Аналоги соматостатина, меченные радионуклидами, для терапии онкологических заболеваний. Обзор
Автор: Тищенко В.К., Петриев В.М., Крылов В.В., Власова О.П., Шегай П.В., Иванов С.А., Каприн А.Д.
Рубрика: Научные статьи
Статья в выпуске: 2 т.31, 2022 года.
Бесплатный доступ
В настоящее время основным требованием к радионуклидной терапии онкологических заболеваний является специфическое воздействие на опухолевые клетки с минимальной токсичностью в отношении здоровых тканей. Молекулярная основа селективной противоопухолевой терапии обеспечивается сверхэкспрессией соматостатиновых рецепторов (ССТр) на клеточной мембране различных опухолей и их метастазов. Природный соматостатин не может быть использован в качестве векторной молекулы для доставки радионуклидов ввиду короткого периода полураспада в крови (1-3 мин). Синтетические аналоги соматостатина пептидной природы, меченные терапевтическими радионуклидами (радиопептиды), также обладают высоким сродством с ССТр и улучшенной фармакокинетикой по сравнению с соматостатином, и потому представляют огромный интерес для направленной терапии рака, именуемой пептид-рецепторной радионуклидной терапией (ПРРТ). В обзоре представлена информация о наиболее значимых на сегодняшний день аналогах соматостатина, меченных радионуклидами 111In, 90Y, 177Lu, для терапии опухолей со сверхэкспрессией ССТр. Приведены данные об эффективности лечения и профиле токсичности ПРРТ, проводимой с различными поколениями радиофармацевтических лекарственных препаратов (РФЛП), нацеленных на ССТр, включая рандомизированное контролируемое исследование NETTER-1. Кроме того, обсуждаются некоторые стратегии оптимизации ПРРТ, такие как тандемная терапия, внутриартериальный способ введения РФЛП, модификация препаратов для оптимизации фармакокинетики, а также разработка новых препаратов, содержащих a-излучающие радионуклиды, или являющихся антагонистами ССТр.
Аналоги соматостатина, радиопептиды, пептид-рецепторная радионуклидная терапия, соматостатиновые рецепторы, производные октреотида, радиофармацевтический лекарственный препарат (рфлп), dotatoc, dotatate, нейроэндокринные опухоли
Короткий адрес: https://sciup.org/170195069
IDR: 170195069 | УДК: 616-006.04-085.849.2.03:577.175.85 | DOI: 10.21870/0131-3878-2022-31-2-76-96
Radiolabeled somatostatin analogs for cancer treatment. Review
Currently, a specific action on tumor cells with minimal toxicity to healthy tissues is the main requirement for radionuclide therapy of cancer. The molecular target of selective antitumor therapy is determined by somatostatin receptors (SSTR) overexpression in various tumors and its metastases. Natural somatostatin cannot be used as a vector molecule for radionuclide delivery due to its short half-life in blood (1-3 min). Synthetic peptide somatostatin analogs labeled with therapeutic radionuclides (radiopeptides) also have high affinity to SSTR and better pharmacokinetics compared to somatostatin and therefore they are of great interest for targeted cancer therapy, also called peptide-receptor radionuclide therapy (PRRT). The data about the most important to date somatostatin analogs labeled with 111In, 90Y, 177Lu radionuclides for therapy of tumors overexpressing SSTR is presented. The results of treatment efficacy and toxicity profile of PRRT, which is administered with various generations of targeting SSTR radiopharmaceuticals, including the randomized controlled trial NETTER-1, is reviewed. In addition, some strategies for optimization of PRRT such as tandem therapy, intra-arterial administration of radiopharmaceuticals, their modification for better pharmacokinetic properties, and the development of new compounds containing a-emitting radionuclides or SSTR antagonist analogs are discussed.
Текст научной статьи Аналоги соматостатина, меченные радионуклидами, для терапии онкологических заболеваний. Обзор
Онкологические заболевания представляют серьёзную медицинскую проблему из-за неуклонного роста заболеваемости и смертности. По данным Глобальной онкологической обсерватории (GCO) в 2020 г. было зарегистрировано более 19 млн новых случаев рака, а умерло – 9,96 млн больных [1]. По прогнозам к 2040 г. смертность от онкологических заболеваний увеличится до 16,3 млн случаев в год [1].
За последние 20 лет в ядерной медицине достигнут существенный прогресс, заключающийся в разработке новых радиофармацевтических лекарственных препаратов (РФЛП) для диагностики и терапии различных заболеваний. В настоящее время большой интерес вызывают пептиды, обладающие избирательным распределением и высоким сродством к специфическим рецепторам, сверхэкспрессированным в опухолевых клетках. Это позволяет использовать РФЛП пептидной природы в ядерной онкологии как для диагностической визуализации, так и таргетной радионуклидной терапии опухолей. Пептид-рецепторная радионуклидная терапия (ПРРТ)
Тищенко В.К. – вед. науч. сотр., д.б.н.; Петриев В.М.* – зав. лаб., д.б.н., проф. НИЯУ МИФИ; Крылов В.В. – зав. отд., д.м.н.; Власова О.П. – вед. науч. сотр., к.б.н.; Иванов С.А. – директор, д.м.н., проф. РАН. МРНЦ им. А.Ф. Цыба – филиал ФГБУ «НМИЦ радиологии» Минздрава России. Шегай П.В. – зам. ген. директора, к.м.н.; Каприн А.Д. – ген. директор, акад. РАН, д.м.н., проф. ФГБУ «НМИЦ радиологии» Минздрава России.
предполагает использование радиопептидов для направленного и тканеспецифичного облучения опухолевых клеток, что позволяет обеспечивать более эффективное лечение диссеминированного рака с минимальными побочными эффектами.
Растущее число свидетельств сверхэкспрессии соматостатиновых рецепторов (ССТр) различными опухолями вызвало огромный интерес к разработке специфических пептидов для их диагностики и терапии [2]. Из-за короткого биологического периода полураспада природного соматостатина (ССТ) были разработаны более стабильные синтетические аналоги ССТ. Они не подвергаются ферментативному гидролизу, имеют благоприятную фармакокинетику, накапливаются в опухоли в высоких концентрациях и быстро выводятся из крови и нецелевых тканей, обеспечивая тем самым тканеспецифическое облучение опухоли [3]. Более того, пептиды способны проникать в опухоль и связываться с соответствующими рецепторами быстрее, чем моноклональные антитела [3].
Целью данного обзора является обобщение имеющихся на сегодняшний день данных о меченных радионуклидами аналогах ССТ, предназначенных для ПРРТ опухолей со сверхэкспрессией ССТр.
Соматостатин – полипептидный гормон, впервые идентифицированный в 1973 г. [4]. Известно, что он ингибирует различные метаболические процессы и подавляет секрецию многих гормонов. Существует две изоформы этого гормона: ССТ-14 и ССТ-28, содержащие 14 или 28 аминокислотных остатков соответственно. ССТ-14 преобладает в центральной нервной системе, а ССТ-28 – в желудочно-кишечном тракте [4].
Разнообразные биологические эффекты ССТ опосредуются через семейство ССТр, наличие которых было обнаружено во многих тканях человеческого организма, опухолях и перитумо-ральных сосудах. В начале 1990-х годов, одновременно с исследованиями связывающих свойств и механизмов действия ССТ, были идентифицированы пять подтипов ССТр (ССТр1-ССТр5), каждый из которых обладает специфическими фармакологическими и физиологическими свойствами [4].
Экспрессия подтипов ССТр на опухолевых клетках зависит от типа опухоли, однако преимущественно встречаются ССТр2 [5]. Самая высокая частота экспрессии ССТр отмечена в группе нейроэндокринных опухолей (НЭО) [2]. Например, в гастроэнтеропанкреатических НЭО ССТр присутствуют в 80-100% случаев, за исключением инсулина, которые имеют более низкую частоту экспрессии ССТр – от 50 до 70% [2]. Отмечена широкая вариабельность плотности ССТр и экспрессии их подтипов в таких НЭО, как аденома гипофиза, феохромоцитома, параганглиома, карциноиды лёгких, мелкоклеточные опухоли лёгких, медуллярная карцинома щитовидной железы и нейробластома [6]. ССТр присутствуют в клетках многих солидных и гематологических новообразований, например, менингиомах, глиомах, лимфомах, раке молочной железы, лёгких, почки, панкреатобилиарного тракта, печени, яичников, предстательной железы, колоректального рака [2, 5, 6]. Поэтому ССТр представляют собой идеальную мишень для таргетной радионуклидной терапии таких опухолей.
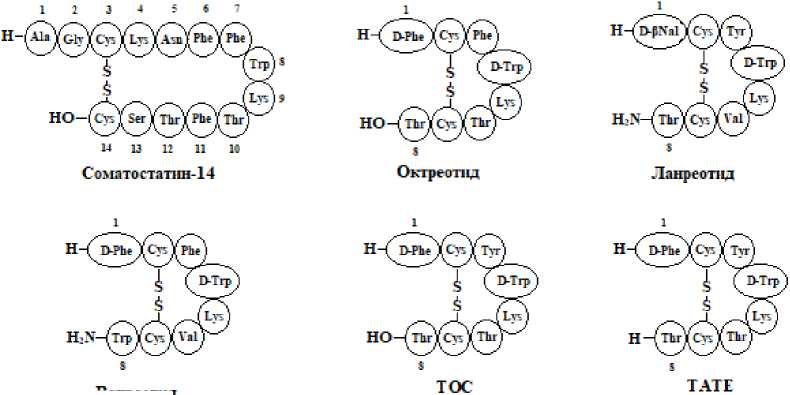
Природный ССТ обладает коротким периодом полураспада в кровотоке (1-3 мин), быстро разрушаясь под действием пептидаз, что затрудняет его практическое использование в качестве векторной молекулы для доставки радионуклидов. Поэтому были разработаны синтетические производные ССТ с пролонгированным действием. Все аналоги ССТ представляют собой циклические октапептиды (реже – гексапептиды) с периодом полувыведения от 1,5 до 12 ч и разным сродством к подтипам ССТр (табл. 1) [4]. Структурно-функциональные исследования показали, что аминокислотные остатки Phe7, Trp8, Lys9 и Thr10 в ССТ-14 необходимы для его биологической активности. При этом наиболее существенными являются остатки Trp8 и Lys9, поскольку именно они обеспечивают связывание с ССТр, в то время как Phe7 и Thr10 могут подвергаться незначительным заменам [4]. Основными способами модификации ССТ, приводящими к улучшению биологических характеристик их аналогов, являются введение D-аминокислот и укорачивание моле- кулы до последовательности биоактивного ядра (рис. 1).
Таблица 1
Аффинность некоторых аналогов ССТ к различным подтипам ССТр [7, 8], концентрация полумаксимального ингибирования (IC50), нмоль/л
|
Пептид |
ССТр1 |
ССТр2 |
ССТр3 |
ССТр4 |
ССТр5 |
|
IC 50 , нмоль/л |
|||||
|
ССТ-14 |
0,93±0,12 |
0,15±0,02 |
0,56±0,17 |
1,5±0,4 |
0,29±0,04 |
|
ССТ-28 |
5,2±0,3 |
2,7±0,3 |
7,7±0,9 |
5,6±0,4 |
4,0±0,3 |
|
Октреотид |
>10000 |
2,0±0,7 |
187±55 |
>1000 |
22±6 |
|
In-DTPA-октреотид |
>10000 |
22±3,6 |
182±13 |
>1000 |
237±52 |
|
Y-DOTATOC |
>10000 |
11±1,7 |
389±135 |
>10000 |
114±29 |
|
Y-DOTATATE |
>10000 |
1,6±0,4 |
>1000 |
523±239 |
187±50 |
|
Y-DOTALAN |
>10000 |
23±5 |
290±105 |
>10000 |
16±3,4 |
|
Lu-DOTATATE |
>1000 |
2,0±0,8 |
162±16 |
>1000 |
>1000 |
|
Lu-DOTA-JR11 |
>1000 |
0,73±0,15 |
>1000 |
>1000 |
>1000 |
Вапреотид
[Туг3]-октреотат
[Туг3]-октреотид
Рис. 1. Структурные формулы ССТ и некоторых аналогов ССТ.
Аналоги ССТ можно разделить на две основные категории: агонисты (вещества, способные активировать ССТр) и антагонисты (соединения, которые взаимодействуют с ССТр и блокируют или снижают их активность). При связывании агонистов с ССТр происходит интернализация комплекса агонист-рецептор за счёт рецепторно-опосредованного эндоцитоза, в результате чего радионуклид оказывается максимально близко к ядру – наиболее радиочувствительному органоиду клетки.
Первым пептидным аналогом из группы агонистов, одобренным для клинического использования, был октреотид (SMS 201-995, Сандостатин®). В отличие от ССТ, у октреотида имеется
D-Trp, D-Phe и дисульфидный мостик, расположенный ближе к активному ядру для улучшенной стабильности (рис. 1). В ядерной медицине наибольшее распространение получили производные октреотида TOC (Tyr3-октреотид) и TATE (Tyr3-октреотат) (рис. 1).
-
123I-Tyr3-октреотид стал первым РФЛП на основе аналогов ССТ и предназначался для визуализации опухолей с повышенной экспрессией ССТр. Соединение продемонстрировало биологическую активность и сродство к ССТр, аналогичное нативному ССТ [4]. Однако из-за сложной процедуры введения радиоактивной метки, высокой стоимости и ограниченной доступности 123I, а также значительного накопления активности в кишечнике и мочевом пузыре вследствие дегалогенирования in vivo , что затрудняло интерпретацию полученных изображений, 123I-Tyr3-октреотид не получил широкого распространения и вскоре был заменён на 111In-DTPA-октреотид [7].
-
111In-DTPA-октреотид (111In-DTPA-D-Phe-пентреотид, ОктреосканTM) долгое время был стандартом в визуализации ССТр [7]. После внутривенного введения 111In-DTPA-октреотид быстро выводится с мочой (около 50% от введённой активности в течение 6 ч), обеспечивая более качественные изображения по сравнению с 123I-Tyr3-октреотидом [7].
Учитывая возможность визуализации опухолей со сверхэкспрессией ССТр, следующим логическим шагом была попытка использования аналогов ССТ, меченных терапевтическими радионуклидами, в качестве нового метода лечения этой группы опухолей. Применение терапевтических радионуклидов определяется их ядерно-физическими характеристиками, такими как период полураспада (Т 1/2 ), энергия и тип излучения, доступность и химическая способность связываться с лигандами. Для ПРРТ опухолей, экспрессирующих ССТр, используются α-, β-излучающие радионуклиды, а также радионуклиды, чей распад сопровождается испусканием электронов конверсии или Оже-электронов.
В исследованиях начала 1990-х годов для ПРРТ использовался 111In-DTPA-октреотид, излучатель электронов Оже, поскольку аналоги соматостатина, меченные β-излучающими радионуклидами, не были доступны для клинического использования в то время.
Было проведено несколько клинических испытаний с использованием высоких доз 111In-DTPA-октреотида [9-11]. В одном из первых опубликованном клиническом исследовании 111In-DTPA-октреотид вводился 20 пациентам с терминальной стадией НЭО. Суммарная введённая доза достигала 74 ГБк. Из 16 пациентов, получивших суммарную дозу более 20 ГБк, у 5 пациентов была отмечена стабилизация заболевания, а ещё у 5 – уменьшение размера опухоли [9].
В исследовании [10] 50 пациентов с опухолями, положительными по ССТр, получали многократные инъекции 111In-DTPA-октреотида. У 40 пациентов суммарная введённая доза препарата составила от 20 до 160 ГБк. Терапевтический эффект был отмечен у 21 пациента. У большинства пациентов побочные эффекты были незначительными, однако у 3 пациентов, получивших дозу 111In-DTPA-октреотида более 100 ГБк, развился миелодиспластический синдром или лейкемия, а доза в 100 ГБк была признана максимально переносимой.
Delpassand et al. [11] пролечили 32 пациента высокими дозами 111In-DTPA-октреотида. 14 пациентов получили один курс терапии с 111In-DTPA-октреотидом, 18 – два курса терапии по 18,5 ГБк. У 2 пациентов, завершивших оба курса лечения, наблюдалась частичная регрессия заболевания, у остальных (16 пациентов) отмечена стабилизация заболевания по критериям RECIST. У 17 пациентов (53%) была отмечена гематологическая токсичность 1 или 2 степени, а у 1 пациента – тромбоцитопения 3 степени. Ни у одного пациента не было почечной токсичности.
Следующим этапом развития ПРРТ опухолей с экспрессией ССТр стало создание препаратов на основе аналогов TOC и TATE, конъюгированных с циклическим хелатором DOTA
(1,4,7,10-тетраазациклододекан-1,4,7,10-тетрауксусная кислота) для связывания с радиоактивными металлами, в частности β-излучателями 90Y и 177Lu.
-
90Y, чистый β-излучатель (Т 1/2 =64 ч, E β max =2,3 МэВ), и 177Lu (Т 1/2 =6,67 сут, E βmax =497 кэВ, Е γ =208 кэВ) являются в настоящее время наиболее используемыми радионуклидами для ПРРТ. Каждый из этих радионуклидов обладает рядом преимуществ. Проникающая способность β-частиц при распаде 90Y выше по сравнению с 177Lu, поэтому препараты с 90Y предпочтительнее использовать для терапии крупных опухолей. С другой стороны, 177Lu испускает β-излучение со средней энергией, более подходящей для лечения небольших опухолей. Кроме того, 177Lu является γ-излучателем, благодаря чему можно визуализировать распределение РФЛП с 177Lu в организме или проводить дозиметрические расчёты.
Первым препаратом на основе аналогов ССТ, меченных β-излучающими радионуклидами, стал 90Y-DOTATOC (90Y-DOTA,Tyr3-октреотид, OctreoTher®). Первые клинические результаты показали, что 90Y-DOTATOC обладает высокой терапевтической эффективностью. У подавляющего большинства пациентов 90Y-DOTATOC приводит к улучшению симптомов заболевания [12, 13]. Так, в исследованиях пациентам с НЭО проводили 4 цикла терапии с 90Y-DOTATOC с интервалом в 6 недель. Суммарная введённая доза составила 6,0-7,4 ГБк/м2. Ответ на терапию в соответствии с критериями ВОЗ был отмечен у 24% пациентов. При опухолях поджелудочной железы (13 пациентов) частота ответа составила 38%. Полная ремиссия наблюдалась у 5%, частичная ремиссия – у 18%, стабилизация заболевания – у 69% больных. У большей части пациентов достигнуто значительное уменьшение клинических симптомов.
Ещё одним аналогом ССТ, используемым для ПРРТ, стал ланреотид, меченный 90Y (90Y-DOTALAN). Он обладает схожим с октреотидом сродством к ССТр2, но более высоким сродством к ССТр5, поэтому он особенно подходит для опухолей, экспрессирующих этот подтип ССТр [14].
Эффективность и безопасность ПРРТ с 90Y-DOTALAN изучалась в разных центрах в рамках исследования MAURITIUS [15, 16]. Суммарные вводимые дозы 90Y-DOTALAN достигали 8,58 ГБк. После анализа результатов лечения у 154 пациентов регрессия опухоли была отмечена у 14%, а стабилизация заболевания – у 41% пациентов. О тяжёлой острой или хронической гематотоксичности или изменениях параметров функции почек или печени под воздействием 90Y-DOTALAN не сообщалось. Кроме того, у большинства пациентов накопление 90Y-DOTALAN опухолью было ниже, чем 90Y-DOTATOC.
Третьим поколением РФЛП для ПРРТ опухолей с гиперэкспрессией ССТр стали препараты, меченные 177Lu, в частности, 177Lu-DOTATOC и 177Lu-DOTATATE. Наиболее широкое применение получил препарат 177Lu-DOTATATE, в основном, из-за более высокого сродства аналога TATE (Tyr3-октреотат) к ССТр2 по сравнению с TOC и, как следствие, более длительного удержания препарата опухолью [5, 17].
В 2011 г. было начато многоцентровое рандомизированное контролируемое клиническое исследование III фазы (NETTER-1) с участием 229 пациентов с местнораспространёнными или метастатическими высокодифференцированными НЭО тонкого кишечника со сверхэкспрессией ССТр и прогрессированием заболевания на фоне приёма фиксированных доз октреотида пролонгированного действия. Сравнивалось лечение с использованием 177Lu-DOTATATE плюс октреотид пролонгированного действия (30 мг) с двойной дозой октреотида пролонгированного действия (60 мг) [18]. 177Lu-DOTATATE вводился внутривенно в дозе 7,4 ГБк каждые 8 недель;
всего в соответствии с планом лечения проводилось 4 введения этого препарата. Октреотид вводился внутримышечно каждые 4 недели [18].
В группе пациентов после терапии с 177Lu-DOTATATE медиана выживаемости без прогрессирования составила 28,4 мес. против 8,5 мес. в группе пациентов, получавших октреотид. Риск прогрессирования заболевания после терапии с 177Lu-DOTATATE снизился на 79% (p<0,0001). Отмечена более высокая частота объективного ответа на терапию у пациентов, получавших 177Lu-DOTATATE, по сравнению с пациентами контрольной группы (18% против 3%, p<0,001) [18]. Медиана общей выживаемости (ОВ) в группе пациентов, получавших 177Lu-DOTATATE, составила 48,0 мес. против 36,3 мес. в контрольной группе [19]. Проводимая терапия обеспечивала значительное улучшение качества жизни пациентов. Тяжёлых побочных эффектов отмечено не было. У некоторых пациентов наблюдалась тошнота, рвота, а также незначительное снижение числа тромбоцитов и лимфоцитов в крови [18]. Основываясь на полученных результатах, 177Lu-DOTATATE под торговой маркой Lutathera® был одобрен в Европе, а затем и в США для терапии гастроэнтеропанкреатических НЭО у взрослых пациентов [20].
Несмотря на высокую эффективность ПРРТ, отмечено, что проводимая терапия, особенно с 90Y, может оказывать токсическое действие на почки и костный мозг [8]. Почки считаются основным органом, ограничивающим вводимую активность РФЛП. Облучение почек в значительной степени связано с гломерулярной фильтрацией меченного радионуклидом аналога ССТ с последующей активной реабсорбцией в проксимальных канальцах, что приводит к задержке радиоактивности в почках. Кроме того, клетки почечных клубочков экспрессируют ССТр2, а почечные канальцы – все подтипы ССТр [21]. Эти два механизма приводят к формированию высокой поглощённой дозы в почках, что может стать причиной развития почечной недостаточности. Инфузия растворов аминокислот (содержащих L-лизин и L-аргинин отдельно или в сочетании с другими аминокислотами) во время инъекции РФЛП приводит к снижению нефротоксичности за счёт ингибирования канальцевой реабсорбции РФЛП [22].
Кроме того, более высокая почечная токсичность ПРРТ с 90Y-DOTATOC по сравнению с 177Lu-DOTATATE обусловлена различием в ядерно-физических характеристиках радионуклидов 90Y и 177Lu. В частности, считается, что более ограниченный диапазон пробега β-частиц, испускаемых 177Lu (2,2 мм), по сравнению с 90Y (12 мм) является причиной значительно меньшей нефротоксичности у пациентов, получавших 177Lu-DOTATATE. Большое ретроспективное исследование с участием 1109 пациентов, получавших 90Y-DOTATOC, продемонстрировало развитие нефротоксичности 4-5 степени у 9,2% пациентов [8], тогда как при ретроспективной оценке 610 пациентов, получавших 177Lu-DOTATATE, долгосрочная нефротоксичность не возникала [23].
Отмечено, что у пациентов, особенно с нарушением функции почек, может наблюдаться длительная циркуляция аналогов ССТ, меченных радионуклидами, что может привести к чрезмерному облучению костного мозга и гематотоксичности высокой степени [8]. Гематологическая токсичность может проявляться в двух различных формах: подострая миелотоксичность и стойкая гематологическая дисфункция, которая включает миелодиспластический синдром (МДС) и острый миелоидный лейкоз (ОМЛ) [8]. Подострая миелотоксичность проявляется в виде обратимой гематотоксичности 3-4 степени и возникает с сопоставимой частотой при терапии 90Y-DOTATOC и 177Lu-DOTATATE примерно у 5-12% пациентов [8, 18]. Надир отмечается примерно через 4-6 недель после введения РФЛП [18]. В зависимости от тяжести и типа гематотоксичности можно снижать вводимую активность и/или отложить проводимую терапию до восстановления показателей крови до приемлемых значений.
Частота встречаемости стойкой гематологической дисфункции после ПРРТ составляет от 1,8 до 4,8% со средним латентным периодом до развития заболевания 41 мес. [24-26]. У таких пациентов резко снижена ОВ, которая составляет 6-7 мес. при ОМЛ и 13,5-15 мес. при МДС [24, 25]. При этом никаких значимых факторов риска развития ОМЛ или МДС обнаружено не было [26].
В настоящее время проводятся клинические исследования, оценивающие эффективность терапии с 177Lu-DOTATATE в комбинации с другими методами лечения, а также при других видах НЭО, таких как феохромоцитома или параганглиома [4, 8]. Многообещающие результаты были получены при проведении тандемной радионуклидной терапии НЭО различных локализаций с использованием аналогов ССТр, меченных 177Lu и 90Y [27-30]. Данный подход заключается в комбинации высокоэнергетического β-излучателя 90Y с большой величиной пробега для нацеливания на крупные опухолевые поражения и β-/γ-излучателя средней энергии 177Lu для нацеливания на более мелкие очаги. Как правило, под тандемной ПРРТ подразумевается совместное введение смеси 90Y/177Lu-DOTATATE/TOC или инфузия обоих РФЛП в один и тот же день. Возможно также чередование циклов 90Y-DOTATATE/ТОС и 177Lu-DOTATATE/ТОС (в англоязычной литературе такой подход именуется «dual therapy», «дуэтная терапия») [8].
Впервые такой подход к терапии был описан de Jong et al. [27]. В исследованиях на крысах было показано, что введение смеси, состоящей из 50% 177Lu-DOTATATE и 50% 90Y-DOTATOC, приводило к 3-кратному увеличению продолжительности жизни животных. Таким образом, авторы пришли к выводу, что совместное использование обоих радионуклидов позволяет достичь более значимого противоопухолевого эффекта при наличии опухолей различного размера.
Kunikowska et al. [29] провели первое нерандомизированное клиническое исследование, в котором сравнивалась монотерапия 90Y-DOTATATE (n=25) с тандемной терапией 90Y/177Lu-DOTATATE (n=25) у пациентов с прогрессирующими НЭО. Значительное увеличение ОВ было достигнуто в группе пациентов, получавших тандемную терапию, по сравнению с группой, получавшей 90Y-DOTATATE. Однако различий по частоте объективного ответа между группами отмечено не было.
Значительное увеличение выживаемости было также продемонстрировано в работах [28, 31]. Так, исследование с участием 486 пациентов показало, что в группе, получавшей чередующиеся циклы 90Y-DOTATOC и 177Lu-DOTATOC (n=249), выживаемость была значительно выше, чем в группе, получавшей монотерапию 90Y-DOTATOC (n=237) (5,51 против 3,96 лет соответственно, p=0,006) [28]. В исследовании, проводимом Baum et al. [31], было включено 1048 пациентов, которым проводили ПРРТ с 177Lu (36%), 90Y (15%) или комбинацией 90Y и 177Lu (49%). Была отмечена значительная разница в ОВ в пользу тандемной ПРРТ (64 мес.), тогда как ОВ в группах, получавших монотерапию с 177Lu или 90Y, составила 44 и 24 мес. соответственно.
Таким образом, в ряде клинических исследований показана высокая эффективность тандемной ПРРТ с приемлемым уровнем токсичности. При этом есть данные, свидетельствующие о преимуществе в выживаемости после тандемной терапии. Необходимы рандомизированные контролируемые клинические испытания, которые бы подтвердили преимущества такого подхода к лечению НЭО.
Возможности терапии пациентов с прогрессирующими гастроэнтеропанкреатическими нейроэндокринными новообразованиями и метастатическим поражением печени часто ограничены. Поэтому логично предположить, что локальная доставка меченных аналогов ССТ позволит увеличить эффективность противоопухолевой терапии и снизить системную токсичность. Для этого предлагается внутриартериальное введение (в печеночную артерию) РФЛП. Клинические исследования показали, что внутриартериальная ПРРТ с различными РФЛП (90Y/177Lu-DOTATOC/TATE, 90Y-DOTALAN) является безопасным и эффективным методом терапии НЭО, осложнённых печеночными метастазами [32-34]. Например, при внутриартериальном введении 177Lu-DOTATATE пациентам с неоперабельными метастатическими гастроэнтеропанкреатиче-скими НЭО накопление препарата в опухоли было в 3 раза выше, чем при внутривенном введении [34]. В ряде исследований [32, 33] было отмечено увеличение ОВ после внутриартериальной ПРРТ.
Одним из ограничений использования 177Lu-DOTATATE является необходимость введения нескольких доз РФЛП для достижения противоопухолевого эффекта. Поэтому в целях улучшения фармакокинетики данного препарата была предпринята попытка конъюгации аналога красителя синий Эванса (Evans Blue, EB) с TATE (EB-TATE). Фрагмент EB обеспечивал обратимое связывание EB-TATE с сывороточным альбумином, тем самым увеличивая его биологический период полувыведения из крови [35]. В клиническом исследовании I фазы была изучена безопасность, фармакокинетика и дозиметрия препарата 177Lu-DOTA-EB-TATE при однократном введении [36]. Из 8 пролеченных пациентов с прогрессирующими метастатическими НЭО 5 получили 177Lu-DOTA-EB-TATE (0,35-0,70 ГБк); остальные 3 – 177Lu-DOTATATE (0,28-0,41 ГБк). У пациентов, получивших 177Lu-DOTA-EB-TATE, отмечалось пролонгированное нахождение препарата в крови, а накопление препарата в опухоли в 7,9 раза выше, чем 177Lu-DOTATATE. При этом поглощённая доза в костном мозге и почках, формируемая при введении 177Lu-DOTA-EB-TATE, была значительно выше (в 18,2 и 3,2 раза соответственно) по сравнению с 177Lu-DOTATATE [36].
В опубликованной недавно работе [37] была оценена безопасность и эффективность введения до трёх циклов 177Lu-DOTA-EB-TATE у 32 пациентов с НЭО (3 группы: А – 1,17±0,09 ГБк/цикл, Б – 1,89±0,53 ГБк/цикл, С – 3,97±0,84 ГБк/цикл). Тяжёлых побочных эффектов отмечено не было. Гематотоксичность 3 степени зарегистрирована у 1 больного (16,6%) в группе Б и у 3 больных (21,4%) в группе С. Нефротоксичность отсутствовала. Эффективность терапии, оцениваемая по снижению SUV, была выше в группах пациентов, получавших 177Lu-DOTA-EB-TATE в дозах 1,89 и 3,97 ГБк/цикл.
Среди других β-излучающих радионуклидов, которые потенциально могут быть использованы для ПРРТ опухолей с гиперэкспрессией ССТр, следует отметить 188Re. Он обладает оптимальными ядерно-физическими свойствами (Т 1/2 =16,9 ч, E βmax =2,12 МэВ, E γ =155 кэВ (15%)), а также следует отметить возможность получения этого радионуклида с высокой удельной активностью с помощью генератора 188W/188Re.
В литературе имеются данные об аналогах ССТ, меченных 188Re. Например, были синтезированы 188Re-вапреотид (188Re-RC-160) и 188Re-ланреотид. Их биораспределение было изучено на различных экспериментальных моделях опухолей (в частности, рака предстательной железы, поджелудочной железы, толстого кишечника, лёгких, шейки матки и др.) [38, 39]. 188Re-RC-160 оказывал дозозависимый противоопухолевый эффект (торможение или регресс опухоли) и увеличивал выживаемость животных [38]. 188Re-ланреотид накапливался в опухолевых клетках рака шейки матки HeLa до 3,1%/г в срок 1,5 ч после инъекции и удерживался на высоком уровне в течение 24 ч [39]. Расчётная поглощённая доза в опухоли составила 0,46 Гр/МБк, а экстраполированная эффективная доза для человека – 0,018 мЗв/МБк. При этом предполагаемая поглощённая доза в красном костном мозге не превышала 7,49·10-3 мЗв/МБк.
Новый аналог ССТ, меченный 188Re, 188Re-P2045, продемонстрировал многообещающие результаты in vivo при лечении перевитого рака поджелудочной железы у мышей, существенно ингибируя рост опухоли и не оказывая токсического действия [40]. Была проведена I фаза клинических исследований 188Re-P2045 с участием 15 пациентов с распространённым раком лёгких
-
[41] . У 5 из 8 пациентов заболевание стабилизировалось не менее чем на 8 недель, а медиана ОВ составила 11,5 мес.
Потенциальный интерес для ПРРТ могут представлять и такие β-излучающие радионуклиды как 47Sc (T 1/2 =3,35 дней, E βmax =600,8 кэВ), 67Cu (T 1/2 =2,58 дней, E βmax =577 кэВ) и 161Tb (T 1/2 =6,91 дней, E βmax =593 кэВ) [4]. На сегодняшний день наиболее изученным изотопом для ПРРТ является 161Tb, свойства распада которого аналогичны свойствам 177Lu. Важно отметить, что 161Tb испускает также значительное количество конверсионных и Оже-электронов, что, как считается, делает 161Tb терапевтически более эффективным, чем 177Lu.
Первое введение 161Tb-DOTATOC людям было осуществлено в Германии [42]. Двум пациентам с метастатическими НЭО вводили 161Tb-DOTATOC (596 и 1300 МБк) для анализа распределения радиопептида на планарных изображениях всего тела и ОФЭКТ/КТ. Профиль распределения 161Tb-DOTATOC был аналогичен таковому для 177Lu-DOTATOC с преимущественным накоплением в печени, селезёнке, кишечнике, почках и мочевом пузыре. Была продемонстрирована возможность визуализации даже небольших метастазов после введения относительно низкой активности 161Tb-DOTATOC с помощью γ-сцинтиграфии и ОФЭКТ/КТ. О побочных эффектах после инъекции 161Tb-DOTATOC не сообщалось.
Менее изученным радионуклидом для ПРРТ, нацеленной на ССТр, является 47Sc. На сегодняшний день опубликована единственная работа, в которой сообщается о рецептор-специфич-ном поглощении 47Sc-DOTATOC клетками опухоли поджелудочной железы AR42J in vitro [43]. Тем не менее, тераностическая пара (43Sc)44Sc/47Sc обладает большим потенциалом для использования в диагностике и терапии рака [4].
ПРРТ опухолей со сверхэкспрессией ССТр не ограничивается использованием β-излучаю-щих радионуклидов. Огромный потенциал имеют изотопы, распадающиеся с излучением α-частиц. Альфа-излучение характеризуется высокой линейной передачей энергии, вызывающей двухцепочечные разрывы ДНК, что приводит к быстрой гибели клеток даже от однократного воздействия нескольких α-частиц [44]. Невысокая величина пробега α-частиц (50-100 мкм, т.е. около двух или трёх диаметров клеток) ограничивает повреждение здоровых тканей, но в то же время требует локализации в ядре клетки для оказания терапевтического эффекта.
-
225Ac/213Bi-DOTATOC, 225Ac/213Bi-DOTATATE, а также 212Pb-DOTAMTATE уже продемонстрировали многообещающие противоопухолевые эффекты в исследованиях на животных [45-47]. При введении 213Bi-DOTATOC крысам с экспериментальной моделью карциномы поджелудочной железы CA20948 накопление препарата в почках и костном мозге было соответственно в 3 и 5 раз ниже, чем свободного 213Bi. 213Bi-DOTATOC продемонстрировал дозозависимый противоопухолевый эффект с минимальной органной токсичностью. 225Ac-DOTATOC также демонстрировал более выраженный противоопухолевый эффект по сравнению с 177Lu-DOTATOC [46].
Высокая эффективность и безопасность ПРРТ с 225Ac-DOTATOC была показана в ряде клинических исследований [48, 49]. Лечение было признано безопасным при вводимой активности 18,5 МБк, введённой с интервалом в 2 мес. Максимально переносимая доза составила 40 МБк. Несмотря на эффективность проводимой терапии, отмеченной у многих пациентов, авторы подчёркивают необходимость проведения дальнейших исследований с более крупными группами пациентов [48]. Отмечается, что ПРРТ с 225Ac-DOTATOC может быть эффективна в случае радиорезистентности опухолей к β-излучателям [49]. В недавнем ретроспективном исследовании [50] было показано, что вводимая активность около 20 МБк (с интервалом в 4 мес.) и кумулятивные дозы до 60-80 МБк позволяли избежать как острой, так и хронической гематотоксичности 3/4 степени у пациентов с поздними стадиями злокачественных новообразований.
Недавно были опубликованы результаты проспективного исследования эффективности 225Ac-DOTATATE для терапии пациентов с метастатическими гастроэнтеропанкреатическими НЭО, у которых была отмечена стабилизация или прогрессирование заболевания после терапии с 177Lu-DOTATATE [51]. Всем пациентам были проведены 2 курса системной терапии с 225Ac-DOTATATE (100 кБк/кг массы тела) с интервалом 8 недель. Суммарная введённая активность составила 7770-44400 кБк. Плановый промежуточный анализ морфологического ответа, проведённый через 8 недель после второго курса терапии, выявил частичный ответ у 62,5%, а стабилизацию заболевания – у 37,5% из 24 обследованных пациентов. Ни у одного из пациентов не было выявлено гематологической, почечной или гепатоксичности 3/4 степени.
Были опубликованы первые результаты клинического исследования 212Pb-DOTAMTATE (AlphaMedixTM) с участием 20 пациентов с гистологически подтверждёнными НЭО и не прошедших ПРРТ с 90Y, 177Lu или 111In [52]. К настоящему времени 10 пациентов, получивших 212Pb-DOTAMTATE в максимальной дозе 2,50 кБк/кг/цикл, завершили все 4 цикла лечения. Объективный ответ по критерию RECIST 1.1 был достигнут у 80% пациентов. Серьёзных побочных эффектов зарегистрировано не было.
Таким образом, первые результаты клинических исследований показали, что ПРРТ с α-излучателями может быть эффективна в случае радиорезистентности к β-излучателям, имеет низкий профиль токсичности и улучшает качество жизни пациентов.
В течение долгого времени считалось, что процесс интернализации и последующего накопления меченных лигандов внутри клетки является крайне важным для осуществления цитотоксического действия радионуклидов. Однако в 2006 г. была опубликована работа [53], в которой сообщалось, что антагонисты ССТр связываются с большим числом рецепторных сайтов, чем агонисты. В отличие от агонистов антагонисты не интернализуются в клетку, однако было показано, что нацеливание на ССТр может быть эффективным и без последующей интернализации комплекса лиганд-рецептор. Например, антагонисты характеризуются повышенным накоплением в опухоли, низким содержанием в почках и быстрым клиренсом организма по сравнению с агонистами [53]. В химической структуре основным отличием антагонистов стало изменение первой аминокислоты в структуре агонистов с D на L-изомер и Cys2 – с L на D-изомер [4].
Логично предположить, что более высокое поглощение опухолью антагонистов ССТр может увеличить эффективность противоопухолевой терапии. Первым антагонистом, меченным терапевтическим радионуклидом, стал 177Lu-DOTA-sst2-ANT (177Lu-DOTA-BASS). В экспериментах in vitro на образцах ССТр2-позитивных опухолей человека было показано, что препарат связывался со значительно большим числом рецепторных сайтов по сравнению с 177Lu-DOTATATE [54]. Накопление 177Lu-DOTA-sst2-ANT in vivo в ксенотрансплантатах опухолей, сверхэкспресси-рующих ССТр2, было крайне высоким (до 30%/г через 4 ч после инъекции) [55]. Полученные результаты были весьма обнадёживающими и доказали перспективность дальнейших разработок РФЛП на основе антагонистов ССТр.
Позднее были синтезированы антагонисты второго поколения с более высокой аффинностью к ССТр. В частности, наиболее высокое сродство к ССТр2 продемонстрировало соединение DOTA-JR11, из-за чего именно оно было выбрано для ПРРТ. Доклинические исследования, сравнивающие 177Lu-DOTA-JR11 (177Lu-OPS201, 177Lu-IPN01072, 177Lu-satoreotide) с 177Lu-DOTATATE, показали значительно более высокое поглощение опухолью (до 23,9±4,5%/г через 4 ч после инъекции) и более длительное удержание 177Lu-DOTA-JR11 по сравнению с 177Lu-DOTATATE (до 17,8±4,4%/г в срок 4 ч). Введение 177Lu-DOTA-JR11 приводило к формированию поглощённой дозы в опухоли в 2,5 раза выше, чем после инъекции 177Lu-DOTATATE (0,854 мГр/МБк против 0,333 мГр/МБк для опухоли размером 4 см) [56].
В работе [57] также было продемонстрировано более высокое поглощение опухолью 177Lu-DOTA-JR11 (в 5 раз) по сравнению с 177Lu-DOTATATE. Поглощённая доза в опухоли при введении 177Lu-DOTA-JR11 составила 1,8±0,7 Гр/МБк, что в 4,4 раза выше по сравнению с 177Lu-DOTATATE. В группе животных, получавших 177Lu-DOTA-JR11, была отмечена более высокая средняя выживаемость (71 день против 61 дня) и более длительная задержка роста опухоли (26±7 дней по сравнению с 18±5 дней). Несмотря на то, что лишь 10% 177Lu-DOTA-JR11 оказывалось внутри опухолевых клеток, препарат вызывал как минимум на 60% больше двухцепочечных разрывов ДНК по сравнению с 177Lu-DOTATATE, уровень интернализации которого составил 74±3%.
В пилотном клиническом исследовании у 4 пациентов с прогрессирующими НЭО накопление 177Lu-DOTA-JR11 в опухоли было в 1,7-10,6 раза выше, чем 177Lu-DOTATATE [58]. Терапия с 177Lu-DOTA-JR11 привела к частичной ремиссии заболевания у 2 пациентов, смешанному ответу у 1 пациента и стабилизации заболевания у 1 пациента.
В исследовании I фазы оценивали эффективность и безопасность 177Lu-DOTA-JR11 у 20 пациентов с прогрессирующими ССТр-позитивными НЭО [59]. 6 пациентов получили один курс ПРРТ с 177Lu-DOTA-JR11, а 14 – два. Максимальная вводимая активность за курс составила 7,4 ГБк. Гематотоксичность после 1 курса была незначительной. Однако после 2 курса у 4 пациентов наблюдалась гематотоксичность 4 степени, вследствие чего предписанная активность для 2 курса была снижена на 50%. Объективный ответ на лечение получен у 45% пациентов. Медиана выживаемости без прогрессирования составила 21 мес. В настоящее время продолжаются исследования для определения оптимальной терапевтической дозы и режима введения 177Lu-DOTA-JR11.
Перспективным соединением для ПРРТ опухолей, экспрессирующих ССТр, может стать LM3 – ещё один антагонист ССТр. Первые результаты клинических исследований продемонстрировали более высокое накопление 177Lu-DOTA-LM3 в опухолевых очагах по сравнению с 177Lu-DOTATOC (0,12%/мл против 0,05%/мл), а также более длительный период полувыведения из организма (76 ч против 50 ч) [60]. Из-за этого средние поглощённые дозы в органах и опухолях, формируемые при введении 177Lu-DOTA-LM3 (21 Гр/ГБк в опухоли, 2,0 Гр/ГБк – в почках), были выше, чем для 177Lu-DOTATOC (6 и 0,6 Гр/ГБк в опухоли и почках соответственно).
В другом исследовании оценивали безопасность и эффективность 177Lu-DOTA-LM3 [61]. 51 пациенту с метастатическими НЭО была проведена ПРРТ. Было обнаружено более высокое поглощение и более длительный период полувыведения 177Lu-DOTA-LM3 всеми органами по сравнению с агонистом 177Lu-DOTATOC. Согласно критериям RECIST 1.1, после терапии 177Lu-DOTA-LM3 частичный ответ отмечался у 17 (36,2%) пациентов, а стабилизация заболевания – у 23 (48,9%) пациентов. Все пациенты переносили терапию без каких-либо серьёзных острых побочных эффектов. Наиболее тяжёлым отсроченным нежелательным явлением была тромбоцитопения 3 степени у 3 (5,9%) пациентов. Не наблюдалось значительного снижения почечной функции и гепатотоксичности.
Были проведены исследования противоопухолевой эффективности 161Tb-DOTA-LM3 и 177Lu-DOTA-LM3 по сравнению с 161Tb-DOTATOC и 177Lu-DOTATOC [62]. В экспериментах in vitro поглощение антагонистов 161Tb-DOTA-LM3 и 177Lu-DOTA-LM3 опухолевыми клетками AR42J было в 4-6 раз выше, чем агонистов 161Tb-DOTATOC и 177Lu-DOTATOC. Во всех случаях аналоги ССТ c 161Tb оказывались более эффективными в отношении опухолевых клеток по сравнению с препаратами на основе 177Lu. Активность 161Tb-DOTA-LM3 была в 102 раза выше, чем для 177Lu-DOTA-LM3. Поглощение и удержание опухолью 161Tb-DOTA-LM3 in vivo достигало 35±7%/г в срок 24 ч и 21±4%/г в срок 48 ч, что было значительно выше по сравнению с 161Tb-DOTATOC (6,3±0,6%/г в срок 24 ч и 3,7±0,7%/г в срок 48 ч). Основываясь на полученных данных, авторы предполагают, что 161Tb-DOTA-LM3 может оказаться клинически более эффективным, чем имеющиеся в настоящее время РФЛП, для лечения пациентов с НЭО [62].
Таким образом, несмотря на очень низкую степень интернализации, антагонисты ССТр демонстрируют благоприятную фармакокинетику и повышенное накопление в опухоли по сравнению с агонистами, что может повысить эффективность ПРРТ с антагонистами ССТр. Более высокое поглощение антагонистов ССТр опухолями может быть весьма полезным для ПРРТ опухолей с пониженной экспрессией ССТр, например, рака молочной железы, мелкоклеточного рака лёгкого, почечно-клеточного рака, неходжкинских лимфом, феохромоцитомы и медуллярного рака щитовидной железы [63]. Поэтому необходимы дальнейшие исследования эффективности противоопухолевой терапии с антагонистами ССТр.
Заключение
Пептид-рецепторная радионуклидная терапия (ПРРТ) с использованием аналогов ССТ, меченных радионуклидами, стала общепринятым методом лечения пациентов с неоперабельными метастатическими НЭО, сверхэкспрессирующими ССТр. На сегодняшний день наиболее часто используемыми являются агонисты ССТр, меченные β-излучающими радионуклидами 90Y и 177Lu. 177Lu-DOTATATE (Lutathera®) стал первым зарегистрированным в Европе и США РФЛП для терапии гастроэнтеропанкреатических НЭО у взрослых пациентов, который значительно увеличивает выживаемость и улучшает качество жизни без серьёзных побочных эффектов.
Текущие исследования сосредоточены на повышении эффективности ПРРТ и снижении их токсичности в отношении здоровых органов и тканей. Для этого можно использовать комбинацию радиопептидов, меченных 90Y и 177Lu, применяемых последовательно или тандемно. Внутриартериальное введение радиопептидов, нацеленных на ССТр, представляет эффективную и безопасную альтернативу внутривенной ПРРТ при метастазах в печень. На основании опубликованных работ можно констатировать, что предпринимаются значительные усилия для получения новых аналогов ССТ с улучшенными фармакокинетическими свойствами и специфичностью к ССТр для снижения их интенсивного поглощения нецелевыми органами (печенью, почками и др.). Потенциальный интерес для ПРРТ с аналогами ССТ представляют радионуклиды 161Tb, 47Sc, а также α-излучающие радионуклиды 225Ac, 213Bi, 212Pb с высокой линейной передачей энергии. Аналоги ССТ, меченные α-излучателями, особенно эффективны у пациентов с резистентными к β-излучению метастатическими НЭО.
Важным достижением в ПРРТ, нацеленной на ССТр, стало использование антагонистов ССТр, которые распознают больше сайтов связывания на рецепторах и демонстрируют улучшенную фармакокинетику по сравнению с агонистами. Использование радиопептидов с антагонистическими свойствами может представлять интерес для терапии опухолей с более низкой экспрессией ССТр по сравнению с НЭО, однако это требует дальнейших исследований.
Список литературы Аналоги соматостатина, меченные радионуклидами, для терапии онкологических заболеваний. Обзор
- Global Cancer Observatory. [Электронный ресурс]. URL: https://gco.iarc.fr/ (дата обращения 08.02.2022).
- Reubi J.C., Laissue J., Krenning E., Lamberts S.W. Somatostatin receptors in human cancer: incidence, characteristics, functional correlates and clinical implications //J. Steroid Biochem. Mol. Biol. 1992. V. 43, N 1-3. P. 27-35.
- Okarvi S.M. Peptide-based radiopharmaceuticals and cytotoxic conjugates: potential tools against cancer //Cancer Treat. Rev. 2008. V. 34, N 1. P. 13-26.
- Eychenne R., Bouvry C., Bourgeois M., Loyer P., Benoist E., Lepareur N. Overview of radiolabeled somatostatin analogs for cancer imaging and therapy //Molecules. 2020. V. 25, N 17. P. 4012.
- Reubi J.C., Waser B., Schaer J.C., Laissue J.A. Somatostatin receptor sst1-sst5 expression in normal and neoplastic human tissues using receptor autoradiography with subtype-selective ligands //Eur. J. Nucl. Med. 2001. V. 28, N 7. P. 836-846.
- Volante M., Rosas R., Allia E., Granata R., Baragli A., Muccioli G., Papotti M. Somatostatin, cortistatin and their receptors in tumours //Mol. Cell Endocrinol. 2008. V. 286, N 1-2. P. 219-229.
- Pauwels E., Cleeren F., Bormans G., Deroose C.M. Somatostatin receptor PET ligands - the next generation for clinical practice //Am. J. Nucl. Med. Mol. Imaging. 2018. V. 8, N 5. P. 311-331.
- Bidakhvidi A.N., Goffin K., Dekervel J., Baete K., Nackaerts K., Clement P., van Cutsem E., Verslype C., Deroose C.M. Peptide receptor radionuclide therapy targeting the somatostatin receptor: basic principles, clinical applications and optimization strategies //Cancers (Basel). 2022. V. 14, N 1. P. 129.
- Krenning E.P., Valkema R., Kooji P.P., Breeman W.A., Bakker W.H., de Herder W.W., van Eijck C.H., Kwekkeboom D.J., de Jong M., Pauwels S. Scintigraphy and radionuclide therapy with [indium-111-labelled-diethyl triamine penta-acetic acid-D-Phe1]-octreotide //Ital. J. Gastroenterol. Hepatol. 1999. V. 3, N S2. P. S219-S223.
- Valkema R., de Jong M., Bakker W.H., Breeman W.A., Kooij P.P., Lugtenburg P.J., de Jong F.H., Christiansen A., Kam B.L., de Herder W.W., Stridsberg M., Lindemans J., Ensing G., Krenning E.P. Phase I study of peptide receptor radionuclide therapy with [In-DTPA]octreotide: the Rotterdam experience //Semin. Nucl. Med. 2002. V. 32, N 2. P. 110-122.
- Delpassand E.S., Sims-Mourtada J., Saso H., Azhdarinia A., Ashoori F., Torabi F., Espenan G, Moore W.H., Woltering E., Anthony L. Safety and efficacy of radionuclide therapy with high-activity In-111 pentetreotide in patients with progressive neuroendocrine tumors //Cancer Biother. Radiopharm. 2008. V. 23, N 3. P. 292-300.
- Waldherr C., Pless M., Maecke H.R., Haldemann A., Mueller-Brand J. The clinical value of [90Y-D0TA]-D-Phe1-Tyr3-octreotide (90Y-D0TAT0C) in the treatment of neuroendocrine tumours: a clinical phase II study //Ann. Oncol. 2001. V. 12, N 7. P. 941-945.
- Waldherr C., Pless M., Maecke H.R., Schumacher T., Crazzolara A., Nitzsche E.U., Haldemann A., Mueller-Brand J. Tumor response and clinical benefit in neuroendocrine tumors after 7.4 GBq (90)Y-D0TAT0C //J. Nucl. Med. 2002. V. 43, N 5. P. 610-616.
- Krenning E.P., Kwekkeboom D.J., Valkema R., Pauwels S., Kvols L.K., de Jong M. Peptide receptor radionuclide therapy //Ann. N.Y. Acad. Sci. 2004. V. 1014, N 1. P. 234-245.
- Virgolini I., Traub T., Novotny C., Leimer M., Füger B., Li SR., Patri P., Pangerl T., Angelberger P., Raderer M., Burggasser G., Andreae F., Kurtaran A., Dudczak R. Experience with indium-111 and yttrium-90-labeled somatostatin analogs //Curr. Pharm. Des. 2002. V. 8, N 20. P. 1781-1807.
- Virgolini I., Britton K., Buscombe J., Moncayo R., Paganelli G., Riva P. In- and Y-D0TA-lanreotide: results and implications of the MAURITIUS trial //Semin. Nucl. Med. 2002. V. 32, N 2. P. 148-155.
- Esser J.P., Krenning E.P., Teunissen J.J., Kooij P.P., van Gameren A.L., Bakker W.H., Kwekkeboom D.J. Comparison of [177Lu-D0TA0,Tyr3]octreotate and [177Lu-D0TA0,Tyr3]octreotide: which peptide is preferable for PRRT? //Eur. J. Nucl. Med. Mol. Imaging. 2006. V. 33, N 11. P. 1346-1351.
- Strosberg J., El-Haddad G., Wolin E., Hendifar A., Yao J., Chasen B., Mittra E., Kunz P.L., Kulke M.H., Jacene H., Bushnell D., O'Dorisio T.M., Baum R.P., Kulkarni H.R., Caplin M., Lebtahi R., Hobday Y., Delpassand E., van Cutsem E., Benson A., Srirajaskanthan R., Pavel M., Mora J., Berlin J., Grande E., Reed N., Seregni E., Öberg K., Sierra M.L., Santoro P., Thevenet T., Erion J.L., Ruszniewski P., Kwekkeboom D., Krenning E., NETTER-1 Trial investigators. Phase 3 trial of 177Lu-Dotatate for midgut neuroendocrine tumors //N. Engl. J. Med. 2017. V. 376, N 2. P. 125-135.
- Strosberg J., Caplin M.E., Kunz P.L., Ruszniewski P.B., Bodei L., Hendifar A., Mittra E., Wolin E.M., Yao J.C., Pavel M.E., Grande E., van Cutsem E., Seregni E., Duarte H., Gericke G., Bartalotta A., Mariani M.F., Demange A., Mutevelic S., Krenning E.P., NETTER-1 investigators. 177Lu-Dotatate plus long-acting octreotide versus high-dose long-acting octreotide in patients with midgut neuroendocrine tumours (NETTER-1): final overall survival and long-term safety results from an open-label, randomised, controlled, phase 3 trial //Lancet Oncol. 2021. V. 22, N 12. P. 1752-1763.
- Hennrich U., Kopka K. Lutathera®: the first FDA- and EMA-approved radiopharmaceutical for peptide receptor radionuclide therapy //Pharmaceuticals (Basel). 2019. V. 12, N 3. P. 114.
- Vegt E., de Jong M., Wetzels J.F.M., Masereeuw R., Melis M., Oyen W.J.G., Gotthardt M., Boerman O.C. Renal toxicity of radiolabeled peptides and antibody fragments: mechanisms, impact on radionuclide therapy, and strategies for prevention //J. Nucl. Med. 2010. V. 51, N 7. P. 1049-1058.
- Bodei L., Cremonesi M., Zoboli S., Grana C., Bartolomei M., Rocca P., Caracciolo M, Mäcke H.R., Chinol M., Paganelli G. Receptor-mediated radionuclide therapy with 90Y-DOTATOC in association with amino acid infusion: a phase I study //Eur. J. Nucl. Med. Mol. Imaging. 2003. V. 30, N 2. P. 207-216.
- Brabander T., van der Zwan W.A., Teunissen J.J.M., Kam B.L.R., Feelders R.A., de Herder W.W., van Eijck C.H.J., Franssen G.J.H., Krenning E.P., Kwekkeboom D.J. Long-term efficacy, survival, and safety of [177Lu-D0TA(0),Tyr(3)]octreotate in patients with gastroenteropancreatic and bronchial neuroendocrine tumors //Clin. Cancer Res. 2017. V. 23, N 16. P. 4617-4624.
- Goncalves I., Burbury K., Michael M., Iravani A., Ravi Kumar A.S., Akhurst T., Tiong I.S., Blombery P., Hofman M.S., Westerman D., Hicks R.J., Kong G. Characteristics and outcomes of therapy-related myeloid neoplasms after peptide receptor radionuclide/chemoradionuclide therapy (PRRT/PRCRT) for metastatic neuroendocrine neoplasia: a single-institution series //Eur. J. Nucl. Med. Mol. Imaging. 2019. V. 46, N 9. P. 1902-1910.
- Chantadisai M., Kulkarni H.R., Baum R.P. Therapy-related myeloid neoplasm after peptide receptor radionuclide therapy (PRRT) in 1631 patients from our 20 years of experiences: prognostic parameters and overall survival //Eur. J. Nucl. Med. Mol. Imaging. 2021. V. 48, N 5. P. 1390-1398.
- Bergsma H., van Lom K., Raaijmakers M.H.G.P., Konijnenberg M., Kam L.R., Teunissen J.J.M., de Herder W.W., Krenning E.P., Kwekkeboom D.J. Persistent hematologic dysfunction after peptide receptor radionuclide therapy with 177Lu-D0TATATE: incidence, course, and predicting factors in patients with gastroenteropancreatic neuroendocrine tumors //J. Nucl. Med. 2018. V. 59, N 3. P. 452-458.
- de Jong M., Breeman W.A.P., Valkema R., Bernard B.F., Krenning E.P. Combination radionuclide therapy using 177Lu- and 90Y-labeled somatostatin analogs //J. Nucl. Med. 2005. V. 46, N S1. P. 13S-17S.
- Villard L., Romer A., Marincek N., Brunner P., Koller M.T., Schindler C., Ng Q.K.T., Mäcke H.R., MüllerBrand J., Rochlitz C., Briel M., Walter M.A. Cohort study of somatostatin-based radiopeptide therapy with (90)Y-D0TA-T0C versus (90)Y-D0TA-T0C plus (177)Lu-D0TA-T0C in neuroendocrine cancers //J. Clin. Oncol. 2012. V. 30, N 10. P. 1100-1106.
- Kunikowska J., Krolicki L., Hubalewska-Dydejczyk A., Mikotajczak R., Sowa-Staszczak A., Pawlak D. Clinical results of radionuclide therapy of neuroendocrine tumours with 90Y-D0TATATE and tandem 90Y/177Lu-DOTATATE: which is a better therapy option? //Eur. J. Nucl. Med. Mol. Imaging. 2011. V. 38, N 10. P. 1788-1797.
- Parghane R.V., Mitra A., Bannore T.U., Rakshit S., Banerjee S., Basu S. Initial clinical evaluation of indigenous 90Y-DOTATATE in sequential duo-PRRT approach (177Lu-DOTATATE and 90Y-DOTATATE) in neuroendocrine tumors with large bulky disease: observation on tolerability, 90Y-D0TATATE post-PRRT imaging characteristics (bremsstrahlung and PETCT) and early adverse effects //World J. Nucl. Med. 2021. V. 20, N 1. P. 73-81.
- Baum R.P., Kulkarni H.R., Singh A., Kaemmerer D., Mueller D., Prasad V., Hommann M., Robiller F.C., Niepsch K., Franz H., Jochems A., Lambin P., Hörsch D. Results and adverse events of personalized peptide receptor radionuclide therapy with 90Yttrium and 177Lutetium in 1048 patients with neuroendocrine neoplasms //0ncotarget. 2018. V. 9, N 24. P. 16932-16950.
- Singh A., Zhang J., Kulkarni H., Baum R. Intra-arterial PRRT of sstr-expressing tumors in patients with hepatic only versus extrahepatic tumor: efficacy and safety evaluation //J. Nucl. Med. 2019. V. 60, N S1. P. 625.
- Kolasinska-Cwikta A., Nowicki M.L., Sankowski A.J., Patucki J.M., Buscombe J.R., Glinka L., Cwikta J.B. Radiological and clinical efficacy of intra-arterial 90Y-D0TATATE in patients with unresectable, progressive, liver dominant neuroendocrine neoplasms //J. Clin. Med. 2021. V. 10, N 8. P. 1794.
- Thakral P., Sen I., Das S.S., Manda D., Cb V., Malik D. Dosimetric analyses of intra-arterial versus standard intravenous administration of 177Lu-D0TATATE in patients of well differentiated neuroendocrine tumor with liver-dominant metastatic disease //Br. J. Radiol. 2021. V. 94, N 1126. P. 20210403.
- Tian R., Jacobson O., Niu G., Kiesewetter D.O., Wang Z., Zhu G., Ma Y., Liu G., Chen X. Evans blue attachment enhances somatostatin receptor subtype-2 imaging and radiotherapy //Theranostics. 2018. V. 8, N 3. P. 735-745.
- Zhang J., Wang H., Jacobson O., Cheng Y., Niu G., Li F., Bai C., Zhu Z., Chen X. Safety, pharmacokinetics, and dosimetry of a long-acting radiolabeled somatostatin analog 177Lu-D0TA-EB-TATE in patients with advanced metastatic neuroendocrine tumors //J. Nucl. Med. 2018. V. 59, N 11. P. 1699-1705.
- Liu Q., Zang J., Sui H., Ren J., Guo H., Wang H., Wang R., Jacobson O., Zhang J., Cheng Y., Zhu Z., Chen X. Peptide receptor radionuclide therapy of late-stage neuroendocrine tumor patients with multiple cycles of 177Lu-D0TA-EB-TATE //J. Nucl. Med. 2021. V. 62, N 3. P. 386-392.
- Zamora P.O., Gulhke S., Bender H., Diekmann D., Rhodes B.A., Birsack H.J., Knapp Jr.F.F. Experimental radiotherapy of receptor-positive human prostate adenocarcinoma with 188Re-RC-160, a directly-radiolabeled somatostatin analogue //Int. J. Cancer. 1996. V. 65, N 2. P. 214-220.
- Molina-Trinidad E.M., Arteaga de Murphy C., Ferro-Flores G., Murphy-Stack E., Jung-Cook H. Radio-pharmacokinetic and dosimetric parameters of 188Re-lanreotide in athymic mice with induced human cancer tumors //Int. J. Pharm. 2006. V. 310, N 1-2. P. 125-130.
- Nelson C.A., Aure M.T., Adams C.T., Zinn K.R. The somatostatin analog 188Re-P2045 inhibits the growth of AR42J pancreatic tumor xenografts //J. Nucl. Med. 2014. V. 55, N 12. P. 2020-2025.
- Edelman M.J., Clamon G., Kahn D., Magram M., Lister-James J., Line B.R. Targeted radiopharmaceutical therapy for advanced lung cancer: phase I trial of rhenium Re188 P2045, a somatostatin analog //J. Thorac. 0ncol. 2009. V. 4, N 12. P. 1550-1554.
- Baum P., Singh A., Kulkarni H., Bernhardt P., Ryden T., Schuchardt C., Gracheva N., Grundler P.V., Köster U., Müller D., Pröhl M., Zeevaart J.R., Schibli R., van de Meulen N.P. First-in-humans application of 161Tb: a feasibility study using 161Tb-D0TAT0C //J. Nucl. Med. 2021. V. 62, N 10. P. 1391-1397.
- Loveless C.S., Radford L.L., Ferran S.J., Queern S., Shepherd M.R., Lapi S.E. Photonuclear production, chemistry, and in vitro evaluation of the theranostic radionuclide 47Sc //EJNMMI Res. 2019. V. 9, N 1. P. 42.
- King A.P., Lin F.I., Escorcia F.E. Why bother with alpha particles? //Eur. J. Nucl. Med. Mol. Imaging. 2021. V. 49, N 1. P. 7-17.
- Norenberg J.P., Krenning B.J., Konings I.R., Kusewitt D.F., Nayak T.K., Anderson T.L., de Jong M., Garmestani K., Brechbiel M.W., Kvols L.K. 213Bi-[D0TA0, Tyr3]octreotide peptide receptor radionuclide therapy of pancreatic tumors in a preclinical animal model //Clin. Cancer Res. 2006. V. 12. P. 897-903.
- Miederer M., Henriksen G., Alke A., Mossbrugger I., Quintanila-Martinez L., Senekowitsch-Schmidtke R., Essler M. Preclinical evaluation of the alpha-particle generator nuclide 225Ac for somatostatin receptor radiotherapy of neuroendocrine tumors //Clin. Cancer Res. 2008. V. 14, N 11. P. 3555-3561.
- Rozgaja Stallons T.A., Saidi A., Tworowska I., Delpassand E.S., Torgue J.J. Preclinical investigation of 212Pb-DOTAMTATE for peptide receptor radionuclide therapy in a neuroendocrine tumor model //Mol. Cancer. Ther. 2019. V. 18, N 5. P. 1012-1021.
- Kratochwil C., Bruchertseifer F., Giesel F.L. Ac-225-DOTATOC - dose finding for alpha particle emitter based radionuclide therapy of neuroendocrine tumors //Eur. J. Nucl. Med. Mol. Imaging. 2015. V. 42, N S1. P. S36.
- Zhang J., Kulkarni H.R., Baum R.P. 225Ac-DOTATOC-targeted somatostatin receptor a-therapy in a patient with metastatic neuroendocrine tumor of the thymus, refractory to ß-radiation //Clin. Nucl. Med. 2021. V. 46, N 12. P. 1030-1031.
- Kratochwil C., Apostolidis L., Rathke H., Apostolidis C., Bicu F., Bruchertseifer F., Choyke P.L., Haberkorn U., Giesel F.L., Morgenstern A. Dosing 225Ac-DOTATOC in patients with somatostatin-receptor-positive solid tumors: 5-year follow-up of hematological and renal toxicity //Eur. J. Nucl. Med. Mol. Imaging. 2021. V. 49, N 1. P. 54-63.
- Ballal S., Yadav M.P., Bal C., Sahoo R.K., Tripathi M. Broadening horizons with 225Ac-DOTATATE targeted alpha therapy for gastroenteropancreatic neuroendocrine tumour patients stable or refractory to 177Lu-DOTATATE PRRT: first clinical experience on the efficacy and safety //Eur. J. Nucl. Med. Mol. Imaging. 2020. V. 47, N 4. P. 934-946.
- Delpassand E.S., Tworowska I., Esfandiari R., Torgue J., Hurt J., Shafile A., Nunez R. Targeted alphaemitter therapy with 212Pb-DOTAMTATE for the treatment of metastatic SSTR-expressing neuroendocrine tumors: first-in-human, dose-escalation clinical trial //J. Nucl. Med. 2022. V. 63, N 3. P. jnumed.121.263230.
- Ginj M., Zhang H., Waser B., Cescato R., Wild D., Wang X., Erchegyi J., Rivier J., Mäcke H.R., Reubi J.C. Radiolabeled somatostatin receptor antagonists are preferable to agonists for in vivo peptide receptor targeting of tumors //Proc. Natl. Acad. Sci. USA. 2006. V. 103, N 44. P. 16436-16441.
- Cescato R., Waser B., Fani M., Reubi J.C. Evaluation of 177Lu-DOTA-sst2 antagonist versus 177Lu-DOTA-sst2 agonist binding in human cancers in vitro //J. Nucl. Med. 2011. V. 52, N 12. P. 1886-1890.
- Wang X. Comprehensive evaluation of a somatostatin-based radiolabelled antagonist for diagnostic imaging and radionuclide therapy //Eur. J. Nucl. Med. Mol. Imaging. 2012. V. 39, N 12. P. 1876-1885.
- Nicolas G.P., Mansi R., McDougall L., Kaufmann J., Bouterfa H., Wild D., Fani M. Biodistribution, pharmacokinetics, and dosimetry of 177Lu-, 90Y-, and ^In-labeled somatostatin receptor antagonist OPS201 in comparison to the agonist 177Lu-DOTATATE: the mass effect //J. Nucl. Med. 2017. V. 58, N 9. P. 1435-1441.
- Dalm S.U., Nonnekens J., Doeswijk G.N., de Blois E., van Gent D.C., Konijnenberg M.W., de Jong M. Comparison of the therapeutic response to treatment with a 177Lu-labeled somatostatin receptor agonist and antagonist in preclinical models //J. Nucl. Med. 2016. V. 57, N 2. P. 260-265.
- Wild D., Fani M., Fischer R., Del Pozzo L., Kaul F., Krebs S., Fischer R., Rivier J., Reubi J.C., Maecke H.R., Weber W.A. Comparison of somatostatin receptor agonist and antagonist for peptide receptor radionuclide therapy: a pilot study //J. Nucl. Med. 2014. V. 55, N 8. P. 1248-1252.
- Reidy-Lagunes D., Pandit-Taskar N., O'Donoghue J.A., Krebs S., Staton K.D., Lyashchenko S.K., Lewis J.S., Raj N., Gönen M., Lohrmann C., Bodei L., Weber W.A. Phase I trial of well-differentiated neuroendocrine tumors (NETs) with radiolabeled somatostatin antagonist 177Lu-satoreotide tetraxetan //Clin. Cancer Res. 2019. V. 25, N 23. P. 6939-6947.
- Schuchardt C., Kulkarni H., Singh A., Müller D., Fani M., Maecke H., Baum R. Peptide receptor radionuclide therapy using somatostatin receptor antagonists: first results of biodistribution and dosimetry of Lu-177 DOTA-LM3 compared to Lu-177 DOTATOC //J. Nucl. Med. 2019. V. 60, N S1. P. 352.
- Baum R.P., Zhang J., Schuchardt C., Müller D., Mäcke H. First-in-humans study of the SSTR antagonist 177Lu-DOTA-LM3 for peptide receptor radionuclide therapy in patients with metastatic neuroendocrine neoplasms: dosimetry, safety, and efficacy //J. Nucl. Med. 2021. V. 62, N 11. P. 1571-1581.
- Borgna F., Haller S., Monne Rodriguez J.M., Ginj M., Grundler P.V., Zeevaart J.R., Köster U., Schibli R., van der Meulen N.P., Müller C. Combination of terbium-161 with somatostatin receptor antagonists -a potential paradigm shift for the treatment of neuroendocrine neoplasms //Eur. J. Nucl. Med. Mol. Imaging. 2022. V. 49, N 4. P. 1113-1126.
- Fani M., Nicolas G.P., Wild D. Somatostatin receptor antagonists for imaging and therapy //J. Nucl. Med. 2017. V. 58, N S2. P. 61S-66S.


