Influence of Benzene Hexachloride (Hexachlorocyclohexane) on Certain Endocrine Glands of South Indian Gerbil, Tatera indica cuvieri
Author: K.A. Hashim, M. Badusha
Journal: Журнал стресс-физиологии и биохимии @jspb
Article in issue: 1 т.22, 2026.
Free access
The present study investigates the histopathological effects of Benzene Hexachloride (BHC/Hexachlorocyclohexane) on selected endocrine glands of the South Indian gerbil, Tatera indica cuvieri. Fifteen adult male gerbils were divided into three groups: normal, control, and experimental. Experimental animals were administered BHC orally mixed with tapioca powder at a dose of 250 mg/kg body weight. Pituitary, thyroid, adrenal, and pancreas were examined histologically after 7 and 14 days using Haematoxylin–Eosin staining. Distinct degenerative changes were observed in pituitary, thyroid, and adrenal glands of BHC-treated animals, while the pancreas remained largely unaffected. Behavioral dullness and a reduction in body weight accompanied histological degeneration. This study demonstrates that BHC exerts toxic effects predominantly on the endocrine system of gerbils, except the pancreas.
BHC, hexachlorocyclohexane, endocrine glands, gerbil, histopathology, pesticide toxicity
Short address: https://sciup.org/143185424
IDR: 143185424
Text of the scientific article Influence of Benzene Hexachloride (Hexachlorocyclohexane) on Certain Endocrine Glands of South Indian Gerbil, Tatera indica cuvieri
It is well known that chemicals cause various types of damages and necrotic effects on the different organs of animals. Various chemicals and heavy metals that caused serious damages to animals were reported by Platt and Steward (1967), Rao and Sarkar (1975), Hasan et al. (1977), Bedwal and Mathur (1980), Bayoumi et al. (1981) and Puttannaiah and Seshadri (1983). The toxicity of various pesticides is of general importance because of their wide use in agriculture. While these pesticides destroy insects, they may have a direct or indirect action on various body systems of animals, including the endocrine system inhabiting the area. Changes induced by organo-chlorine derivatives on male reproductive organs of rats were reported by Dikshith and Datta (1972) and Starr and Clifford (1972). Roy Chowdhury et al. (1987) and Khanna et al. (1989) also reported testicular changes in rats. Changes induced by sub lethal doses of DDT on albino rats treated from four to six weeks were reported by Baronia and Sahai (1993).
The pesticide may enter the animal either directly or indirectly as an environment pollutant. Reports do indicate that herbicides like diuron, Linuron, fenuron cause functional disturbances of haemopoietic system in animals during their short & long term exposure (Hodge et al., 1967; Wager 1983; Vrochinskii et al., 1974; Perelygin et al 1975). Sub lethal levels of DDT were shown to cause increase in adrenal cortical tissue of Bobwhite quails which resulted in increased secretion of corticosteroids which in turn affects reproduction (Lehman et al., 1974). A delicate homeostatic balance exists within in the avian endocrine system which may be disturbed by feeding sub lethal levels of pesticides. This adverse effect on the endocrine system may cause subtle reproductive failures which go unnoticed until the population is greatly reduced (Wadwa et al., 1991). Only very little work has been reported to investigate the effects of pesticides in the endocrine glands of Antelope Rats, Gerbils. The present work embodies observation on the toxicity studies of Benzanehexachloride on four endocrine glands i.e., Pituitary, Thyroid, Adrenal and Pancreas of South Indian Gerbil, Tatera indica cuvieri.
MATERIALS AND METHOD
About 15 male adult healthy Gerbils of almost same size weighing 150-200 grams caught from the coastal belt of Trivandrum city and were housed in metallic wire netted cages in a room at a temperature 30-32°C and on a 12 hour light: 12 hour dark Schedule. They were fed with bits of tapioca on every third day, with water given ad libitum. After a week of acclimatization they were divided into three groups of five each. Group I served as Normal gerbils, Group II control gerbils and group III experimental gerbils. Group I were given the normal diet the Tapioca bits ad libitum, group II were fed only with 200 grams tapioca powder per animal and group III were fed with toxic dose of BHC in 200 grams of tapioca powder per animal.
250 milligrams per kilo body weight of BHC (commercial grade) with 200 grams of Tapioca powder uniformly mixed together to produce a toxic dose BHC contained food.. 200 grams of tapioca powder was given to each control animal. Likewise toxic dose of BHC contained food was given separately to each experimental antelope rat. Control animals consumed the 200 grams of tapioca powder within two to three days. But the experimental animals consumed the toxic dose of BHC + food completely within seven days. The second dose of the control food and BHC + food was given to control animal and experimental animal respectively on the seventh day when both groups had completed their Ist doses. The experimental animal completely consumed the 2nd dose of BHC food only on the 14th day. Their activity pattern was observed during the experimental period. The experiment was repeated for confirmation.
One set of normal gerbil, control gerbil and experimental gerbil were killed by decapitation on the seventh day when they completed the 1st dose of food. The endocrine glands- Pituitary, Thyroid, Adrenal and pancreas were dissected out from this sets and fixed in aqueous Bouins fluid. The other sets of animal normal, control and experimental were killed by decapitation on 14th day, 12 hours after the complete consumption of 2nd dose of BHC + food by experimental animals. The endocrine glands – Pituitary, thyroid, Adrenal and
Pancreas were dissected out and fixed in aqueous Bouins fluid. Paraffin blocks were prepared with fixed tissues and cut at 6 μ thicknesses, stained with haematoxylin and eosin, clearly observed and studied under microscope.
Haematoxylin – Eosin staining method was used to stain the fixed thin paraffin sections. The procedure followed were
-
• Deparaffinise and hydrated the sections.
-
• Stained in undiluted Ehrlic's haematoxylin for about 60-90 minutes.
-
• Washed in water
-
• Destained in aqueous picric acid or acid water if needed.
-
• Washed in running water until nuclei are blue.
-
• Dehydrated through graded series of alcohol (30%, 50%, 70%)
-
• Counter stain in 1% solution of alcoholic eosin for about 10 minutes.
-
• Differentiated in 90% alcohol.
-
• Transferred it in absolute alcohol.
-
• Cleared in xylene.
-
• Mounted in DPX
This slide section is used for further study under microscope.
RESULTS
The pitutory gland is composed of three lobes: anterior, intermediate and posterior. The anterior lobe is a dorsal evagination called the adenohypophysis and posterior to be derived from central infundibular evagination called neurohypophysis. There is an intermediate enlargement of posterior part of adenohypophysis called intermediate lobe. (Figure 4). Normal and control Gerbils did not show notable histopathological changes in pituitary glands. The pituitary gland of BHC treated animals showed remarkable changes on the 7th day. The cells of these glands became constricted. The cells of the peripheral part separated from each other. The adenohypophyseal and neurohypophyseal parts of pituitary gland deformed. On 14 days of treatment there developed vacuoles inside the cells. There appeared binucleate stage due to the breakage of adjacent cells. Constriction and shrinkage of cells made spaces and clefts in the various parts of the gland. Certain cells broken into bits. Aggregations of haematoxylin and eosin staining granules were present here and there. It is clear that the pesticide BHC caused damage to the pituitary gland. (Figure 3 and 5).
Thyroid gland is a bilobed gland lying on the central side of the trachea. The gland is composed of varying numbers of irregularly rounded thyroid follicles of different sizes. The follicles are closely packed together and each is surrounded by a basement membrane. The epithelial cells may be flattened or tall and columnar. (Figure 4). The normal and control antelope rats showed normal nature of the gland. The thyroid glands of BHC treated animals were damaged. Cells constricted and separated on the 7th day analysis. Due to the formation of vacuoles inside the thyroid follicles the section of the 14 day treated BHC fed animals appeared in the form of a loose net. Cleft formations were many when compared to pituitary gland on the 14th day analysis. Separated cells and splitted cells were found due to decaying of thyroid gland. (Figure 3 and 5).
The adrenal glands lie at the anterior end of the kidneys. They are a pair of compact bodies and each with two distinct regions, inner medulla of ectodermal origin and outer cortex of mesodermal origin. The medulla contains fine cytoplasmic granules called chromaffin granules. The adrenal cortex is found to consist of an outer zona glomerulosa layer, middle zona fasciculata layer and an inner zona reticularis . The cells of the medulla are more homogeneous in appearance than those of cortex. The cells of the medulla are irregularly arranged and secretory in nature. (Figure 5).
The adrenal glands of experimental gerbils only showed histopathological changes. On the seventh day of BHC treatment, the cortex was damaged, clumps of nuclei and space formation observed. In medulla region also shrinkage of cells, damaged chromaffin cells and space formation were noted. The experimental gerbils that consumed the 2nd dose of BHC on the 14th day, zona fasciculata of the cortex were ruptured and their nuclei were pycnotic and vacuolations developed. Some of the cells of this region were binucleated and space formation noticed. In medulla region due to shrinkage of cells spaces formed and ruptured chromaffin cells in compact mass. Chromaffin cells were hypertrophied and their nuclei were pycnotic. Due to splitting big venules were formed in medulla. BHC damaged the adrenal gland of gerbil. (Figure 3 and 5).
Pancreas is provided with two distinct kinds of tissues, the exocrine and endocrine tissues. In the exocrine portion, the secretory cells making up the alveoli and among the alveoli there lies masses of epithelial cells which are in variable size, the islets of Langerhans, the endocrine tissues. (Figure 4). On examination, the histology of the pancreas of normal, control and experimental antelope rats found to be normal after consuming completely the first and 2nd doses of food i.e. after the 7th day as well as after the 14th day. The cells of the alveolar tissue as well as the islets of Langerhans were not disturbed. It was noticed that the pesticide BHC had only very little toxicity effect in the pancreas of gerbil. (Figure 3 and 5).
Table 1. Effect of BHC on Activeness of Gerbils
|
Day spent |
Normal |
Control |
Experimental |
|
7 |
Very active |
Very active |
Less active |
|
10 |
Very active |
Active |
Dull |
|
14 |
Very active |
Active |
Semi conscious |
Note: Decrease of activeness in experimental gerbils noticed.
Table 2. Effect of HCH (BHC) in Connection to Food Consumption of Antelope Rats
|
Number of Days |
Food Consumption – Control (g) |
Food Consumption – Experimental (g) |
|
1 |
22.0 ± 0.82 |
10.3 ± 0.77 |
|
4 |
128.3 ± 5.89 |
107.0 ± 4.02 |
|
7 |
Fully consumed – Again food given |
Food consumed completely – Next dose given |
|
10 |
140.7 ± 6.82 |
121.38 ± 3.88 |
|
14 |
Fully consumed – Food completed |
Second dose of food – completed |
Note: Food consumption rate is slow in experimental antelope rats
Table 3. Effect of BHC on Body Weight of Antelope Rats
|
Number of days |
Body weight – Normal (g) |
Body weight – Control (g) |
Body weight – Experimental (g) |
|
1 |
151.33 ± 0.98 |
149.90 ± 1.02 |
156.83 ± 2.11 |
|
7 |
150.97 ± 0.88 |
147.99 ± 2.33 |
149.27 ± 3.22 |
|
14 |
150.99 ± 1.26 |
142.58 ± 2.17 |
140.01 ± 2.02 |
Note: Body weight of experimental animals was found to be decreased at a higher rate when compared to normal and control antelope rats.
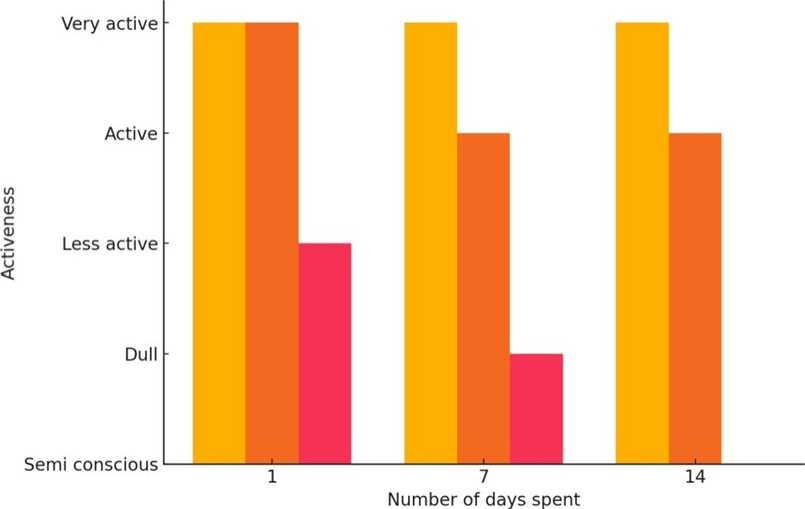
■ Normal ^H Control ^H Experimental
Figure 1. Graphical Representation of the Activeness of Normal, Control and Experimental Gerbil during Experiment
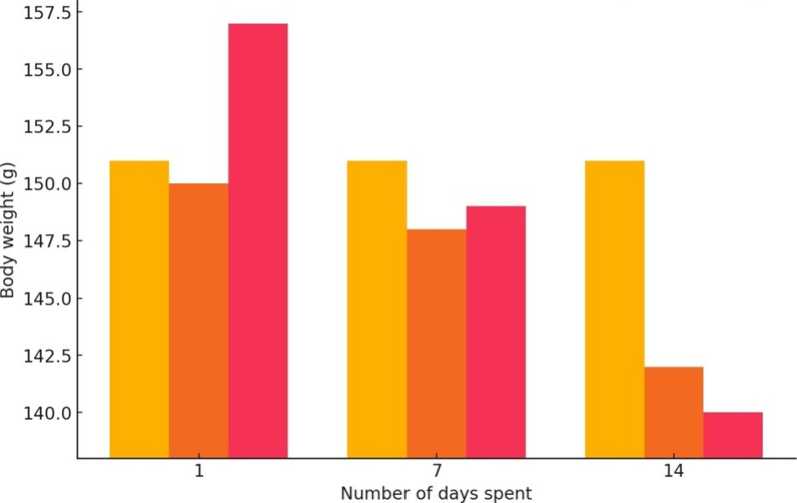
^H Normal ^H Control Mi Experimental
Figure 2. Graphical Representation of the Body Weight of Normal, Control and Experimental Gerbil during Experiment
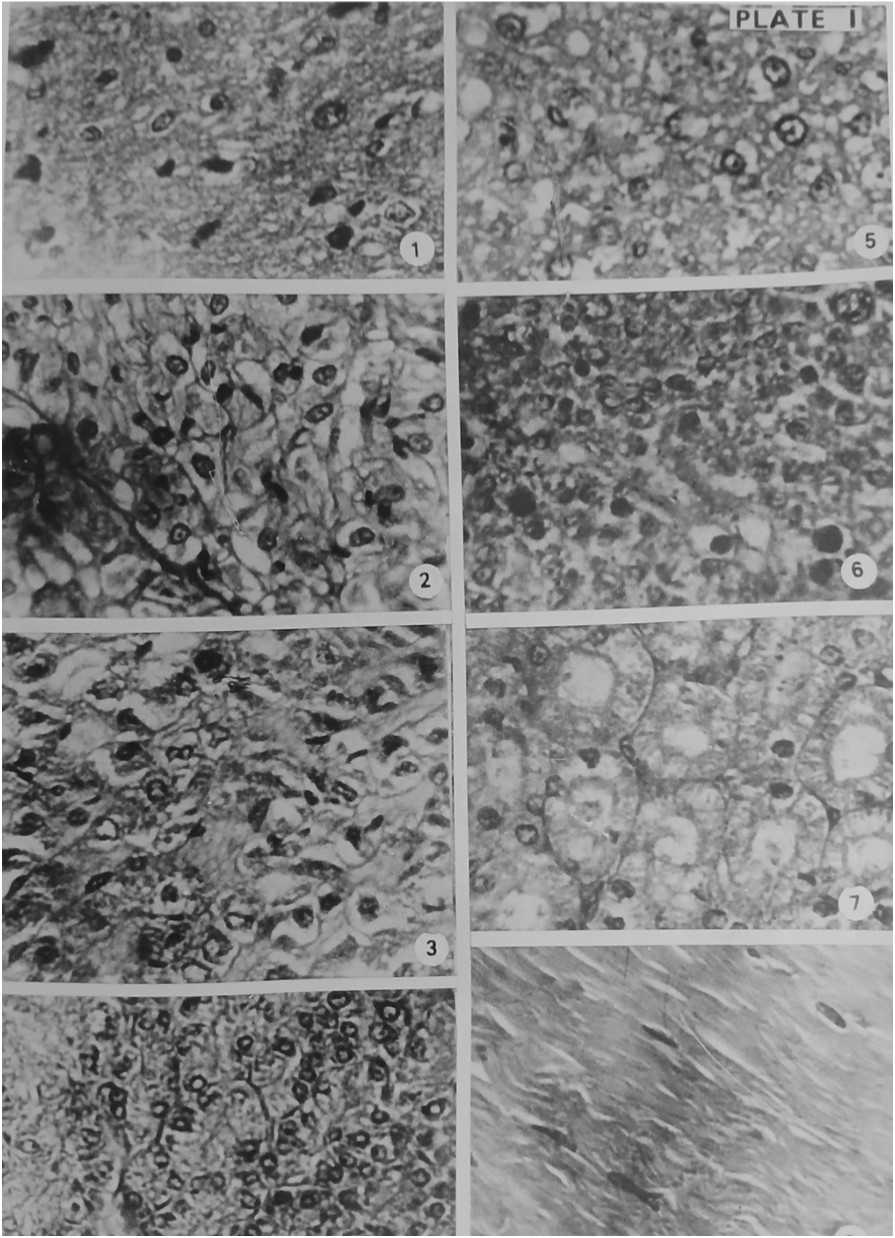
Figure 3. Section of Pituitary gland (1), Thyroid gland (2), Adrenal gland (3), Pancreas (4), Liver (5), Testis (6), Kidney (7) and Muscle tissue (8) of Experimental Gerbil after 1st dose of BHC + Food treatment. Haematoxylin- Eosin × 400
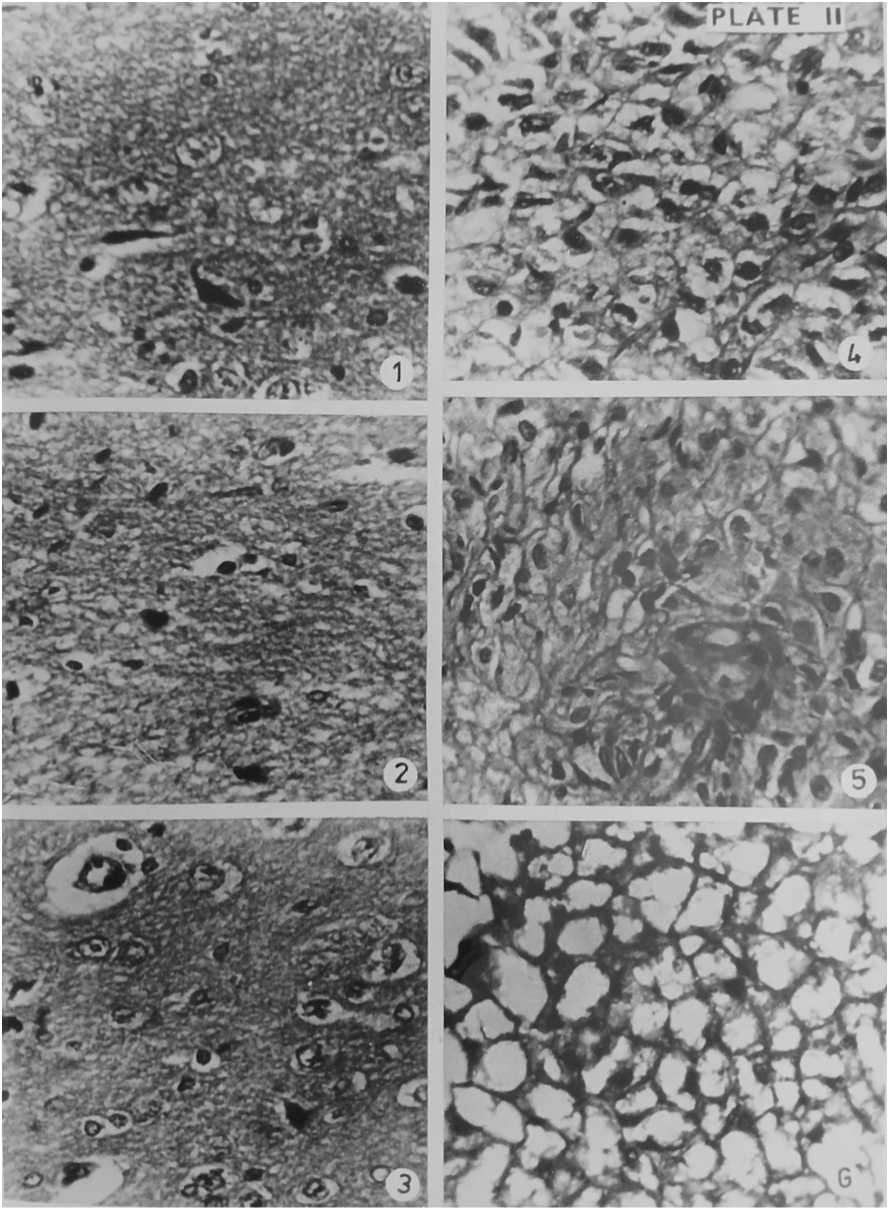
Figure 4. Section of Pituitary gland of Normal Gerbil (1), Control Gerbil (2), Experimental Gerbil after 14 days of BHC + Food treatment (3) & Section of Thyroid gland of Normal Gerbil (4), Control Gerbil (5), Experimental Gerbil after 14 days of BHC + Food treatment (6). Haematoxylin- Eosin × 400
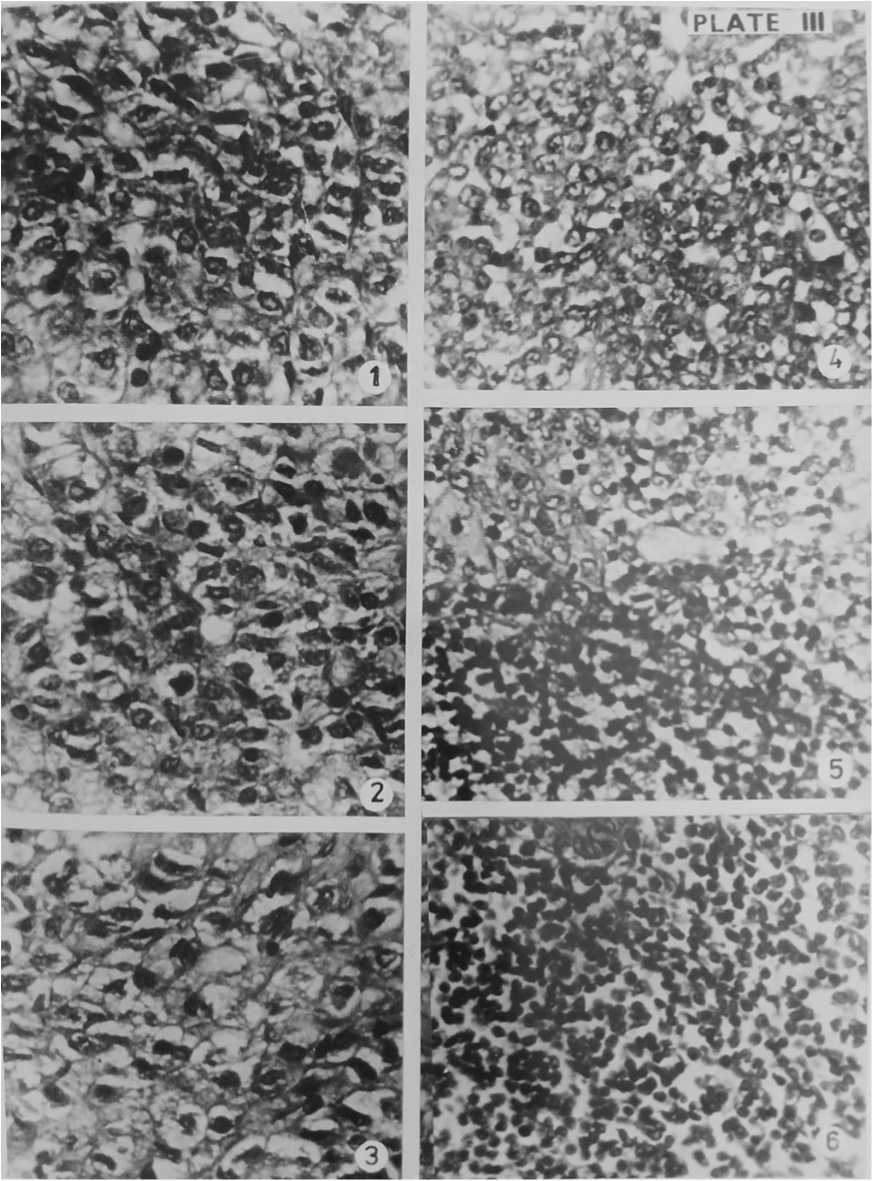
Figure 5. Section of Adrenal gland of Normal Gerbil (1), Control Gerbil (2), Experimental Gerbil after 14 days of BHC + Food treatment (3) & Section of Pancreas of Normal Gerbil (4), Control Gerbil (5), Experimental Gerbil after 14 days of BHC + Food treatment (6). Haematoxylin- Eosin × 400
DISCUSSION
In the present experiment histopathological changes in pituitary, thyroid and adrenal gland were noticed clearly in BHC treated experimental gerbils. But the pancreas of the three groups of gerbils showed normal structure. In the present experiment, the pesticide BHC affected the pituitary gland of antelope rat and showed many changes including constriction, shrinkage, breakage of cells vacuolar and space formations. Similar observations are noted by few workers. The values reported in the literature of WHO (WHO, 1985) indicates that mice and Rats are more sensitive to the insecticides. Oral administration of cypermethrin produced a dose dependent toxicity in Rats, cypermethrin degenerated the glands and other tissues in varying degree on microscopic observations (Ahamed et al., 1989). Here also the pesticide BHC damaged the pituitary gland.
In the experimental animals, like pituitary gland the thyroid also affected seriously by the pesticide. Separation of cells, splitting of cells, vacuolar and cleft formation was also noticed here. The gland appeared as loose broken net under micro scope. Nazareth et al. (1975) noted that certain doses of DDT in animals create the structural aberrations. He reported the pesticide induced increasing frequency of chromatid lesions in workers occupationally exposed to DDT. Considering his view deletions in the form of fragments of cells were one of the most common abnormalities of glands due to influence of pesticides. The toxic effect of BHC also damaged the thyroid gland of gerbil.
In the present study, the Adrenal gland showed serious histopathological changes. Pycnotic nuclei, vacuolations, binucleated cells, space formations, venules formation were noticed after BHC treatment. Similar observations are done by few workers in adrenal glands of rat. Shtenberg and Rybacova (1968) reported an increase in size and mitotic activity of the cells of zona glomerulosa, enlargement of cells with two nuclei in the fascicular zone induced by Carbaryl in rats. Hasan et al. (1977) observed degranulation and hypertrophy of adrenal cortex and medullary cells after exposure to 60 co irradiation. Hypertrophy due to BHC is reported here. Dikshith et al. (1980) reported small as well as big vacuoles in the adrenal glands of male rats treated with carbon tetrachloride (CCl4) and noticed necrotic cells. Roy Chowdhury et al. (1984) described significant morphological changes in the adrenal glands of rats due to low dose of lead acetate but with higher dose the chromaffin cells of adrenal medulla also atrophy. Singh et al. (1984) reported the tendency for individualisation of cells of adrenal glands after exposure to aldrin.
The pancreas is not affected by the pesticide. In our present study no sharp deviation in the structure of pancreas were noticed in the normal, control and experimental gerbils up to 7th and 14th day of treatment of BHC. Notable works in the action of pesticide on pancreas were not noticed. However our present study reveals that the pancreas resisted the toxic action of BHC. It is assumed that the degeneration of most of the endocrine glands of Antelope Rat may be due to the metabolic blocking of the pesticide and also its serious lethal actions in glands. The stable nature of the pancreas both in the direction of chemical actions and arrangement of cells may resist the action of BHC. The stability of pancreas may due to its dual function in the body, the exocrine and endocrine functions. It can be concluded that BHC causes damage to the endocrine glands of gerbil except pancreas.
CONCLUSION
BHC induces severe histopathological damage in pituitary, thyroid, and adrenal glands of Tatera indica , accompanied by behavioral dullness and body weight loss. Pancreatic tissue remains resistant to these toxic effects. The results underscore the endocrine-disrupting potential of BHC in non-target mammals.
ACKNOWLEDGMENT
The authors express sincere thanks to the department of Zoology, Mahatma Gandhi College, Thiruvananthapuram, University of Kerala for all the facilities provided throughout the study.
CONFLICTS OF INTEREST
The authors declares that they have no potential conflicts of interest.


