Implantation of embryonic cardiomyocytes into the severely damaged myocardium (after 2 infarctions): 23-year follow-up
Author: Shevchenko Yu.L., Matveev S.A., Gudymovich V.G.
Journal: Вестник Национального медико-хирургического центра им. Н.И. Пирогова @vestnik-pirogov-center
Section: Обзоры литературы
Article in issue: 2 т.16, 2021.
Free access
N this article, we describe our pioneering experience of implantation of human embryonic cardiomyocytes to a patient with severe ischemic heart lesions. In 1998, we injected suspension of human embryonic cardiomyocytes into the myocardium of a 58-year-old male patient during coronary artery bypass surgery. Long follow-up (23 years) shows that the patient, who is currently 81 years old, feels good and has a good quality of life. He has no signs of circulatory insufficiency. No complications associated with cell implantation have ever been reported during this period.
Ischemic heart disease, cell replacement therapy, implantation of embryonic cardiomyocytes
Short address: https://sciup.org/140257784
IDR: 140257784 | DOI: 10.25881/20728255_2021_16_2_142
3-летний результат имплантации культуры эмбриональных кардиомиоцитов в инвалидный (после двух инфарктов) миокард
В статье анализируется первое в мире наблюдение имплантации культур эмбриональных кардиомиоцитов пациенту с тяжелым ишемическим поражением сердца. Больному 58 лет в 1998 году во время операции аорто-коронарного шунтирования была введена в миокард предварительно подготовленная суспензия культуры эмбриональных кардиомиоцитов. При длительном наблюдении (23 года) отмечается хорошее самочувствие пациента и вполне удовлетворительное качество жизни. В настоящее время ему 81 год, чувствует себя хорошо. признаков недостаточности кровообращения нет. Осложнений, связанных с имплантацией культур клеток, на протяжении всего периода наблюдения не выявлено.
Text of the scientific article Implantation of embryonic cardiomyocytes into the severely damaged myocardium (after 2 infarctions): 23-year follow-up
Background
Organ and tissue transplantation aimed at restoration of some lost function has become a landmark of the 20th century. In fact, researchers in technics and medicine had been pursuing this goal for a long time. The development of the idea of autojector, proposed by S.S. Bryukhonenko, resulted in the creation of the cardiopulmonary bypass machine, or artificial blood circulation. Transplantations of kidney, heart, liver, and other organs has become an every-day practice.
Cell cultures were a new step in this area. It seemed that the main problem of partial organ and tissue replacement was almost addressed, and implantation of cell cultures would become an easy alternative to organ transplantation in near future. Autologous transplantation of pancreatic islet cell cultures [Pepper A. R. 2017], extracorporeal application of isolated donor hepatocytes [Lee C. A, et al., 2018], use of keratinocyte or fibroblast cultures, and a number of other studies have raised some hopes. Moreover, the use of pluripotent embryonic cells implied an opportunity of organ growing from these cells [Jackman C.P. et al., 2015]. However, new obstacles have emerged, including donorrecipient compatibility, suppression of graft rejection, ethical issues, etc. Most importantly, it is still extremely challenging to achieve appropriate conditions for the cultivation of fully functional structures outside an organism.
Preparation of a case report
Our team has become involved in this highly interesting, laborious, and even mysterious work. In a very short time, we managed to achieve interesting and unexpected results [Shevchenko Yu.L., 1998; Shevchenko Yu.L., 2005]. We conducted experimental studies aimed at:
-
• cultivation of embryonic cardiomyocytes
and their implantation into the damaged myocardium;
-
• application of endothelial cell cultures to prevent thrombosis of vascular grafts and calcification of valve grafts;
-
• stimulation of angiogenesis in tissues affected by ischemia.
Using enzymatic suspension of fetal myocardial tissue, we managed to obtain up to 95% of viable cells, the vast majority of which were cardiomyocytes (as confirmed by a positive reaction with glycogen (marker of cardiomyocytes in culture) in a cell monolayer, as well as by immunofluorescent staining with a label for alpha-actinin) (Fig. 1).
Experiments with cell cultures demonstrated some very interesting stages of the gradual syncytium development, such as single attached and scattered cells (1–2 days of incubation), formation of a monolayer and its densification (days 2–7), and formation of multicellular three-dimensional aggregates (after 7 days). Since days 3–7, cells and conjugates showed electromechanical activity, which was initially asynchronous and chaotic; after 7 days, we observed synchronous rhythmic contractions of many cell clusters (Fig. 2) in vials with organized cell aggregates with formed links between them seen as transparent strands.
The main effects of embryonic cell implantation have been studied in a number of experimental models, both with cell cultures and in a series of animal experiments. As a result, we obtained the evidence that

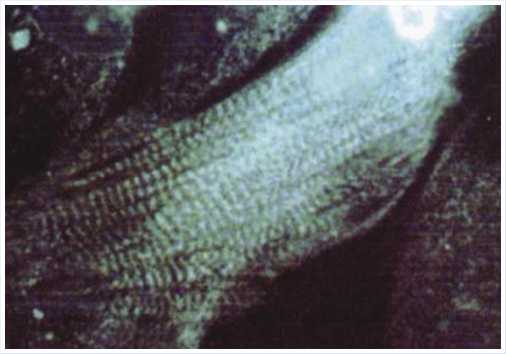
Fig. 1. Immunofluorescence label for α -actinin in a cell monolayer. Transverse striation of the contractile structures of the cardiomyocyte can be clearly seen (x800).
I
0,02 ml
0,02 ml
0,02 ml
0,02 ml
V
0,02 ml
N
7 days after acute myocardial infarction
N = 9
intact myocardium
LCA ligation infarction suspension of embryonic cardiomyocytes with a concentration of 4.8 ■ 10е cels/cm3(on average.
9.6x10* celb)
Isotonic solution suspensions of cardiomyocyte fragments (control for the inject on of dead cells)
suspension of embryonic cardiomyocytes with a concentraoon of 4.8 x 10* cels/cm3(on average.
9.6 x 10* celb)
Kotonk solution
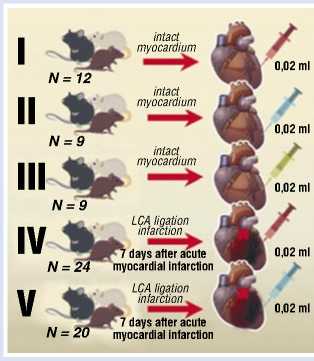
Fig. 3. Series of in vivo experiments with implantation of embryonic cardiomyocytes in the area of myocardial infarction.

Fig. 2. 5-day-old culture of embryonic cardiomyocytes in an electromagnetic field. The developing cell structures are co-directed with the impulse propagation vector (the vector is directed vertically) (x160).
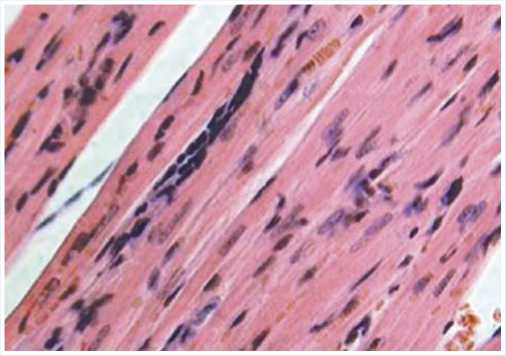
Fig. 4. Microscopy image: histological section made at the site of embryonic cardiomyocyte implantation in the area of myocardial infarction (Day 21). Muscle fiber with poorly differentiated cells. Hematoxylin and eosin stain (x200).
all of them are provided by a powerful induction of reparative processes at the site of damage. For example, experimental cell cultures were placed under prolonged hypoxic conditions and then treated with a suspension of embryonic cardiomyocytes, after which they demonstrated very high resistance to ischemia, appeared intact, and continued synchronous contractions [Shevchenko Yu.L., 1999].
We evaluated the state of embryonic cardiomyocytes implanted in the myocardium in a series of in vivo experiments (on laboratory rats) (Fig. 3). We have obtained some interesting findings; the most important one was the regression of electrocardiographic signs of acute myocardial damage caused by ligation of the branches of the left coronary artery by day 28. Histological examination demonstrated muscle cells at various stages of differentiation (Fig. 4).
Promising results obtained in animal experiments allowed us to attempt implantation of embryonic cardiomyocytes in a patient with severe ischemic heart disease. This case is described below.
Case report
A 58-year-old male patient was admitted to P.A. Kupriyanov Clinic of Cardiovascular Surgery, Military Medical Academy on January 26, 1998. He had severe angina and circulatory insufficiency (symptoms included pain after minor physical exertion, climbing to the second floor, fast walking as long as 100–150 m; all of them subsided after administration of nitroglycerin). The patient had been ill since
1979; he had two myocardial infarctions (in 1996 and 1997). Electrocardiography revealed atrial fibrillation, as well as signs of ischemic and cicatricial changes in the apical septal region and in the anterolateral wall of the left ventricle, accompanied by hypokinesia of the lateral and posterior walls of the left ventricle, and decreased overall myocardial contractility (ejection fraction 41%).
Coronary angiography findings: subocclusion of the middle third of the anterior interventricular artery, blood extravasation to the right coronary artery, subocclusion of the middle third of the right coronary artery, 90% stenosis in the distal third of the right coronary artery, stenosis of the posterior interventricular artery (Fig. 5).
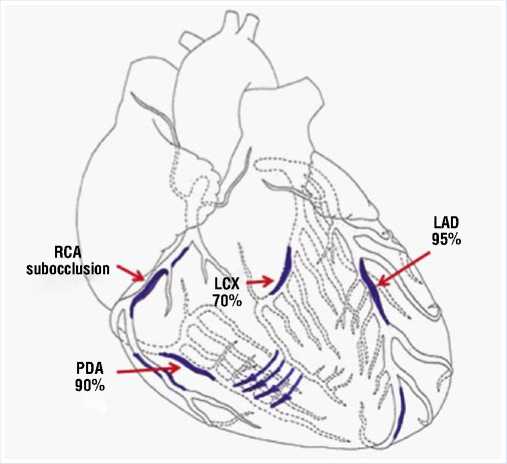
Fig. 5. Scheme demonstrating lesions to the coronary bed in a 58-year-old male patient (LAD — Left anterior descending artery, LCX — Left circumflex coronary artery, RCA — Right coronary artery, PDA — Posterior descending artery).
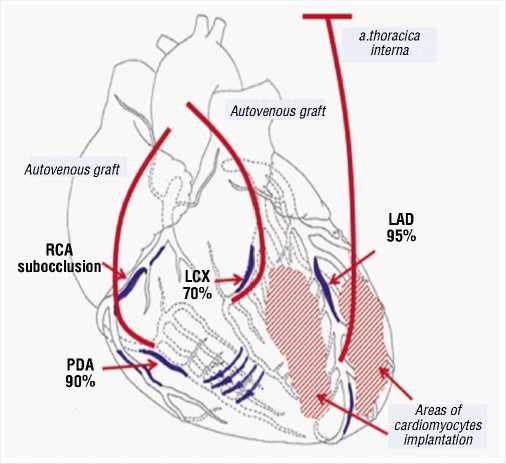
Fig. 6. Scheme of surgery (LAD — Left anterior descending artery, LCX — Left circumflex coronary artery, RCA — Right coronary artery, PDA — Posterior descending artery).
Considering extensive damage to the coronary bed, nature of the disease, no possible benefits of conservative therapy, we attempted recanalization of the anterior interventricular artery and right coronary artery. However, it was unsuccessful due to anatomical characteristics of the coronary bed in this patient. It was decided to perform direct myocardial revascularization with artificial blood circulation. It was also decided to try implantation of embryonic cardiomyocytes, since the patient had severe lesions in the coronary bed, diffuse lesions in the peripheral coronary arteries, and poor performance status. In addition to that, we realized that conservative therapy would be of no use. The patient received all necessary explanations about the surgery and gave his written consent.
On 26.02.1998, he underwent coronary artery bypass surgery that involved affected arteries (three shunts) (Fig. 6) with extracorporeal blood circulation and pharmacological/cold cardioplegia.
As soon as the main stage of the surgery was completed, heart-lung machine was disconnected, and hemodynamics was stabilized, we injected a suspension of human embryonic cardiomyocytes in the anterolateral wall and the apex of the left ventricle (4,8x106 cells) (Fig. 7). The cells were isolated from embryonic material of a 19-week fetus, obtained after abortion from a woman examined for main infections.
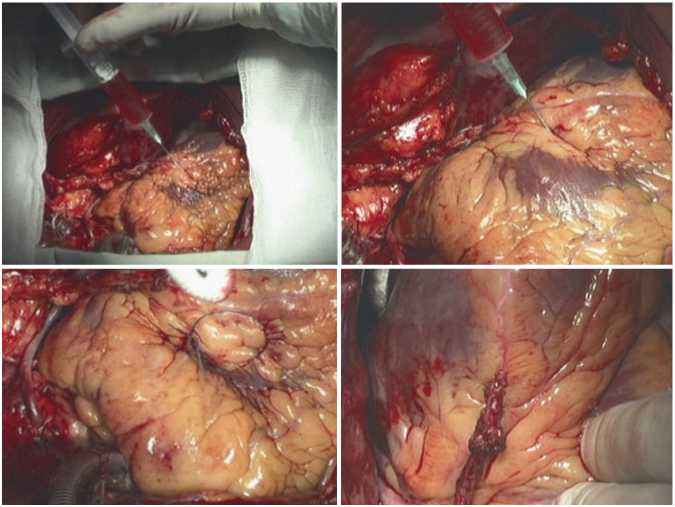
Fig. 7. Stages of injection of embryonic cardiomyocyte suspension and marking of one of implantation areas with a prolene thread.
The postoperative period was uneventful. The patient was followed-up for all this time and regularly visited the clinic for examination. He had no new angina episodes and refused nitroglycerin. He also reported an increased tolerance to physical activity. Electrocardiography demonstrated restoration of the sinus rhythm, although cicatricial changes in the myocardium were still present. Five years after this examination, we noticed an improvement of the contractile function of the myocardium (ejection fraction 56%) (Fig. 8). In 2003, we recorded a video interview with him, where he told us about this in details. He said that despite being retired, he continued to work at home, in the garden, felt good, tolerated adequate physical activity, and drove a car.
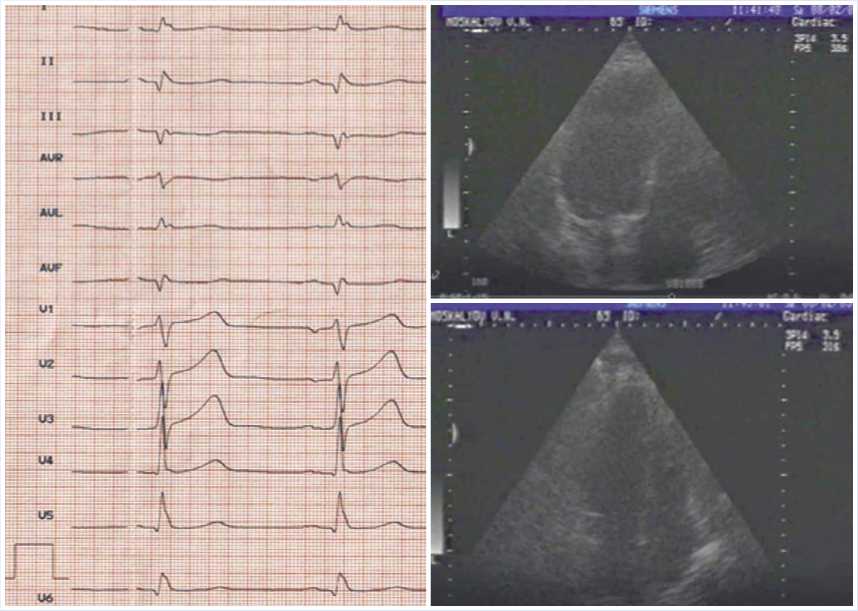
Fig. 8. Results of postoperative examination in 2003 (Cardiography and echocardiography: LV cavity in systole and diastole; good LV myocardial contractility).
For several years, the patient did not visit the clinic, probably because he had no need. However, 23 years later, we invited him to the clinic for comprehensive assessment of his treatment results. The information we obtained was very useful for the assessment of long-term outcome of this surgery. After the operation, the patient has been long working as a construction engineer. He was still active, had no problems with climbing stairs, could walk on the escalator at the age of 80, could do housework, worked in the garden, and felt good.
Conclusion
In this article, we probably report one of the first cases of embryonic cardiomyocyte implantation in the human heart (at least we failed to find any similar reports in the literature). During the time that has passed since implantation, our team had some second thoughts about this issue and changed the view as a result of better understanding of underling mechanisms, since more clinical and experimental data became available. We are now free from the illusion that implanted cardiomyocytes were integrated into the structure of the myocardium and started to function as a part of the heart. Most probably, biological mechanisms prevented this. Aggressive local factors presumably eliminated injected foreign cells. However, produced derivatives and cellular components containing growth factors acted as powerful stimulators of angiogenesis and, possibly, activated mitotic processes in own cardiomyocytes, thereby contributing to revascularization of heart tissue damaged by ischemia. Extracardial revascularization of the myocardium developed after sternotomy, pericardiotomy, and natural aseptic inflammation of the mediastinal tissues apparently played some role as well.
In conclusion, we would like to emphasize that cell therapy is possible and have good outlooks, while its cancer risk is probably exaggerated, as demonstrated in our study.
This case report is the only one in our study. Due to the ethical and legal problems at that time, and their active discussion, we decided not to perform such interventions with implantation of human embryonic cardiomyocytes anymore; however, similar studies using various experimental animals were continued.
Авторы заявляют об отсутствии конфликта интересов (The authors declare no conflict of interest).
References Implantation of embryonic cardiomyocytes into the severely damaged myocardium (after 2 infarctions): 23-year follow-up
- SHevchenko YUL, Matveev SA, Pinaev GP, et al. Pervyj opyt implantacii embrional'nyh kardiomiocitov (EK) cheloveka v kompleksnom lechenii bol'nyh s ishemicheskoj bolezn'yu serdca. IV Vserossijskij s"ezd SSKH. M.,1998. P.85.
- Шевченко Ю.Л., Матвеев С.А., Пинаев Г.П. и соавт. Первый опыт имплантации эмбриональных кардиомиоцитов (ЭК) человека в комплексном лечении больных с ишемической болезнью сердца. IV Всероссийский съезд ССХ. - М.,1998. - С.85.
- SHevchenko YUL, Matveev SA. Kletochnye tekhnologii v serdechno-sosudistoj hirurgii. M.: Medicina, 2005. 160 р.
- Шевченко Ю.Л., Матвеев С.А. Клеточные технологии в сердечно-сосудистой хирургии. - М.: Медицина, 2005. - 160 с.
- Jackman CP, Shadrin IY, Carlson AL, Bursac N. Human Cardiac Tissue Engineering: From Plu-ripotent Stem Cells to Heart Repair. Curr Opin Chem Eng. 2015 Feb; 7: 57-64. 10.1016/ j.coche.2014.11.004. DOI: 10.1016/j.coche.2014.11.004
- Lee CA, Sinha S, Fitzpatrick E, Dhawan A. Hepatocyte transplantation and advancements in alternative cell sources for liver-based regenerative medicine. J Mol Med (Berl). 2018; 96(6): 469-481. DOI: 10.1007/s00109-018-1638-5
- Pepper AR, Pawlick R, Brun A, et al. Transplantation of Human Pancreatic Endoderm Cells Reverses Diabetes Post Transplantation in a Prevascularized Subcutaneous Site. Stem Cell Reports. 2017; 8(6): 1689-1700. 10.1016/ j.stemcr.2017.05.004. DOI: 10.1016/j.stemcr.2017.05.004
- Shevchenko YuL. Experimental Validation of Embryonic Cardiomyocyte Implantation in Complex Therapy of Myocardial Failure. Progress in Biomedical Research. 1999; 4(5): 468-474.


