Key Genes for Biosynthesis and Recycling of Ascorbic Acid in M. baccata Fruit Tissues
Author: Katyshev A.I., Stavitskaya Z.O., Dudareva L.V., Rudikovskaya E.G., Fedoseeva I.V., Osipova S.V., Rudikovskii A.V.
Journal: Журнал стресс-физиологии и биохимии @jspb
Article in issue: 1 т.22, 2026.
Free access
Apple (M. × domestica (Suckow) Borkh.) is a major fruit crop worldwide, particularly in harsh climates in Siberia and the Russian Far East. Increasing the ascorbic acid content of apples is a priority in breeding. M. baccata, a frost-resistant apple variety native to these regions, contains high levels of vitamin C and is used in breeding as a donor of genes that improve the consumer qualities and winter hardiness of M. × domestica. This study analyzed the genetic characteristics that determine the accumulation of high AA content in M. baccata fruit tissues at different stages of development. A significant increase in the relative expression of the GGP3 gene was observed in tissues of all genotypes toward maturity; however, no correlation with the accumulation of high AA content was found. In M. × domestica fruits, the DHAR gene plays a dominant role in AA recycling. Experiments revealed that in M. baccata fruits, unlike in M. × domestica, expression of the ascorbic acid recycling gene MDHAR1 increased significantly at fruit maturity, exceeding the expression level of this gene in other genotypes in both the peel and pulp. This may be one of the reasons for the higher AA content in M. baccata fruits compared to M. × domestica. The obtained results indicate that M. baccata fruit tissues are characterized by a unique strategy for accumulating and maintaining AA levels. F1 hybrids partially inherit M. baccata's mechanisms for maintaining high AA levels. Further studies of gene expression and the activity of AA recycling and biosynthetic enzymes in apple trees may help in the creation of markers that will reduce the breeding time and select genotypes with increased expression of this gene already at the seedling stage.
M. baccata, M. × domestica, ascorbic acid
Short address: https://sciup.org/143185427
IDR: 143185427
Text of the scientific article Key Genes for Biosynthesis and Recycling of Ascorbic Acid in M. baccata Fruit Tissues
The importance of apples in the human diet is difficult to overestimate. They are a valuable food product that helps prevent certain diseases. There are over 10,000 apple varieties worldwide (Sedov, 2022). The intensification of fruit growing and horticulture necessitates the development of new varieties with specific characteristics, the cultivation of which will ensure a year-round supply of fresh and processed fruit.
Apple breeding strategies require the inclusion of a number of traits in new varieties. Fruit trees should be characterized by early and regular fruiting, resistance to diseases common to the region, and adaptability to the climatic conditions of the planned growing region. Furthermore, the fruit should possess valuable consumer qualities, a long shelf life, and an attractive appearance (Sedov, 2015; Pereira-Lorenzo et al. , 2018).
E.N. Sedov (2015) notes that each region of the country has its own specific criteria for developing new varieties. For example, in continental climates, emphasis should be placed on qualities such as winter hardiness, early fruiting, increased sugar and ascorbic acid (AA) content in fruits, and others (Kichina, 2011). Identifying and developing new M. baccata hybrids with the most optimal combination of agronomic and consumer qualities, as well as systematizing and analyzing biochemical parameters, including analysis of high AA accumulation, is a crucial task in adaptive breeding, facilitating the development of new varieties in risky farming conditions.
Pre-breeding is crucial for developing a new apple variety. This stage is the very first step in cultivating new variety. Understanding the genealogy of apple varieties and studying the biochemical profiles and metabolic pathways of parental species allows for a more complete assessment of the likelihood of valuable traits being expressed in offspring and is an invaluable tool in developing new varieties.
An extremely important milestone in research on the Malus genus was the sequencing of the genomes of M × domestica (Velasco et al., 2010) and M. baccata (Chen et al., 2019). This allowed the identification of genes associated with resistance, fruit flavor, such as the MADS-box genes involved in flowering and sorbitol metabolism (Zhang et al., 2024), and other agronomic characteristics. Genes and enzymes involved in the AA biosynthetic pathways in various plants were also identified and clarified.
As is known, the L-galactose pathway, or the Smirnov-Wheeler pathway (Wheeler et al. , 1998), is considered the main pathway of AA biosynthesis in plant fruits and includes ten sequential stages. The first five stages are necessary for the conversion of glucose to mannose and are reversible. The rate-limiting step in this metabolic pathway of vitamin C synthesis is considered to be the subsequent reaction, which directly leads to the synthesis of AA and is encoded by the GGP gene. It is the intensity of GGP activity that is key to AA formation and is a regulatory point throughout the entire synthesis pathway (Bulley et al. , 2012; Wang et al. , 2014). The AA recycling system is also of great importance for maintaining high AA content. The DHAR gene, which is involved in the AA recycling system, is considered to be the gene of M. × domestica that maintains AA content in fruits (Li et al. , 2011). However, it should be noted that the AA recycling system also includes the MDHAR gene. Its contribution to maintaining optimal AA levels in plant tissues may be more significant than that of DHAR for certain species or cultivars.
An international trend is to incorporate landraces and native species into breeding, which often possess a wide range of beneficial properties. Populations of wild apple species are a valuable resource for identifying and conserving allelic variants responsible for critical phenotypic traits, such as pathogen resistance, frost hardiness, and fruit flavour characteristics (Ignatov and Bodishevskaya, 2010). It should be noted that information on the metabolome of native species is currently fragmentary and limited to general data (Rudikovskaya et al., 2019). For M. × domestica, the Siberian apple (Malus baccata (L.) Borkh.) is a promising donor of beneficial traits. In addition to winter hardiness and resistance to certain fungal diseases, it is distinguished by an extremely high AA content in its fruits (Fang et al., 2017; Stavitskaya et al., 2023).
The use of native species to improve fruit quality and the agronomic properties of trees not only has practical value but also represents a significant area of fundamental research. This requires a detailed study not only of metabolic processes but also of the complex interrelations between them, which, in turn, will allow for a deeper understanding of the mechanisms of plant adaptation and productivity in various ecosystems. This will also facilitate the identification and long-term conservation of the unique gene pool of wild apple species, which is an integral part of global biodiversity.
In light of the above, the aim of this study was to investigate the genetic characteristics that determine the accumulation of high AA content in M. baccata fruit tissues at different stages of fruit development.
MATERIALS AND METHODS
The study involved fruits of the Siberian crabapple ( M. baccata (L.) Borkh.), its F1 hybrids of the Dobrynya, Ranetka purpurovaya, and Palmetta varieties, and the domestic crabapple ( M. × domestica (Suckow) Borkh.) varieties Chudnoye and Ranneye Bolonyaeva. All genotypes used were grown on the experimental plot of the Bioresource Center of the Siberian Institute of Plant Physiology and Biochemistry, Siberian Branch of the Russian Academy of Sciences (52º16´ N, 104º17´ E).
M. baccata was brought from a natural population in the Selenga River valley. The grafting material for the Dobrynya and Ranetka purpurovaya varieties was taken from the Irkutsk State University Botanical Garden (Irkutsk), the Palmetta variety from the Central Siberian Botanical Garden of the Siberian Branch of the Russian Academy of Sciences (Novosibirsk), the Chudnoye variety from the Shushenskoye State Fruit and Berry Variety Testing Plot, and the Ranneye Bolonyaeva variety from the Far Eastern Agricultural Research Institute. M. baccata was used as the rootstock. Fruits were selected 50 days after full bloom (DAFB) and at biological maturity. Fruit harvesting took place in 2020, 2021, and 2022. The peel and pulp were removed in a thin layer with a stainless-steel knife. Fruit tissue was weighed, fixed in liquid nitrogen, and stored at –27°C until analysis.
Isolation of ascorbic acid. A 0.3–0.5 g sample of plant tissue (peel and pulp) was suspended in liquid nitrogen and ground in a pre-cooled mortar until homogeneous. Then, 10 ml of 0.1% metaphosphoric acid (Sigma-Aldrich, USA) was added. The resulting solution was quantitatively transferred into centrifuge tubes and sonicated at 4°C for 10 minutes in an ultrasonic bath (Sapphire, Russia). The mixture was then centrifuged (Allegra 64R centrifuge, BeckmanCoulter Life Sciences, USA) for 10 minutes at 4°C, 8000 rpm.
Ascorbic acid content was determined by HPLC. Ascorbic acid content was determined by HPLC using a Milichrom A-02 chromatograph (Econova, Novosibirsk), Prontosil 120-5 C18AQ column, 2 × 75, 5 μm bead diameter, 35°C. A 25% methanol solution in 0.1% metaphosphoric acid (Sigma-Aldrich, USA) was used as the eluent. A standard L-ascorbic acid sample (Sigma-Aldrich, USA) was used to construct a calibration curve. Identification was performed by comparing retention time with the standard sample, UV spectrum, and by adding a standard sample of known concentration to the test sample.
RNA Isolation and Analysis. Total RNA was isolated from fruit tissues using the Spectrum Plant Total RNA kit according to the manufacturer's instructions (Sigma-Aldrich, USA). The purity and quantity of the isolated total RNA were determined using an NP80 nanophotometer (Implen, Germany). One microgram of total RNA was treated with DNase (Themo Fisher Scientific, Lithuania) before reverse transcription. The first strand of cDNA was synthesized from 1 μg of total RNA using RevertAid reverse transcriptase (Themo Fisher Scientific, Lithuania) according to the manufacturer's instructions using oligo(dT) primers.
qRT-PCR was performed using the CFX96 RealTime PCR Detection Sensor System (Bio-Rad, USA) with qPCRmix-HS SYBR (Evrogen, Russia). All reactions were performed in duplicate, containing 1 μl of cDNA template (5 ng), 5 μl of 5X qPCRmix-HS SYBR, and 0.5 μl of each primer (10 μm) in a final volume of 25
μl. The conditions were as follows: denaturation step at 95 °C for 5 min, then 50 cycles of denaturation at 95 °C for 30 s, annealing at 60 °C for 30 s, and elongation at 68 °C for 30 s. Melting curve analysis was performed. The gene-specific primer sequences of the tested genes are listed in Table 1. eIF-4A transcripts were used to standardize cDNA samples of various genes (Zubini, 2007). Data were analyzed using the ddCT method in Bio-Rad CFX Manager software (Bio-Rad Laboratories).
Statistical analysis of the experimental data was performed using the statistical analysis package in Microsoft Office Excel 2016, SigmaPlot 12.5. The experiment to determine the AA content in tissues was carried out in 3 biological replicates (n = 3). The experiment to determine the relative gene expression was carried out in 4 biological replicates (n = 4). A biological replicate was considered to be the tissues of one fruit. The obtained data were presented as the arithmetic mean, and the spread of values - as a standard deviation or standard error of the mean. The Shapiro-Wilk test was used to check the normality of distribution. Differences between the experimental data were considered statistically significant at P ≤ 0.05, P ≤ 0.01, P ≤ 0.001. In the case of a normal distribution, a paired two-sample t-test was used to prove the presence of significant differences between the means. For data deviating from a normal distribution, the presence of significant differences in data series was proven using the Mann-Whitney test. Spearman's coefficient was used for correlation analysis.
RESULTS AND DISCUSSION
AA Content in Tissue at Different Stages of Fruit Development. AA metabolism in fruits differs significantly from that in photosynthetic tissues. These differences are due to the fact that fruits undergo physiological and biochemical changes during development, ripening, and postharvest storage (Lemmens et al. , 2020). The mode of AA accumulation during fruit development varies among plant species. Three AA accumulation strategies can be distinguished: with peak accumulation during the juvenile period, with peak accumulation during the period of active growth, and with peak accumulation as fruits reach maturity (Ioannidi et al. , 2009; Luo et al. ,
2019, Mellidou et al. , 2021). In M. × domestica , AA accumulates according to the first strategy: maximum AA content is found in fruits during the juvenile period of development, then, as they reach maturity, its content declines (Li et al. , 2008).
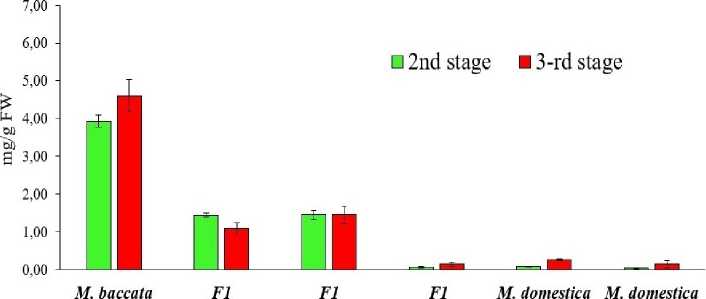
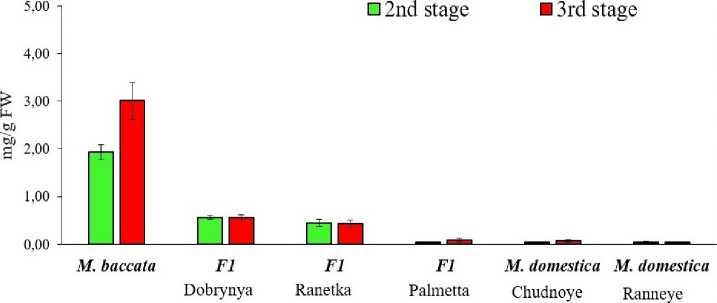
To gain insight into the mechanisms regulating AA accumulation in fruits, it is necessary to monitor its accumulation dynamics at different stages of development: 50 days after full bloom (the active growth stage, 2nd stage and the stage of biological maturity (stage 3), determined for each hybrid and species based on a combination of traits. The studied genotypes had clearly distinct differences in the dynamics of AA accumulation and maintenance (Figs. 1, 2). In the fruit tissues of the Ranetka purpurovaya and Dobrynya genotypes, AA content in the pulp did not undergo significant changes after active fruit growth. In the tissues of fruits of genotypes that were characterized by a low content of AA at the stage of active growth – Palmetta, Chudnoye, Ranneye Bolonyaeva, at the stage of maturity there was no tendency for a strong increase in AA in either the peel or the pulp.
Therefore, it is possible to identify genotypes with a tendency to accumulate high levels of AA at maturity, even at the early stages of fruit development. In M. baccata fruit tissues, AA accumulation began early in fruit development and continued until maturity in the peel and, importantly, in the pulp. M. baccata fruits apparently possess a unique strategy for accumulating and maintaining high levels of AA in fruit tissues at all stages of development, distinct from all other genotypes.
No drastic decline in AA content was observed in the tissues of any of the studied genotypes at the onset of active fruit growth, as described for M. × domestica in (Fang et al. , 2017, Li et al. , 2008). It can be assumed that the different dynamics of AA content changes during apple fruit growth are species- and cultivarspecific.
Relative expression of the GGP3, DHAR3, and MDHAR1 genes in tissues at different stages of fruit development. Currently, the L-galactose pathway is considered the main pathway for AA biosynthesis in plant cells; it comprises ten stages. The regulatory point for AA synthesis in the L-galactose pathway is the conversion of L-galactose to galactose-1-P by the enzyme galactose phosphorylase, encoded by the GGP gene. It is also known that AA content in plant organs and tissues depends on both its biosynthesis and recycling. During recycling, oxidized forms are reduced to AA by two reductases: monodehydroascorbate reductase and dehydroascorbate reductase, encoded by the MDHAR and DHAR genes. The important role of the MDHAR and DHAR genes in regulating AA levels has been demonstrated in transgenic plants of several species, where increased expression of the DHAR and MDHAR genes resulted in increased vitamin C content (Li et al., 2011).
Therefore, to understand the specifics of AA regulation in the fruit tissues of M. baccata and its hybrids, it was necessary to determine the relative expression of the GGP , DHAR , and MDHAR genes at different stages of fruit development.
A significant increase in the relative expression of the GGP3 gene at maturity was observed in the tissues of all genotypes; however, no correlation with the accumulation of high AA levels was found (Fig. 3).
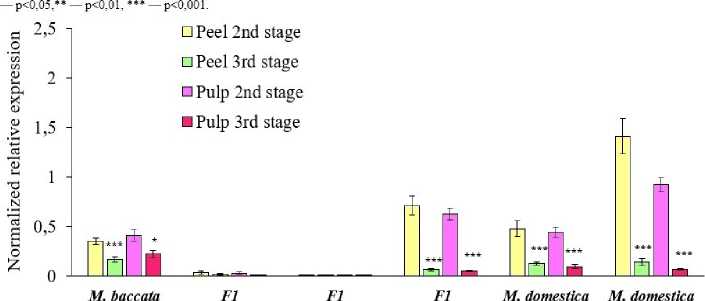
Tissues of the Dobrynya and Ranetka purpurovaya genotypes had minimal relative expression of the DHAR 3 gene during the active growth stage; this indicator did not significantly decrease towards maturity (Fig. 4).
The highest level of relative DHAR 3 expression was observed in the skin and pulp of the Ranneye Bolonyaeva and Palmetta genotypes at the 2nd stage of fruit development. By maturity, relative expression of this gene decreased: by more than 10 times in the Ranneye Bolonyaeva genotype, and by more than 11 times in the Palmetta genotype. Relative DHAR 3 expression in the tissues of the Chudnoye genotype was approximately 4
times lower at maturity. Based on the experimental results, it can be hypothesized that DHAR 3 plays a dominant role in regulating AA recycling during the second stage of fruit development in genotypes such as Palmetta, Chudnoye, and Ranneye Bolonyaeva, but is not an indicator of high AA accumulation.
Relative expression of the MDHAR1 gene significantly increased toward maturity in both the peel and pulp of the M. baccata , Dobrynya, Palmetta, and Chudnoye genotypes (Fig. 5).
Relative expression of the MDHAR1 gene at maturity in all tissues of M. baccata fruits was more than 7 times higher than that of other genotypes. MDHAR1 expression in M. baccata fruits correlated with high AA content, with a correlation coefficient of 0.75. The Chudnoye and Ranneye Bolonyaeva genotypes showed a significant increase in relative expression at maturity only in the pulp. All other genotypes studied showed only a slight increase in relative expression of this gene in the pulp at this stage, including genotypes with low vitamin content (Palmetta, Chudnoye, and Ranneye Bolonyaeva).
The results of this experiment suggest that a significant, several-fold increase in MDHAR1 expression in the pulp of M. × domestica at maturity is one of the characteristics associated with the high AA content of its ripe fruits. Based on the obtained results, it is logical to assume that MDHAR plays a major role in AA recycling in M. baccata tissues at maturity. F1 hybrids likely partially inherit this peculiarity of maintaining AA levels in fruit tissues. The DHAR gene plays a major role in AA recycling during the second stage of fruit development in the Palmetta, Chudnoye, and Ranneye Bolonyaeva genotypes. The dominance of one or another gene regulating the recycling process is likely species- and cultivar-specific.
Table 1. Gene-specific primer sequences of the tested genes.
|
Gene |
Primer sequence |
GenBank № |
|
MdMDHAR1-1 (MDHAR1) |
F:5’GAGAGAGATTGCTTCCTGACTGGTACAA-3’ R:5’GGGAAGATCAGCGTTAACTATTTCAGTGT-3’ |
XM_008393540 |
|
MdDHAR3-3 (DHAR3) |
F: 5’- AACCCTCACGGTGGCAATGGTT-3’ R: 5’- CCGAGCTTGTTGGGAACAGTGGT-3’ |
XM_008370485 |
|
MdGGP3 (GGP3) |
F: 5’CAGTTTAACGAAGGATGGAGTAGGGATT-3’ R: 5’- TCCTCCTGCAGTTCCATCCAACTAA-3’ |
XM_008385175 |
|
MdeIF-4A-8 (reference gene) |
F:5’CTTCATCGTATTGGACGAAGTGGTAGA-3’ R:5’TGTCATAGAGCATTCTCTCGTCGTCTT-3’ |
XM_008394156 |
Dobrynya Ranetka Palmetta Chudnoye Ranneye purpurovaya Bolonyaeva
Figure 1. Dynamics of AA accumulation in the peel of M. baccata , M. × domestica and F1 hybrids at the stage of active growth and fruit maturity in 2020–2022.
purpurovaya
Bolonyaeva
Figure 2. Dynamics of AK accumulation in the pulp of M. baccata , M. × domestica and F1 hybrids at the stage of active growth and fruit maturity in 2020–2022.
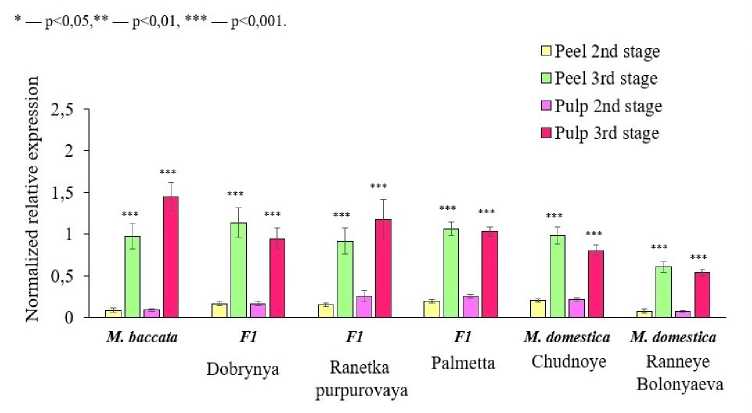
Figure 3. Dynamics of relative GGP3 expression in the tissues of M. baccata , M. × domestica and F1 hybrid fruits at the 2nd and 3rd stages of fruit development in 2020–2022.
Dobrynya
Ranetka puipurovaya
Palmetta Chudnoye Ranneye
Bolonyaeva
Figure 4. Dynamics of relative DHAR 3 expression in tissues of M. baccata , M. × domestica and their F1 hybrids at the 2nd and 3rd stages of fruit development in 2020–2022.
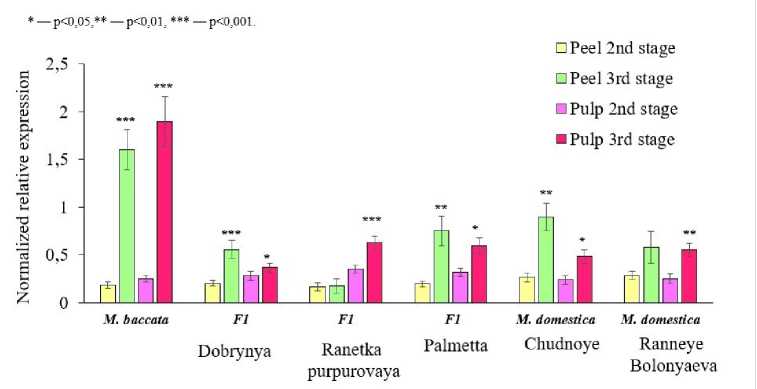
Figure 5. Dynamics of relative expression of MDHAR1 in tissues of M. baccata , M. × domestica and their F1 hybrids at the 2nd and 3rd stages of fruit development in 2020–2022.
CONCLUSION
Currently, one of the priority tasks in apple breeding is the development of varieties with improved agrobiological characteristics, including increased AA and other biologically active substance content in fruit (Sedov, 2015). This requires comprehensive information on the metabolic processes occurring in fruit at different stages of development, as well as an analysis of the relationships between them. Data on the biochemical composition, genetic, and metabolic characteristics of fruits of wild species, including M. baccata , are fragmentary and disparate. Furthermore, there are a large number of hybrid varieties (F1, F2, F3, etc.) of M. baccata with M. × domestica . Despite the widespread distribution of these plants in Siberia, the biochemical composition of their fruits and the specifics of AA metabolism also remain poorly understood.
The AA content in fruits varies significantly depending on the cultivar, species, or genotype studied (Scalzo et al. , 2005). However, M. × domestica fruit tissues are not distinguished by a high AA content—on average, this parameter reaches only 0.04 mg/g fresh weight (Bianchi et al. , 2020). The strategy of AA accumulation in M. × domestica fruits is as follows: at the early stage of fruit development, a certain amount of AA accumulates, mainly in the peel, sufficient to effectively combat oxidative stress. With further growth and development of the fruit, the AA content decreases and continues to decrease until the stage of biological maturity of the fruit (Fang et al. , 2017; Li et al. , 2008). This is possibly due to long-term selection in favor of sweet fruit taste. In the work of T. Fang et al. (Fang et al. , 2017), significant differences in the AA content were shown in the studied sample of apple trees. The highest values, up to 0.28 mg/g fresh weight, were observed primarily in wild species. Even higher AA contents, over 1.2 mg/g fresh weight, were observed in the fruit tissues of wild species typical of Eastern Siberia and the Russian Far East (Stavitskaya et al. , 2023).
Unlike M. × domestica, the fruits of M. baccata and its hybrids demonstrated a different strategy for AA accumulation. M. baccata exhibited high AA content in both the peel and pulp even at an early stage of fruit development. AA content in all tissues increased during active growth stage and continued to increase toward maturity. In the fruit tissues of F1 hybrids, AA content decreased approximately fivefold compared to M. baccata, but was still higher than in the fruit tissues of M. × domestica. In the Ranetka purpurovaya and Dobrynya genotypes, the AA content in the pulp did not change after active growth stage. Genotypes with low AA content, such as Palmetta, Chudnoye, and Ranneye Bolonyaeva, did not show any significant increase in AA content at maturity.
A significant increase in relative GGP3 gene expression was observed in tissues of all genotypes at maturity; however, no correlation with the accumulation of high AA levels was found.
AA recycling plays an important role in maintaining optimal AA levels. The DHAR and MDHAR genes and their corresponding AA recycling enzymes play a key role in this process (Suekawa et al. , 2018). In M. × domestica fruits, the DHAR gene play a dominant role in maintaining AA recycling (Li et al. , 2011).
In M. baccata fruit, the expression of genes involved in AA recycling differed from that in M. × domestica . Our study showed that in M. baccata , the relative expression of the MDHAR1 gene increased significantly at fruit maturity, exceeding the relative expression of this gene in other genotypes in both the peel and pulp. This may be one of the reasons for the higher AA content in M. baccata fruit compared to M. × domestica .
These results indicate that M. baccata fruit tissues have a unique strategy for accumulating and maintaining AA levels in the fruit. F1 hybrids partially inherit M. baccata 's mechanisms for maintaining high AA levels.
Further studies of gene expression and the activity of AA recycling enzymes in apple breeding may involve the development of a marker that will enable the selection of genotypes with increased expression of this gene already at the seedling stage. This could accelerate the development of new apple varieties with higher AA content in their fruit. Such varieties may offer a number of advantages, including improved flavor and nutritional properties and resistance to stress.
CONFLICTS OF INTEREST
The authors declare that they have no potential conflicts of interest.


