity in adult male rats
- Abuelgasim AL, Omer R and Elmahdi B. (2008) Serobiochemical Effects of Potassium Bromate on Wistar Albino Rats. American J of Food Tech; 3: 303-309.
- Adejor EB, Ameh DA, James BD, Owolabi OA, Ndidi US. (2003) Effects of Garcinia kola biflavonoid fractions on serum lipid profile and kidney function parameters in hyperlipidemic rats. Clinical Phytoscience; 2:19. https://doi.org/ 10.1186/s40816-016-0033-4.
- Ahlborn GJ, Delker DA, Roop BC, Geter DR, Allen JW, DeAngelo AB and Winnik WM. (2009) Early alterations in protein and gene expression in rat kidneys following bromate exposure. Food Chem Toxicol 47:1154-1160.
- Akanji MA, Nafiu MO and Yakubu MT. (2008) Enzyme activities and histopathology of selected Tissues in rats treated with potassium bromate. African J Biomed Res; 11:87-95.
- Al-Attar MA, Alrobai AA, Almalki AD. (2015) Protective effect of olive juniper leaves extracts on nephrotoxicity induced by thioacetamide in male mice. Saudi J Biol Sci; 24(1):15-22. https://doi.org/10.1016Zj.sjbs. 2015.08.013.
- Alqasoumi SI. (2014) Evaluation of the hepatoprotective and nephroprotective activities of Scrophularia hypericifolia growing in Saudi Arabia. Saudi Pharmaceutical Journal;22:258-63.
- Atawodi SE, Yakubu OE, Liman ML, Iliemene DU. (2014) Effect of methanolic extract of Tetrapleura tetraptera (Schum and Thonn) Taub leaves on hyperglycemia and indices of diabetic complications in alloxan-induced diabetic rats. Asian Pac J Trop Biomed.; 4(4):272-8.
- Bowen J, Gibson R, Cummins A and Keefe D. (2006) Intestinal mucositis: the role of the Bcl-2 family, p53 and caspases in chemotherapy-induced damage. Support Care Cancer; 14:713-731.
- Claiborne A. (1985) Catalase activity. In: Greenwald RA (ed) CRC handbook of methods in oxygen radical research. CRC Press, Boca Raton,, pp.283-284.
- Corsby LM, Hyder KS, DeAngelo AB, Kepler TB, Gaskil B, Benavides GR, Yoon L and Morgan KT. (2000) Morphological analysis correlates with gene expression changes in cultured F344 rat mesothelial cells. Toxicol Appl Pharmacol; 169:205-221
- De Angelo AB, George MH, Kilburn SR, Moore TM and Wolf DC. (1998) Carcinogenicity of potassium bromate administered in the drinking water to male B6C3F1 mice and F344/Nrats. Toxicologic Pathol 26:587-594.
- Delker D, Hatch G, Allen J, Crissman B, George M, Geter D, Kilburn S, Moore T, Nelson G, Roop B. (2006) Molecular biomarkers of oxidative stress associated with bromate carcinogenicity. Toxicology; 221, 158e165.
- Dimkpa U, Ukoha U, Anyabolu E, Uchefuna R, Anikeh L, Oji O, Besong E and Emenjo O. (2013) Hepatotoxic effects of potassium bromate on adult Wistar rats. J Biol Agri Healthcare; 3:111-115.
- Dorant E, van Din Brandt P and Goldbohm R.A. (1994) prospective cohort study on Allium vegetable consumption, garlic supplement use, and the risk of lung carcinoma in the Netherlands. Cancer Res 54: 6148-6153.
- Dorsch W and Wagner H. (1991) New antiasthmatic drugs from traditional medicine. Int Arch Allergy Appl Immunol 94:262- 265.
- El-Meleigy MA, Ahmed ME, Arafa RA, Ebrahim NA and El-Kholany EE. (2010) Cytotoxicity of four essential oils on some human and bacterial cells. J Appl Sci Environ Sanit; 5:143-159.
- Gibb RI, Taylor DD, Wan T, O'Connor DM, Doeriog DL and GerHel-Taylor T. (1997) Apoptosis as a measure of chemosensitivity to cisplatin and taxol therapy in ovarian cancer cell lines. Gynecolog Oncol; 65:13-22..
- Goldman IL, Kopelberg M, Devaene JE and Schwartz BS. (1996) Antiplatelet activity in onion is sulfur dependent. Thrombosis Haemostasis 76:450-452.
- Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS and Tannenbaum SR. (1982) Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Anal Biochem; 126:131-138
- Habig WH, Pabst MJ and Jakoby WB. (1974) Glutathione-S-transferases: the first enzymatic step in mercapturic acid formation. J Biol Chem; 249:7130-7139.
- Halliwell B. (1999) Establishing the significance and optimal intake of dietary antioxidants: the biomarker concept. Nutr Rev; 57:104-113.
- Hollman PCH, Bijsman MNCP, van Gameren Y, Cnossen EPJ, de Vries JHM, Katan MB. (1999) The sugar moiety is a major determinant of the absorption of dietary flavonoids glycosides in man. Free Radic Res; 31:569-573
- Hsu, S., Raine, L., and Fanger, H. (1981) A comparative study of the peroxidase-anti peroxidase method and an avidin-biotin complex method for studying polypeptide hormones with radioimmunoas? say antibodies. Am. J. Clin. Pathol. 75: 734-738
- Hussain L, Ikram J, Rehman K, Tariq M, Ibrahim M and Hamid MS. (2014) Hepatoprotective effects of Malvasylvestris L. against paracetamol-induced hepatotoxicity. Turk J Biol 38:396-402.
- International Agency for Research on Cancer (IARC). Summaries & Evaluations Ochratoxin A (Group 2B), http://www.inchem.org/documents/iarc/vol56/13-ochra.html (2003, accessed on 25 April 2019).
- Jaloszynski P, Murata S, Shinkai Y, Takahashi S, Kumagai Y, Nishimura S and Yamamoto M. (2007) Dysfunction of Nrf2 decreases KBrO3-induced oxidative DNA damage in Ogg1-null mice. Biochem Biophy Res Comm 364:966-971.
- Jollow DJ, Mitchell JR, Zampaglione N and Gillette JR. (1974) Bromobenzene induced liver necrosis: protective role of glutathione and evidence for 3, 4-bromobenzene oxide as the hepatotoxic metabolite. Pharmacology; 11:151-169
- Khan N and Sultana S. (2005) Inhibition of potassium bromate-induced renal oxidative stress and hyperproliferative response by Nymphaea alba in Wistar rats. J Enzyme Inhib Med Chem; 20:275283.
- Khan N, Sharma S, Alam A, Saleem M and Sultana S. (2001) Tephrosia purpurea ameliorates N-diethylnitrosamine and potassium bromate-mediated renal oxidative stress and toxicity in Wistar rats. Pharmacol Toxicol; 88:294-299.
- Khan RA, Khan MR and Sahreen S. (2012) Protective effects of rutin against potassium bromate induced nephrotoxicity in rats. BMC Complement Altern Med; 12:204. https://doi.org/10.1186/1472-6882-12-204.
- Konturek PC, Kania J, Burnat G, Hahn EG and Konturek SJ. (2005) Prostaglandins as mediators of COX-2-derived carcinogenesis in the gastrointestinal tract. J Physiol Pharmacol; 56: 57-73
- Kurokawa Y, Maekawa A, Takahashi M and Hayashi Y. (1990) Toxicity and carcinogenicity of potassium bromate-a new renal carcinogen. Environ Perspect 87:309-35.
- Lillie RD, Fullmer HM (1976) Histopathologic technic and practical histochemistry, 4th edn. McGraw-Hill, New York.
- Liu Y and Mou S. (2004) Determination of bromate and chlorinated haloacetic acids in bottled drinking water with chromatographic methods. Chemosphere 55:1253-1258.
- Lugli E, Ferraresi R, Roat E, Troiano L, Pinti M, Nasi M, Nemes E, Bertoncelli L., Gibellini L, Salomoni P, Cooper EL and Cossarizza A. (2009) Quercetin inhibits lymphocyte activation and proliferation without inducing apoptosis in peripheral mononuclear cells. Leuk Res; 33:140-150.
- McDorman KS, Pachkowski BF, Nakamura J, Wolf DC and Swenberg JA. (2005) Oxidative DNA damage from potassium bromate exposure in Long-Evans rats is not enhanced by a mixture of drinking water disinfection byproducts. Chem Biol Interact 152:107-117.
- Misra HP and Fridovich I. (1972) The role of superoxide anion in the auto-oxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem; 247:3170-3175.
- Mohandas M, Marshall JJ, Duggin GG, Horvath JS and Tiller D. (1984) Differential distribution of glutathione and glutathione related enzymes in rabbit kidney. Cancer Res.; 44:5086-509.
- National Institute of Health (N.I.H). (1985) Guide for the care and use of laboratory animals. DHEW publication; Office of Science and Health Reports; Bethsaida; U.S.A.
- Nwaehujor CO, Igile GO, Nwinyi FC and Ode JO. (2014) The Protective Role of Extract of Allium cepa Linn. (Liliaceae) (Red Onion) Bulb in Artesunate-induced Testicular Damage in Male Wistar Rats. British J Pharm Res; 4:1715-1724.
- Orsolic N, Knezevic AH, Sver L, Terzic S and Basic I. (2004) Immunomodulatory and antimetastatic action of propolis and related polyphenolic compounds. J Ethnopharmacol; 94:307-315.
- Pick A and Keisari Y. (1981) Superoxide anion and H2O2 production by chemically elicited peritoneal macrophages-induction by multiple nonphagocytic stimulus. Cell Immun.; 59:301-308.
- Raouia D, Maryem BS, Rim M, Serrial H, Khaled Z, Kamiliar K. (2021) Protective effect of Urtica dioica in induced neurobehavioral changes, nephrotoxicity, and hepatotoxicity after chronic exposure to potassium bromate in rats. Environmental pollution; 287, 117657. https://doi.org/10.1016/j.envpol.2021.117657.
- Read MA. (1995) Flavonoids: naturally occurring antiinflammatory agents. Am J Pathol; 147:235-237
- Rose P, Whiteman M, Moore P and Zhu Y. (2005) Bioactive S-alk(en)yl cysteine sulfoxide metabolites in the genus Allium: the chemistry of potential therapeutic agents. Natural Product Res; 22:351368.
- Singh NS, Vats P, Suri S, Shyam R, Kumria MML, Ranganathan S and Sridharan K. (2001) Effect of an antidiabetic extract of Catharanthus roseus on enzymic activities in streptozotocin-induced diabetic rats. J Ethnopharmacol 76:269-277.
- Watanabe S, Tajima Y, Yamaguchi T, Fukui T. (2004) Potassium bromate-induced hyperuricemia stimulates acute kidney damage and oxidative stress. J Health Sci; 50:647-653.
- Wright JR, Colby HD and Miles PR. (1981) Cytosolic factors which affect microsomal lipid peroxidation in lung and liver. Arch Biochem Biophys; 206: 296304


