Differential Expression of Phenylalanine Ammonia-lyase Between Barley Spot Blotch (Cochliobolus sativus) and Net Spot Blotch (Pyrenohora teres f. sp. maculata)
Author: Arabi M.I.E., Al-Shehadah E., Jawhar M.
Journal: Журнал стресс-физиологии и биохимии @jspb
Article in issue: 1 т.22, 2026.
Free access
Cochliobolus sativus and Pyrenophora teres f. sp. maculata, are economically important barley pathogens worldwide. Although they are very different in vegetative and reproductive forms, their spot blotch symptoms are quite similar, so the question is whether barley reacts to the disease similarly. Phenylpropane metabolic pathway plays a crucial role in plant responses to fungal diseases. In the present work, expression of phenylalanine ammonia-lyase (PAL) gene was monitored in susceptible barley genotype at early time points of infection using quantitative real-time PCR (qPCR). Data displayed significant variance in PAL expression between barley C. sativus and P. teres interactions as compared to the non-infected controls. It is remarkable that PAL expression and spot severity were higher and faster in plants infected with C. sativus as compared with those infected with P. teres during the infection process, with a maximum PAL expression (3.55 and 1.43-fold) respectively, at 96h post infection. This indicates that the severity of spot blotch symptoms seems to be associated with the degree of gene expression that will require further investigation. This work may provide useful information regarding barley reaction to two foliar pathogens causing similar disease symptoms.
Barley (Hordeum vulgare L.), spot blotch, Net spot blotch, Cochliobolus sativus, Pyrenophora teres f. sp. maculata, PAL expression, RT-PCR
Short address: https://sciup.org/143185421
IDR: 143185421
Text of the scientific article Differential Expression of Phenylalanine Ammonia-lyase Between Barley Spot Blotch (Cochliobolus sativus) and Net Spot Blotch (Pyrenohora teres f. sp. maculata)
Cochliobolus sativus (Ito & Kurib.) Drechsl. ex Dast. [anamorph: Bipolaris sorokiniana (Sacc. in Sorok.) Shoem.], and Pyrenophora teres Drechsler f. sp. maculata [anamorph Drechslera teres (Sacc.) Shoem], the causing agents of barley ( Hordeum vulgare ) spot blotch net spot blotch diseases, respectively, are two of the most important pathogens that cause significant yield losses in warm and humid regions throughout the world (Al-Sadi 2021; Bakonyi et al., 2024). The two pathogens are very different morphologically, while their spot blotch symptoms are quite similar, so it is confusing to distinguish spot blotch caused by each one (Mathre 1997).
During the process of infection, each pathogen undergoes two developmental phases. The first phase is biotrophic and asymptomatic and it is characterized by penetration into epidermal cells and spread within the mesophyll. The second phase occurs after a few days and manifests by the appearance of chlorosis and necrosis as spot blotch, marking the transition of the pathogen into a necrotrophic stage (Smedegård-Petersen 1971; Kumar et al., 2002). The first symptoms appear at the end of winter and the beginning of spring, often during the tilling phase. The spot-type symptoms of C. sativus are very often confused with barley net spot blotch caused by P. teres f. sp. maculata , and examination of the spore type is necessary to characterize the causal agent (Mathre 1997; McLean et al., 2009). Hence, barley plants have to orchestrate various defense mechanisms against pathogens with contrasting lifestyles. For this, in depth knowledge on the response of barley to spot blotch is required.
Barley plants respond to both C. sativus and P. teres pathogens by activating different mechanisms, the infection cycle begins with the development of penetration structures triggered by the perception of chemical and/or physical signals from the plant surface. During the initial interaction, the expression of phenylalanine ammonia lyase (PAL) can adapt to adversity or resist pathogen invasion by participating in synthesizing important secondary metabolites through phenylpropanoid metabolism (Huang et al., 2010; Arabi et al., 2020). The products of PAL in barley plays an important role in the metabolic pathway of plant disease resistance substances such as lignin and phytoprotectin (Koukal and Conn 1966; Feduraev et al., 2021), and had a functional role against fungal pathogen in wheat (Zhan et al., 2022), tomato (Farahani et al., 2016) and tobacco (Way et al., 2002).
Quantitative PCR (qPCR) is an effective method to analyze modulations in gene expression due to its efficiency to detect and precisely quantify the target genes after plant infection by pathogens (Bates et al., 2001) Presently, the understanding of metabolic processes after barley infection with spot blotch is limited. To complete the picture of barley biochemical responses drawn by Arabi et al., (2020) and Al-Shehadah et al., (2022), the current study investigated the PAL differential expression during barley interaction with spot blotch caused by C. sativus and P. teres f. sp. maculata at different time points.
MATERIALS AND METHOD
Host genotype
Barley seeds of the susceptible cv. ‘WI 2291’ from Australia (Arabi et al., 2003; Arabi et al., 2004) were grown in plastic pots (25-cm in diameter) filled with sterilized peat moss in three replicates, and each replicate consisted of 8 plants. The pots were placed in a greenhouse at 20 ± 2°C with a 75 ± 5%. relative humidity
Infection with C. sativus
A single spore virulent pathotype (Pt4) of C. sativus described by Arabi and Jawhar (2003) was used in the study. Seedlings were inoculated at two youngest fully expanded leaves ( Zadoks et al., 1974) using a spraying bottle. Inoculated seedlings were kept under greenhouse conditions at 21 ± 1°C °C with a 16 h photoperiod and RH 85 ±5%. Control plants were sprayed with tap water.
Infection with P. teres f. sp. maculata
The virulent P. teres f. sp. maculata single conidium strain NB4 (Arabi et al., 2003) was used in this study. Inoculum preparation, inoculation, and post inoculation were similar to those described by Arabi et al. (1992).
RNA isolation and cDNA synthesis.
Barley seedling leaves were collected at 24, 47, 72 and 96 hours post inoculation ( hpi) and ground to a fine powder in liquid nitrogen, and total RNA was extracted using Nucleotrap mRNA mini kit (Macherey-Nagel, MN, Germany). cDNA synthesis was performed by the Quanti TectReverse Transcription Kit (Qiagen) following the manufacturer’s instructions.
Quantitative real-time PCR (qPCR)
PAL was assayed in Step One Plus, 96 well using SYBR Green Master kit (Roche, USA) according to Wen et al. (2005). The sequence of RT-PCR primers is presented in Table 1. The expression level of PAL was determined according to the 2 -ΔΔCT method of Livak and Schmittgen (2001) using Ct-values of EF1α gene for normalization. The statistical analysis was conducted using ANOVA software and Tukey's test at the 0.05 significance level.
RESULTS AND DISCUSSION
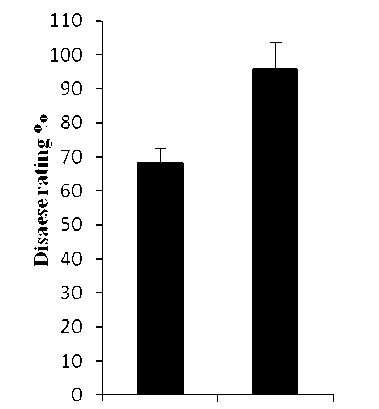
In this investigation, the susceptible barley cv. WI2291 to C. sativus and P. teres f. sp. maculata infections was used. Both pathogens produced similar spot form symptoms that consist of dark-brown, circular to elliptical lesions surrounded by a chlorotic or necrotic halo of varying width (Fig. 1), with high disease ratings for C. sativus as compared with those of P. teres (Fig. 2). These results are in agreement with our previous observations under natural field conditions (Arabi et al., 2003; 2004)
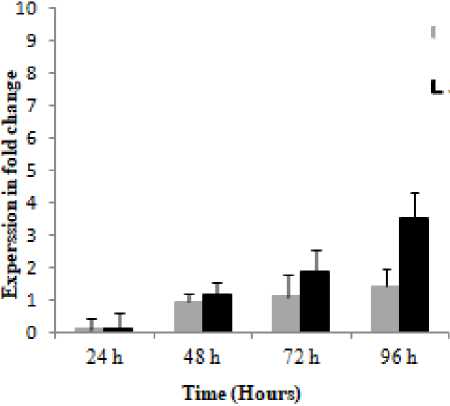
Further studies of barley interactions with spot blotch caused by C. sativus and P. teres measuring PAL expression at four early time points after pathogen challenge, showed that PAL displayed different expressions at P = 0.001, and it was noteworthy that this gene had higher expression and faster induction in C. sativus as compared with the P. teres (Fig. 3).
Data showed that 24 and 96 hpi constitute a significant starting time-point for PAL in demonstrating the differential response of barley plants towards C. sativus and P. teres and that the optimal time-point for both pathogens was 96 hpi (Fig. 3). PAL expression and spot severity were higher and faster in plants infected with C. sativus as compared with those infected with P. teres during the infection process, with a maximum expression (3.55 and 1.43-fold) respectively, at 96h post infection, therefore, the spot blotch symptom severity appears to be connected to the degree of gene expression which requisites further investigation (Figs. 2 and 3).
The present work analyzed expression profiles of PAL gene in susceptible barley genotype WI2291 infected with C. sativus and P. teres. The two pathogens differ in several morphological and biological aspects, while their spot blotch symptoms on barley are quite similar (Kumar et al., 2002; McLean et al., 2009). Therefore, we believe that analysis of phenylalanine ammonia-lyase might provide useful information for a deeper molecular research on barley reaction to spot blotch of the both pathogens. Our data showed that PAL gene exhibited differential expressions by P = 0.05, and were inversely regulated during different time points post infection of barley with both C. sativus and P. teres . Considering that this cultivar had high levels of susceptibility to both pathogens (Arabi et al., 2003; 2004).
On the other hand, it is recognized that PAL plays a crucial function in catalyzing the non-oxidative domination of phenylalanine to trans -cinnamate as a first phase in the phenylpropanoid pathway which is an important regulation stage between primary and secondary metabolism (Vogt 2010). This event might support its role in barley cell wall leakage during C. sativus and P. teres infections.
|
Table 1. Nucleotide sequences of primers used in this study |
||
|
Gene |
Gene description |
Sequence |
|
EF1α |
Elongation factor-1 Alpha |
GGCTGATTGTGCTGTGCTTA TGGTGGCATCCATCTTGTTA |
|
PAL |
Phenylalanine amino lyase |
CCA TTG ATG AAG CCA AAG CAAG ATG AGT GGG TTA TCG TTG ACGG |
(I) (II)
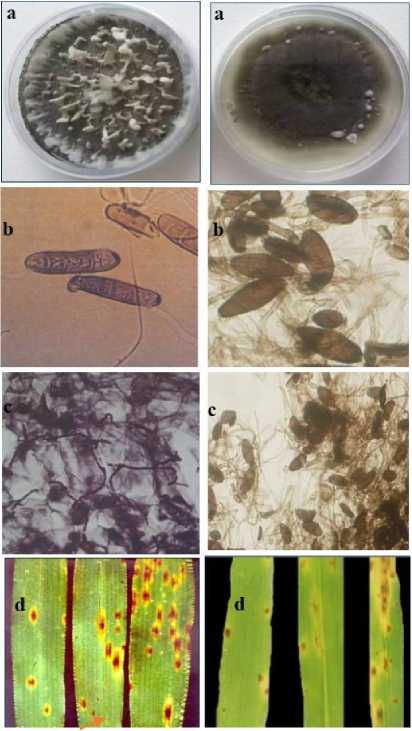
Figure 1. Vegetative forms of P. teres f. sp. maculata (I) and C. sativus (II) and their symptoms on barley leaves. Mycelium grown on PDA medium (a), conidia (b), mycelium within barley leaf (c), and spot blotch (chlorosis and necrosis) symptoms on barley leaf (red arrows) (d) .
P. teres C.sativus
Figure 2. Frequency of disease reactions on the barley susceptible cv. WI2291 by C. sativus and P. teres f. sp. maculata . Error bars are representative of the standard error (mean ± SD, n = 3).
P. teres
Figure 3. Relative expression profiles of PAL gene in the susceptible cv. ‘WI2291’ during the time course following C sativus and P. teres f. sp. maculata infections. Error bars are representative of the standard error (mean ± SD, n = 3). Data are normalized to Elongation factor 1α (EF-1α) gene expression level (to the calibrator, control 0 h, taken as 0).
^C.sath'Hs
CONCLUSION
Spot blotch of C. sativus and P. teres f. sp. maculata pathogens pose an important risk to barley cropping worldwide. In this study, our data revealed that, significant increases in spot severity and PAL expression were found upon barley challenged with the spot blotch of both fungi, with values being consistently higher in plants inoculated with C. sativus , indicating that the severity of spot blotch symptoms might be associated with the degree of gene expression that need further investigation. However, our data showed, although their spot blotch symptoms are quite similar, the barley respond differently through phenylpropane metabolic pathway.
ACKNOWLEDGMENTS
The authors would like to thank the Director General of AECS and the Head of Molecular biology and Biotechnology Department for their much appreciated help throughout the period of this research. They would also like to thank Dr. A. Al-Daoude for critical reading of the manuscript.
CONFLICTS OF INTEREST
The authors declares that they have no potential conflicts of interest.


