МикроРНК, ВПЧ-инфекция и цервикальный канцерогенез: молекулярные аспекты и перспективы клинического использования
Автор: Архангельская Полина Анатольевна, Бахидзе Елена Вилльевна, Берлев Игорь Викторович, Самсонов Роман Борисович, Иванов Михаил Константинович, Малек Анастасия Валерьевна
Журнал: Сибирский онкологический журнал @siboncoj
Рубрика: Обзоры
Статья в выпуске: 4 (76), 2016 года.
Бесплатный доступ
Рак шейки матки (РШМ) является одним из самых распространенных онкологических заболеваний женской репродуктивной системы и одной из лидирующих причин женской онкологической смертности. Эпидемиологические и экспериментальные данные достоверно доказывают ключевую роль вируса папилломы человека (ВПЧ) в патогенезе РШМ. Определение ДНК ВПЧ высокого канцерогенного риска является актуальным скрининговым методом для выявления пациенток с повышенным риском развития цервикальной неоплазии. Однако положительная предсказательная ценность такого анализа недостаточно высока. Анализ дополнительных показателей инфекционного процесса (вирусная нагрузка, соотношение эписомальной и интегрированной форм вируса, длительность его персистенции) может быть использован для повышения диагностической и прогностической значимости ВПЧ-тестирования. В исследованиях последнего десятилетия показано, что ВПЧ индуцирует специфические изменения профиля микроРНК инфицированной клетки, которые отражают этапность заболевания и могут иметь диагностическое / прогностическое значение. В обзоре кратко изложены современные представления об изменениях профиля клеточных микроРНК, вызываемых инфицированием ВПЧ эпителия шейки матки, приведены известные примеры участия микроРНК в цервикальном канцерогенезе и обозначены перспективы возможного использования микроРНК в качестве биомаркеров в диагностике РШМ.
Рак шейки матки, цервикальный канцерогенез, вирус папилломы человека, впч, микрорнк
Короткий адрес: https://sciup.org/140253953
IDR: 140253953 | УДК: 618.146-006.6:577.215:578.827.2 | DOI: 10.21294/1814-4861-2016-15-4-88-97
MicroRNA, HPV and cervical carcinogenesis: molecular aspects and prospects of clinical application
Cervical cancer is one of the most common cancers of the female reproductive system and a leading cause of mortality in women worldwide. Epidemiologic and experimental data have clearly demonstrated a causal role of Human Papillomavirus in cervical carcinogenesis. Determination of HPV DNA of high carcinogenic risk is relevant screening tool to identify patients with an increased risk of cervical neoplasia. However, the positive predictive value of such an analysis is not high enough. Analysis of additional indicators of infection (viral load, the ratio of episomal and integrated forms of the virus, the duration of its persistence) can be used to improve the diagnostic and prognostic value of HPV testing. In the last decade, studies have shown that HPV induces specific changes in miRNA profile of the infected cell, which reflect the phasing of the disease and may have diagnostic / prognostic value. This review summarizes the current understanding of cellular changes in the profile of microRNAs, caused by infection with HPV, given the known examples of microRNA involvement in cervical carcinogenesis and the perspectives of the possible use of microRNAs as biomarkers in the diagnosis of cervical cancer.
Текст научной статьи МикроРНК, ВПЧ-инфекция и цервикальный канцерогенез: молекулярные аспекты и перспективы клинического использования
ВПЧ-инфекция и цервикальный канцерогенез
Рак шейки матки (РШМ) остается серьезной проблемой здравоохранения в мире. По оценке глобальной заболеваемости, проводимой Всемирной организацией здравоохранения (WHO, World Health Organization) в 2012 г., в мире было зарегистрировано 528 000 новых случаев РШМ и 266 000 смертей от этого заболевания [1]. Стабильно высокие показатели заболеваемости и смертности от РШМ регистрируются в России [2, 3].
Персистенция вируса папилломы человека (ВПЧ) является триггерным фактором в цервикальном канцерогенезе. В настоящее время идентифицировано более 150 типов ВПЧ, однако только 60 из них тропны к эпителию шейки матки [4]. В отношении развития РШМ опасны генотипы ВПЧ высокого онкогенного риска, которые выявляются в образцах первичных опухолей РШМ с частотой от 60 до 99 % [5–7].
Продуктивный жизненный цикл ВПЧ тесно связан с плоскоклеточной дифференцировкой клеток. Вирус инфицирует базальные эпителиальные клетки (около 10 геномов ВПЧ на клетку), после нескольких циклов амплификации количество вирусных геномов увеличивается до нескольких десятков на клетку. Для ВПЧ-инфекции характерно латентное течение. В подавляющем большинстве случаев (по различным данным, в 80–95 % случаев) в течение 6–12 мес происходит самопроизвольная элиминация вируса, и инфицирование разрешается без каких-либо последствий для здоровья. Вероятность элиминации вируса из организма пациентки определяется совокупностью многих факторов, включая возраст, продолжительность персистенции вируса, его генотип, историю предшествующих заражений различными генотипами ВПЧ, уровень эстрогенов и иммунный статус [8].
В процессе дифференцировки зараженных эпителиальных клеток происходит репликация
вирусной ДНК и экспрессия так называемых ранних генов (early genes: E1, …, E7). На ранних этапах инфекции белки Е6 и E7 экспрессируются на низком уровне, поскольку в стадии активной репродукции вируса, сопровождающей эти этапы, экспрессия генов Е6 и Е7 регулируется репрессором их транскрипции – продуктом гена Е2. Интеграция провирусной ДНК ВПЧ в геном клеток сопровождается делецией гена Е2, потеря функциональной активности которого приводит к увеличению неконтролируемой активации экспрессии генов Е6 и Е7, запускающих процессы опухолевой трансформации. Белки Е6 и Е7 способны подавлять активность ключевых белков – регуляторов пролиферативной активности клеток (р53 и белок ретинобластомы). Бесконтрольная пролиферация инфицированных клеток обеспечивает накопление генетических повреждений и в конечном счете приводит к малиг-низации. Поэтому интеграция ДНК вируса в ДНК клетки-хозяина считается ключевым событием в процессе злокачественной трансформации эпителиальных клеток. Только интегрированная форма ВПЧ способна к злокачественной трансформации клетки-хозяина, однако лишь эписомная инфекция является продуктивной (ведущей к образованию полноценных вирусных частиц). В случае интеграции ДНК ВПЧ вирусные частицы не производятся, и инфекция является непродуктивной. Поэтому на ранних стадиях опухолевого процесса вирусная ДНК выявляется исключительно или преимущественно в эписомной форме, тогда как на поздних стадиях возрастает доля интегрированной формы [8].
Определение этиологической взаимосвязи ВПЧ с развитием рака шейки матки привело к внедрению ДНК-тестирования в качестве метода первичного скрининга для выявления пациенток с повышенной вероятностью развития РШМ. Проспективные исследования показали, что тестирование на ДНК ВПЧ имеет более высокую диагностическую чувствительность, но меньшую специфичность, по сравнению с цитологическим методом, при выявлении цервикальной интраэпителиальной неоплазии второй степени и выше (CIN2+) [9]. Поскольку ВПЧ-инфекция встречается довольно часто, особенно у молодых девушек, и в большинстве случаев является временной [5], тривиальное тестирование на наличие ДНК ВПЧ имеет относительно низкую диагностическую ценность. Стремление к более эффективному использованию ВПЧ-тестирования в диагностике РШМ стимулировало поиск дополнительных показателей ВПЧ-инфекции, анализ которых сделал бы возможным более точную оценку ее канцерогенного потенциала в каждом конкретном случае. Отечественными [6, 8, 10, 11] и зарубежными исследователями [1, 12] ведется активная работа по созданию и клинической валидации методов анализа вирусной нагрузки и интеграции вирусной ДНК, но в настоящее время не существует общепризнанного подхода к клинической интерпретации результатов таких анализов.
В целом в качестве критериев, которые суммарно отражают состояние инфекционного процесса и его канцерогенный потенциал, могут рассматриваться: генотип ВПЧ, длительность персистенции ВПЧ, вирусная нагрузка, интеграция вирусной ДНК в клеточный геном. Очевидно, что все перечисленные критерии отражают состояние собственно вируса, который играет ключевую, но не «солирующую» роль в процессе малигнизации цервикального эпителия. Поиск новых факторов, отражающих интегрированный процесс злокачественной трансформации эпителия шейки матки, а следовательно, могущих стать предиктивными факторами, имеющими диагностическую ценность, приводит к исследованию в этой связи микроРНК.
МикроРНК, ВПЧ-зависимые механизмы регуляции
Открытие молекул микроРНК (миРНК), участвующих в системе регуляции экспрессии генов на транскрипционном и посттранскрипционном уровне, существенно дополнило современную концепцию канцерогенеза и предоставило новые возможности поиска тканевых онкомаркеров и мишеней противоопухолевой терапии [13, 14]. МиРНК играют существенную роль в процессе развития всех типов онкологических заболеваний, включая РШМ [15]. Определенные сдвиги профиля миРНК всегда сопровождают процесс неопластической трансформации, отражая и отчасти определяя биологические и клинические особенности опухолевого роста. Уровень экспрессии определенных молекул миРНК часто коррелирует с такими биологическими особенностями клеток, как активность пролиферации, направление и степень дифференцировки, секреторная активность, метаболический статус.
В ходе злокачественной трансформации цервикального эпителия персистенция ВПЧ приводит к характерным изменениям профиля экспрессии миРНК и, соответственно, их регуляторной активности. В ряде исследований, осуществленных с помощью высокопропускных технологий, продемонстрированы изменения уровней экспрессии множества миРНК в клетках, инфицированных ВПЧ, но информация о биологическом значении изменений содержания конкретных миРНК пока ограничена [15]. Согласно данным многих исследований, изменения профиля клеточных миРНК при ВПЧ-инфекции в основном опосредованы активностью генов E6 и E7 [16, 17].
|
влияние вирусных белков на экспрессию клеточных мирнк |
таблица 1 |
|
|
миРНК |
Изменение |
Источник |
|
миРНК-23b |
Снижение уровня экспрессии при цервикальной неоплазии, уровень существенно понижен при раке Повышение при цервикальной интраэпителиальной неоплазии CIN 2–3 по сравне- |
[16, 28, 29, 30] |
|
миРНК-27а |
нию с CIN1 (р = 0,023) и в образцах плоскоклеточного рака по сравнению с CIN2–3 |
[26, 31, 32] |
|
(р = 0,033). Генетические варианты miR-27a определяют риск развития РШМ |
||
|
миРНК-34а |
Снижение уровня экспрессии при CIN2-3 по сравнению с образцами при CIN1 и в образцах плоскоклеточного рака по сравнению с уровнем образцов CIN2–3 |
[26, 33, 34] |
|
миРНК-143 |
Снижение уровня экспрессии ассоциировано с прогрессией трансформации цервикального эпителия |
[25, 29, 35, 36] |
|
миРНК-218 |
При СIN2–3 были статистически значимо ниже, чем при CIN1 и нормальной цитологической картине |
[24, 26, 27, 37, 38] |
|
миРНК-203 |
Угнетается в процессе злокачественной трансформации, но коррелирует с активностью метастатической диссеминации |
[26, 39, 40, 41] |
|
миРНК-127 |
Повышается при инвазивных стадиях и достигает максимальных значений при метастазировании в лимфатические узлы |
[42, 43] |
|
миРНК-21 |
Повышается и коррелирует с тяжестью дисплазии цервикального эпителия |
[31, 44, 45, 46] |
Изменения профиля миРНК, индуцируемые белком Е6
Способность вирусного белка Е6 связывать и инактивировать один из ключевых регуляторов пролиферативной активности клетки, белок p53, была продемонстрирована в нескольких исследованиях [18, 19]. p53 является транскрипционным фактором и регулирует экспрессию ряда генов, включая гены миРНК. Так, инактивация p53 приводит к блокаде экспрессии миРНК-34а, которая в нормальных условиях активируется белком p53. Причинно-следственная связь в регуляторной цепи Е6 – p53 – миРНК-34а была показана в ряде экспериментальных работ [20]. МиРНК-34а регулирует экспрессию ряда факторов контроля клеточного цикла (cyclin E2, cyclin D1, CDK4, CDK6, E2F1, E2F3, E2F5, Bcl-2, SIRT1), дисфункция этой молекулы может служить критическим событием в ходе трансформации цервикального эпителия.
Вирусный белок Е6 также регулирует экспрессию миРНК-218 и миРНК-23b. Механизм угнетения экспрессии miR-23b является p53-зависимым и ведет к активации экспрессии ферментов, определяющих инвазивный фенотип клеток (urokinasetype plasminogen activator, uPA) [16, 21]. Механизм угнетения миРНК-218 пока изучен недостаточно и, вероятно, является p53-независимым [22]. Но интересным является тот факт, что угнетение миРНК-218 приводит к активации экспрессии эпителиального маркера LAMB3, который определяет инвазивный потенциал эпителиальных клеток [22].
Изменения профиля миРНК, индуцируемые белком Е7
Патогенный эффект вирусного белка Е7 реализуется путем его взаимодействия с онкосупрессорным белком pRb, которое приводит к деградации этого белка и высвобождению активного транскрипционного фактора E2F [8]. Одним из патогенетических последствий этих реакций является активация экспрессии кластера миРНК: miR-15a, miR-15b и miR-16 [23]. Роль этих молекул в цервикальном карциногенезе еще требует изучения. МиРНК-203 является важным регулятором перехода эпителиоцитов от низкодифференцированных и активно делящихся клеток базального слоя к дифференцированным и не способным делится клеткам остистого и зернистого слоев [24]. Активность этой ключевой регуляторной молекулы блокируется вирусным белком Е7, что закономерно нарушает процесс дифференцировки цервикального эпителия.
Суммарная миРНК-регуляторная активность вирусных белков
Исследование, которое определило бы вклад каждого вирусного белка в изменение глобального профиля клеточных миРНК в развитии цервикальной неоплазии, еще не проведено. Данные многочисленных, но разнородных по дизайну и методологии работ [25, 26, 27] показывают, что персистенция ВПЧ в клетках цервикального эпителия приводит в большинстве случаев к угнетению экспрессии и, соответственно, снижению функциональной активности клеточных миРНК (табл. 1).
Дерегуляция экспрессии миРНК в результате ВПЧ-инфекции является следствием комплексного воздействия вируса на геном клетки, выражающегося как в генетических изменениях (делеции, встройки и точечные мутации), так и в эпигенетических модификациях (аберрантное метилирование ДНК, модификации гистонов). Так, экспрессия генов E6 и E7 приводит к повышению активности метилазы DNMT1. Белок E6 опосредует этот эффект путем участия в деградации белка p53, являющегося негативным регулятором DNMT1 [16, 47]. Белок E7 действует аналогично, связывая белок RBp, но показана также его способность непосредственно связывать DNMT1 [48, 49]. Повышенная таблица 2
Изменения уровней экспрессии некоторых мирнк в процессе цервикального канцерогенеза
Роль отдельных молекул миРНКв развитии РШМ
В ходе ряда исследований были выявлены миРНК, изменение содержания которых коррелирует со степенью преинвазивной неоплазии и/или характерно для инвазивного рака по сравнению с преинвазивными стадиями [15]. Так, в одном из исследований была произведена оценка уровня экспрессии миРНК: mir-21, mir-27а, mir-34а, mir-155, mir-196а, mir-203 – у ВПЧ-позитивных женщин, а также в материале, полученном из эпителия шейки матки женщин с неоплазиями различной степени и плоскоклеточным раком. Уровень экспрессии mir-27а был значительно выше в цервикальной интраэпителиальной неоплазии тяжелой степени (CIN2-3) по сравнению со слабой неоплазией CIN1 и в образцах плоскоклеточного рака по сравнению с CIN2-3. Кроме того, отмечалось снижение уровня экспрессии mir-34а при CIN2-3, по сравнению с образцами при CIN1, и в образцах плоскоклеточного рака, по сравнению с уровнем образцов CIN2-3.
Были обнаружены значимые различия у испытуемых с несколькими типами ВПЧ в mir-27а и mir-203 в CIN2-3 по сравнению с CIN1 и mir-21, и mir-27а и mir-34а в образцах плоскоклеточного рака по сравнению с уровнем образцов CIN2-3 [26].
Аналогичные примеры зависимости уровней экспрессии миРНК от тяжести неопластической трансформации эпителия шейки матки были продемонстрированы и другими исследователями (табл. 2). Причем вовлеченность некоторых миРНК в процесс трансформации цервикального эпителия была показана разными авторами в результате анализа различных групп пациенток и использования различных методов детекции. Эти результаты свидетельствуют о существовании типичных для данной патологии изменений профиля клеточных миРНК, что определяет возможность разработки методов диагностики и прогнозирования течения заболевания.
В эксперименте, проведенном на культурах клеток, инфицированных ВПЧ-31 и находящихся на разных стадиях дифференцировки, было показано, что в нормальных кератиноцитах миРНК miR-145 способна подавлять репликацию вируса, воздействуя на мишени в транскриптах генов E1 и E2. Клеточной мишенью miR-145 является транскрипционный фактор KLF-4, известный способностью индуцировать плюрипотентность стволовых клеток. Экспрессия этого белка достоверно повышена в ВПЧ-инфицированных клетках. В то же время экспрессия онкобелка E7 опосредует снижение уровня содержания miR-145 в инфицированных клетках, что стимулирует вирусную репликацию [56].
Таким образом, анализ ряда крупных исследований позволяет предполагать возможность использования определения показателей миРНК для оценки тяжести преинвазивной неоплазии, а также при дифференциальной диагностике пре-инвазивных стадий от инвазивного рака.
Возможности клинического применения миРНК в диагностике: диагностика и прогнозирование течения РШМ
Обнаружение клеточных миРНК, профиль экспрессии которых отражает состояние церви- кального эпителия, его реакцию на персистенцию ВПЧ, риск или этапность процесса малигнизации, позволяет предположить, что такие миРНК могут быть использованы в качестве потенциальных биомаркеров при диагностике или мишеней при лечении РШМ. Особенности профиля миРНК могли бы служить комплексным диагностическим параметром, отражающим совокупность многих факторов: патологического действия инфекции вирусом определенного типа, активности инфекционного процесса, толерантности клеток эпителия к неопластической трансформации и иммунного статуса пациентки. Возможность разработки методов оценки и интерпретации такого диагностического теста стимулирует новые исследования. Так, в недавней работе Q. Tian et al. сравнили метод выявления изменений миРНК с традиционным цитологическим методом, использующимся в качестве скринингового при диагностике РШМ, и подчеркнули, что при обнаружении снижения экспрессии миРНК-424 и миРНК-375 достигается более высокая чувствительность (76,0 и 74,9 % против 63,8 %), более высокая отрицательная прогностическая ценность (NPV) (от 85,7 и 85,4 % против 79,3 %) по сравнению с традиционной цитологией [57].
Аналогичная задача была поставлена авторами другого исследования, в котором изменения профиля клеточных миРНК в результате вирусной инфекции (ВПЧ-16 и ВПЧ-18) были сначала оценены в условиях in vitro, а затем характерные изменения отдельных «маркерных» молекул были проанализированы в образцах клинического материала [32]. В результате было показано, что путем оценки соотношения уровней экспрессии миРНК-25/92а и миРНК-22/29а можно дифференцировать нормальное состояние цервикального эпителия, цервикальной интраэпителиальной неоплазии и цервикальной карциномы.
В исследовании [58] на основании анализа выборки из более 100 пациенток с различными цервикальными неоплазиями было показано, что содержание единственной миРНК-125 может рассматриваться в качестве потенциального биомаркера инвазивного рака шейки матки (общая точность анализа >80 %).
Таким образом, даже анализ ограниченного числа миРНК, который может быть осуществлен недорогими и высокопропускными методами, основанными на ПЦР, может представлять диагностическую ценность. В то же время более высокая надежность анализа может быть достигнута за счет анализа широкого спектра миРНК, экспрессия которых меняется при канцерогенезе. Метаанализ данных 85 исследований изменений let-7d-5p miR-338-3p miR-200a-3p miR-192-5p miR-181b-5p miR-146a-5p miR-135b-5p miR-133a-3p miR-106a-5p miR-93-5p miR-21-5p miR-18a-5p miR-15a-5p miR-9-5p miR-25-5p miR-10a-5p
miR-425-5p miR-339-5p miR-200c-3p miR-196a-5p miR-185-5p miR-146b-5p miR-133b-5p miR-130b-3p miR-106b-5p miR-155-5p miR-34c-5p miR-20b-5p miR-15b-5p miR-196a-5p miR-92a-3p miR-16-5p
let-7d-5p miR-338-3p miR-200a-3p miR-192-5p miR-181b-5p miR-146a-5p miR-135b-5p miR-133a-3p miR-106a-5p miR-93-5p miR-21-5p miR-18a-5p miR-15a-5p miR-9-5p miR-25-5p miR-10a-5p
miR-425-5p miR-339-5p miR-200c-3p miR-196a-5p miR-185-5p miR-146b-5p miR-133b-5p miR-130b-3p miR-106b-5p miR-155-5p miR-34c-5p miR-20b-5p miR-15b-5p miR-196a-5p miR-92a-3p miR-16-5p
miR-944 miR-200b miR-189-5p miR-96-5p miR-20a-5p let-7d-5p miR-338-3p miR-200a-3p miR-192-5p miR-181b-5p miR-146a-5p miR-135b-5p miR-133a-3p miR-106a-5p miR-93-5p miR-21-5p miR-18a-5p miR-15a-5p miR-9-5p miR-25-5p miR-10a-5p
miR-1246 let-7f-5p miR-224-5p miR-142-5p miR-31-5p miR-425-5p miR-339-5p miR-200c-3p miR-196a-5p miR-185-5p miR-146b-5p miR-133b-5p miR-130b-3p miR-106b-5p miR-155-5p miR-34c-5p miR-20b-5p miR-15b-5p miR-196a-5p miR-92a-3p miR-16-5p
miR-196a-5p miR-25-5p miR-92a-3p miR-10a-5p miR-16-5p
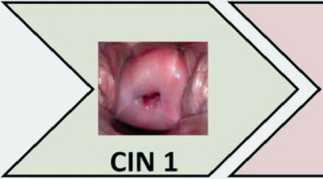
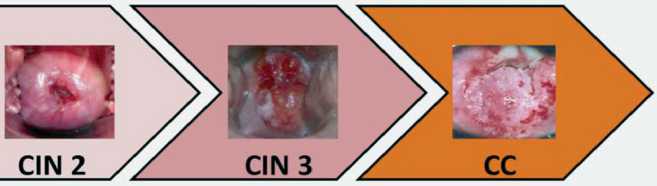
|
miR-29a miR-34a miR-29a miR-99a-5p miR-100-5p miR-99a-5p miR-199a-3p miR-203 miR-199a-3p miR-218-5p miR-218-5p miR-125b-5p miR-375 miR-494-Зр miR-617 |
miR-34a miR-29a miR-34a miR-29a miR-34a miR-100-5p miR-99a-5p miR-100-5p miR-99a-5p miR-100-5p miR-203 miR-199a-3p miR-203 miR-199a-3p miR-203 miR-149-5p miR-218-5p miR-149-5p miR-218-5p miR-149-5p miR-195-5p miR-125b-5p miR-195-5p miR-125b-5p miR-195-5p miR-376a-3p miR-375 miR-376a-3p miR-375 miR-376a-3p miR-497 miR-494-Зр miR-497 miR-494-Зр miR-497 miR-617 miR-376c-3p miR-617 miR-376c-3p miR-99b-5p miR.196b-5p miR-126-Зр mjR-i40-5p miR-1 |
Рис. 1. Изменение профиля микроРНК клеток цервикального эпителия в процессе злокачественной трансформации (перепечатано с разрешения издательства John Wiley and Sons из International Journal of Cancer [45])
профиля миРНК в процессе развития РШМ, в которые суммарно было включено более 6 000 клинических образцов, позволил идентифицировать 63 «маркерные» молекулы миРНК. Профиль этих молекул, по утверждению авторов работы, позволяет дифференцировать последовательные стадии процесса малигнизации цервикального эпителия: CIN1, CIN2, CIN3 и инвазивный рак [59]. Результаты этого масштабного исследования, представленные на рис. 1, показывают не только характерные особенности, но и прогрессивную динамику изменений профиля клеточных миРНК в ходе неопластической трансформации.
Параллельный анализ транскриптомов миРНК и миРНК в 24 цервикальных пробах клеток эпителия, находящихся на разных стадиях прогрессирования (норма, CIN1 и CIN3), не только позволил зафиксировать факт изменения экспрессии множества молекул РНК, коррелирующего со стадией, но и выявил множество пар миРНК-мРНК-мишень, экспрессия которых координированно изменяется при прогрессировании неоплазии [60]. Биоинфор-матический анализ полученных данных указывает на то, что многие миРНК, входящие в пару, являются эффективной клеточной мишенью для соответствующей миРНК. Другие же факты координированной регуляции могут быть объяснены на основании имеющихся литературных данных. Авторами на основании собственных и литературных данных предложены возможные регуляторные сети с участием миРНК, описание которых дает ключ к пониманию механизмов, лежащих в основе прогрессирования интраэпителиальной неоплазии, опосредованной ВПЧ-инфекцией [60].
В качестве диагностически значимого параметра при оценке онкогенного потенциала
Список литературы МикроРНК, ВПЧ-инфекция и цервикальный канцерогенез: молекулярные аспекты и перспективы клинического использования
- World Cancer Report 2014/Stewart B.W., Wild C.P. France: WHO Press, 2014, 630 p.
- Злокачественные новообразования в России в 2012 году (заболеваемость и смертность)/Под ред. А.Д. Каприна, В.В. Старинского, Г.В. Петрова. Москва; 2014, 250 с.
- Мерабишвили В.М., Бахидзе Е.В., Лалианци Э.И., Урманчеева А.Ф., Красильников И.А. Распространенность гинекологического рака и выживаемость больных. Вопросы онкологии, 2014; 60 (3): 288-297.
- Chen Z., Schiffman M., Herrero R., Desalle R., Anastos K., Segondy M., Sahasrabuddhe V.V., Gravitt P.E., Hsing A.W., Burk R.D. Evolution and taxonomic classification of human papillomavirus 16 (HPV16)related variant genomes: HPV31, HPV33, HPV35, HPV52, HPV58and HPV67. PLoS One. 2011; 6 (5): e20183. pone.0020183 DOI: 10.1371/journal
- Евстигнеева Л.А., Бахидзе Е.В., Семиглазов В.В., Шипицына Е.В., Золотоверхая Е.А. Роль вирусологических факторов в развитии рака шейки матки у молодых женщин. Журнал акушерства и женских болезней. 2007; 7 (5): 153.
- Коломиец Л.А., Уразова Л.Н., Севастьянова Н.В., Чуруксаева О.Н. Клинико-морфологические аспектыцервикальнойпапилломавирусной инфекции. Вопросы онкологии. 2002; 48 (1): 43-46.
- Bosch F.X., Burchell A.N., Schiffman M., Giuliano A.R., de Sanjose S., Bruni L., Tortolero-Luna G., Kjaer S.K., Munoz N. Epidemiology and natural history of human papillomavirus infections and type-specific implications in cervical neoplasia. Vaccine. 2008 Aug 19; 26 Suppl 10: K1-16 DOI: 10.1016/j.vaccine.2008.05.064
- Киселев В.И., Киселев О.И. Этиологическая роль вируса папилломы человека в развитии рака шейки матки: генетические и патогенетические механизмы. Цитокины и воспаление. 2003; 2 (4): 31-38.
- Dillner J., Rebolj M., Birembaut Ph., Petry K.U., Szarewski A., Munk C., de Sanjose S., Naucler P., Lloveras B., Kjaer S., Cuzick J., van Ballegooijen M., Clavel C., Iftner T.; Joint European Cohort Study. Long term predictive values of cytology and human papillomavirus testing in cervical cancer screening: joint European cohort study. BMJ. 2008 Oct 13; 337: a1754 DOI: 10.1136/bmj.a1754
- Киселева В.И., Крикунова Л.И., Любина Л.В., Мкртчян Л.С., Безяева Г.П., Панарина Л.В., Скугарев С.А., Куевда Д.А., Трофимова О.В., Саенко А.С. Количественная нагрузка вируса папилломы человека 16 типа и прогнозирование эффективности лечения рака шейки матки. Радиация и риск. 2011; 20 (2): 58-63.
- Комарова Е.В., Минкна Г.Н., Гаврикова М.В., Храмова О.К. Вирус папилломы человека -тестирование и генотипирование в диагностике цервикальных интраэпителиальных неоплазий. Медицина критических состояний. 2010; 1 (1): 54-61.
- Marks M., Gravitt P.E., Utaipat U., Gupta S.B., Liaw K., Kim E., Tadesse A., Phongnarisorn C., Wootipoom V., Yuenyao P., Vipupinyo C., Rugpao S., Sriplienchan S., Celentano D.D. Kinetics of DNA load predict HPV 16 viral clearance. J Clin Virol. 2011 May;51(1):44-9. doi: 10.1016/j. jcv.2011.01.011.
- Колесников Н.Н., Титов С.Е., Веряскина Ю.А., Карпинская Е.В., Шевченко С.П., Ахмерова Л.Г., Иванов М.К., Козлов В.В., Елисафенко Е.А., Гуляева Л.Ф., Жимулев И.Ф. МикроРНК, эволюция и рак. Цитология. 2013; 55 (3): 159-164.
- Di Leva G., Garofalo M., Croce C.M. MicroRNAs in cancer. Annu Rev Pathol. 2014;9:287-314 DOI: 10.1146/annurev-pathol-012513104715
- Pedroza-Torres A., Lopez-Urrutia E., Garcia-Castillo V., Jacobo-Herrera N., Herrera L.A., Peralta-Zaragoza O., Lopez-Camarillo C., De Leon D.C., Fernandez-Retana J., Cerna-Cortes J.F., Perez-Plasencia C. MicroRNAs in cervical cancer: evidences for a miRNAprofile deregulated by HPV and its impact on radio-resistance. Molecules. 2014 May 16; 19 (5): 6263-81 DOI: 10.3390/molecules19056263
- Au Yeung C.L., Tsang T.Y., Yau P.L., Kwok T.T. Human papilloma-virus type 16 E6 induces cervical cancer cell migration through the p53/microRNA-23b/urokinase-type plasminogen activator pathway. Oncogene. 2011 May 26; 30 (21): 2401-10 DOI: 10.1038/onc.2010.613
- Greco D., Kivi N., Qian K., Leivonen S.K., Auvinen P., Auvinen E. Human papillomavirus 16 E5 modulates the expression of host microRNAs. PLoS One. 2011; 6 (7): e21646 DOI: 10.1371/journal.pone.0021646
- Bernard X., Robinson P., Nomine Y., Masson M., Charbonnier S., Ramirez-Ramos J.R., Deryckere F., Trave G., Orfanoudakis G. Proteasomal degradation of p53 by human papillomavirus E6 oncoprotein relies on the structural integrity of p53 core domain. PLoS One. 2011; 6 (10): e25981 DOI: 10.1371/journal.pone.0025981
- Nomine Y., Masson M., Charbonnier S., Zanier K., Ristriani T., Deryckere F., Sibler A. P., Desplancq D., Atkinson R. A., Weiss E., Orfanoudakis G., Kieffer B., Trave G. Structural and functional analysis of E6 oncoprotein: insights in the molecular pathways of human papillomavirusmediated pathogenesis. Mol Cell. 2006 Mar 3; 21 (5): 665-78.
- Wang X., Wang H.K., McCoy J.P., Banerjee N.S., Rader J.S., Broker T.R., Meyers C., Chow L.T., Zheng Z.M. Oncogenic HPV infection interrupts the expression of tumor-suppressive miR-34a through viral oncoprotein E6. RNA. 2009 Apr; 15 (4): 637-47 DOI: 10.1261/rna.1442309
- Turner M.A., Palefsky J.M. Urokinase plasminogen activator expression by primary and HPV 16-transformed keratinocytes. Clin Exp Metastasis. 1995 Jul; 13 (4): 260-8.
- Martinez I., Gardiner A.S., Board K.F., Monzon F.A., Edwards R.P., Khan S.A. Human papillomavirus type 16 reduces the expression of microRNA-218 in cervical carcinoma cells. Oncogene. 2008 Apr 17; 27 (18): 2575-82.
- Myklebust M.P., Bruland O., Fluge O., Skarstein A., Balteskard L., Dahl O. MicroRNA-15b is induced with E2F-controlled genes in HPV-related cancer. Br J Cancer. 2011 Nov 22; 105 (11): 1719-25 DOI: 10.1038/bjc.2011.457
- Yamamoto N., Kinoshita T., Nohata N., Itesako T., Yoshino H., Enokida H., Nakagawa M., Shozu M., Seki N. Tumor suppressive microRNA-218 inhibits cancer cell migration and invasion by targeting focal adhesion pathways in cervical squamous cell carcinoma. Int J Oncol. 2013 May; 42(5): 1523-32 DOI: 10.3892/ijo.2013.1851
- Banno K., Iida M., Yanokura M., Kisu I., Iwata T., Tominaga E., Tanaka K., Aoki D. MicroRNA in cervical cancer: OncomiRs and tumor suppressor miRs in diagnosis and treatment. Scientific World J. 2014 Jan 2; 2014: 178075 DOI: 10.1155/2014/178075
- Gocze K., Gombos K., Kovacs K., Juhasz K., Gocze P., Kiss I. MicroRNA expressions in HPV-induced cervical dysplasia and cancer. Anticancer Res. 2015 Jan; 35 (1): 523-30.
- Kogo R., How C., Chaudary N., Bruce J., Shi W., Hill R.P., Zahedi P., Yip K.W., Liu F.F. The microRNA-218~Survivin axis regulates migration, invasion, and lymph node metastasis in cervical cancer. Oncotarget. 2015 Jan 20; 6 (2): 1090-100.
- Campos-Viguri G.E., Jimenez-Wences H., Peralta-Zaragoza O., Torres-Altamirano G., Soto-Flores D.G., Hernandez-Sotelo D., Alarcon-Romero Ldel C., Jimenez-Lopez M.A., Illades-Aguiar B., Fernandez-Tilapa G.miR-23b as a potential tumor suppressor and its regulation by DNAmethylation in cervical cancer. Infect Agent Cancer. 2015; 10: 42 DOI: 10.1186/s13027-015-0037-6
- Honegger A., Schilling D., Bastian S., Sponagel J., Kuryshev V., Sultmann H., Scheffner M., Hoppe-Seyler K., Hoppe-Seyler F. Dependence of intracellular and exosomal microRNAs on viral E6/E7 oncogene expression in HPV-positive tumor cells. PLoS Pathog. 2015 Mar 11; 11 (3): e1004712 DOI: 10.1371/journal.ppat.1004712
- Zheng Z.M., Wang X. Regulation of cellular miRNA expression by human papillomaviruses. Biochim Biophys Acta. 2011 Nov-Dec; 1809 (11-12): 668-77 DOI: 10.1016/j.bbagrm.2011.05.005
- Ben W., Yang Y., Yuan J., Sun J., Huang M., Zhang D., Zheng J. Human papillomavirus 16 E6 modulates the expression of host microRNAs in cervical cancer. Taiwan J Obstet Gynecol. 2015 Aug; 54 (4): 364-70 DOI: 10.1016/j.tjog.2014.06.007
- Wang X., Wang H.K., Li Y., Hafner M., Banerjee N.S., Tang S., Briskin D., Meyers C., Chow L.T., Xie X., Tuschl T., Zheng Z.M. micro-RNAs are biomarkers of oncogenic human papillomavirus infections.Proc Natl Acad Sci USA. 2014 Mar 18; 111 (11): 4262-7 DOI: 10.1073/pnas.1401430111
- Wang L., Yu J., Xu J., Zheng C., Li X., Du J. The analysis of microRNA-34 family expression in human cancer studies comparing cancer tissues with corresponding pericarcinous tissues. Curr Pharm Des. 2014; 20 (11): 1639-46.
- Yuan F., Sun R., Chen P., Liang Y., Ni S., Quan Y., Huang J., Zhang L., Gao L. Combined analysis of pri-miR-34b/c rs4938723 and TP53 Arg72Pro with cervical cancer risk. Tumour Biol. 2016 May; 37 (5): 6267-73 DOI: 10.1007/s13277-015-4467-y
- Chen Y., Ma C., Zhang W., Chen Z., Ma L. Down regulation of miR-143 is related with tumor size, lymph node metastasis and HPV16 infection in cervical squamous cancer. Diagn Pathol. 2014 Apr 28; 9: 88 DOI: 10.1186/1746-1596-9-88
- Liu L., Wang Y.L., Wang J.F. Differential expression of miR-21, miR-126, miR-143, miR-373 in normal cervical tissue, cervical cancer tissue and Hela cell. Sichuan Da Xue Xue Bao Yi Xue Ban. 2012 Jul; 43 (4): 536-9.
- Yuan W., Xiaoyun H., Haifeng Q., Jing L., Weixu H., Ruofan D., Jinjin Y., Zongji S. MicroRNA-218 enhances the radiosensitivity of human cervical cancer via promoting radiation induced apoptosis. Int J Med Sci. 2014 May 6; 11 (7): 691-6 DOI: 10.7150/ijms.8880
- ZHu Y.K., Cheng N., Hu Y., Cen Y.Z. The role of microRNAs in the pathogenesis of cervical cancer and its relationship to HPV. Sheng Li Ke Xue Jin Zhan. 2012 Aug; 43 (4): 251-6.
- Botezatu A., Goia-Rusanu C.D., Iancu I.V., Huica I., Plesa A., Socolov D., Ungureanu C., Anton G. Quantitative analysis of the relationship between microRNA124a, -34b and -203 gene methylation and cervical oncogenesis. Mol Med Rep. 2011 Jan-Feb;4 (1): 121-8 DOI: 10.3892/mmr.2010.394
- Seifoleslami M., Khameneie M.K., Mashayekhi F., Sedaghati F., Ziari K., Mansouri K., Safari A. Identification of microRNAs (miR-203/miR-7) as potential markers for the early detection of lymph node metastases in patients with cervical cancer. Tumour Biol. 2015 Oct 22.
- Zhu X., Er K., Mao C., Yan Q., Xu H., Zhang Y., Zhu J., Cui F., Zhao W., Shi H. miR-203 suppresses tumor growth and angiogenesis by targeting VEGFA in cervical cancer. Cell Physiol Biochem. 2013; 32 (1): 64-73 DOI: 10.1159/000350125
- Lee J.W., Choi C.H., Choi J.J., Park Y.A., Kim S.J., Hwang S.Y., Kim W.Y., Kim T.J., Lee J.H., Kim B.G., Bae D.S. Altered MicroRNA expression in cervical carcinomas. Clin Cancer Res. 2008 May 1; 14 (9): 2535-42 DOI: 10.1158/1078-0432.CCR-07-1231
- You W., Wang Y., Zheng J. Plasma miR-127 and miR-218 Might Serve as Potential Biomarkers for Cervical Cancer. Reprod Sci. 2015Aug; 22 (8): 1037-41 DOI: 10.1177/1933719115570902
- Tian Q., Li Y., Wang F., Li Y., Xu J., Shen Y., Ye F., Wang X., Cheng X., Chen Y., Wan X., Lu W., Xie X. MicroRNA detection in cervical exfoliated cells as a triage for human papillomavirus-positive women. J Natl Cancer Inst. 2014 Sep 4; 106 (9). pii: dju241 DOI: 10.1093/jnci/dju241
- Han Y., Xu G.X., Lu H., Yu D.H., Ren Y., Wang L., Huang X.H., Hou W.J., Wei Z.H., Chen Y.P., Cao Y.G., Zhang R. Dysregulation of miRNA-21 and their potential as biomarkers for the diagnosis of cervical cancer. Int J Clin Exp Pathol. 2015 Jun 1; 8 (6): 7131-9.
- Shishodia G., Shukla S., Srivastava Y., Masaldan S., Mehta S., Bhambhani S., Sharma S., Mehrotra R., Das B.C., Bharti A.C. Alterations in microRNAs miR-21 and let-7a correlate with aberrant STAT3 signaling and downstream effects during cervical carcinogenesis. Mol Cancer. 2015 Jun 9; 14: 116 DOI: 10.1186/s12943-015-0385-2
- Leonard S.M., Wei W., Collins S.I., Pereira M., Diyaf A., Constandinou-Williams C., Young L.S., Roberts S., Woodman C.B. Oncogenichuman papillomavirus imposes an instructive pattern of DNA methylation changes which parallel the natural history of cervical HPV infection in young women. Carcinogenesis. 2012 Jul; 33 (7): 1286-93. doi: 10.1093/carcin/bgs157.
- Burgers W.A., Blanchon L., Pradhan S., de Launoit Y., Kouzarides T., Fuks F. Viral oncoproteins target the DNA methyltransferases. Oncogene. 2007 Mar 8; 26 (11): 1650-5.
- Jimenez-Wences H., Peralta-Zaragoza O., Fernandez-Tilapa G. Human papilloma virus, DNA methylation and microRNA expression in cervical cancer (Review). Oncol Rep. 2014 Jun; 31 (6): 2467-76 DOI: 10.3892/or.2014.3142
- Wilting S.M., van Boerdonk R.A., Henken F.E., Meijer C.J., Dios-dado B., Meijer G.A., le Sage C., Agami R., Snijders P.J., Steenbergen R.D. Methylation-mediated silencing and tumour suppressive function of hsa-miR-124 in cervical cancer. Mol Cancer. 2010 Jun 26; 9: 167 DOI: 10.1186/1476-4598-9-167
- Wang J.T., Ding L., Jiang S.W., Hao J., Zhao W.M., Zhou Q., Yang Z.K., Zhang L. Folate deficiency and aberrant expression of DNA methyltransferase 1 were associated with cervical cancerization. Curr Pharm Des. 2014; 20 (11): 1639-46.
- Hu Z., Zhu D., Wang W., Li W., Jia W., Zeng X., Ding W., Yu L., Wang X., Wang L., Shen H., Zhang C., Liu H., Liu X., Zhao Y., Fang X., Li S., Chen W., Tang T., Fu A., Wang Z., Chen G., Gao Q., Li S., Xi L., Wang C., Liao S., Ma X., Wu P., Li K., Wang S., Zhou J., Wang J., Xu X., Wang H., Ma D. Genome-wide profiling of HPV integration in cervical cancer identifies clustered genomic hot spots and a potential microhomologymediated integration mechanism. Nat Genet. 2015 Feb; 47 (2): 158-63 DOI: 10.1038/ng.3178
- Calin G.A., Sevignani C., Dumitru C.D., Hyslop T., Noch E., Yendamuri S., Shimizu M., Rattan S., Bullrich F., Negrini M., Croce C.M.Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc Natl Acad Sci USA. 2004 Mar 2; 101 (9): 2999-3004.
- Wentzensen N., Vinokurova S., von Knebel Doeberitz M. Systematic review of genomic integration sites of human papillomavirus genomes in epithelial dysplasia and invasive cancer of the female lower genital tract.Cancer Res. 2004 Jun 1; 64 (11): 3878-84.
- Zubillaga-Guerrero M.I., Alarcon-Romero L de lC., Illades-AguiarB., Flores-Alfaro E., Bermudez-Morales V.H., Deas J., Peralta-Zaragoza O. MicroRNA miR-16-1 regulates CCNE1 (cyclin E1) gene expression in human cervical cancer cells. Int J Clin Exp Med. 2015 Sep 15; 8 (9): 15999-6006.
- Gunasekharan V., Laimins L.A. Human papillomaviruses modulate microRNA145 expression to directly control genome amplification. J Virol. 2013 May; 87 (10): 6037-43 DOI: 10.1128/JVI.00153-13
- Tian Q., Li Y., Wang F., Li Y., Xu J., Shen Y., Ye F., Wang X., Cheng X., Chen Y., Wan X., Lu W., Xie X. MicroRNA detection in cervical exfoliated cells as a triage for human papillomavirus-positive women. J Natl Cancer Inst. 2014 Sep 4; 106 (9). pii: dju241 DOI: 10.1093/jnci/dju241
- Ribeiro J., Marinho-Dias J., Monteiro P., Loureiro J., Baldaque I., Medeiros R., Sousa H. miR-34a and miR-125b Expression in HPV Infection and Cervical Cancer Development Biomed Res Int. 2015; 2015: 304584 DOI: 10.1155/2015/304584
- He Y., Lin J., Ding Y., Liu G., Luo Y., Huang M., Xu C., Kim T.K., Etheridge A., Lin M., Kong D., Wang K. Asystematic study on dysregulated microRNAs in cervical cancer development. Int J Cancer. 2016 Mar 15; 138 (6): 1312-27 DOI: 10.1002/ijc.29618
- Mo W., Tong C., Zhang Y., Lu H. microRNAs’ differential regulations mediate the progress of Human Papillomavirus (HPV)-induced Cervical Intraepithelial Neoplasia (CIN). BMC Syst Biol. 2015 Feb 7; 9: 4 DOI: 10.1186/s12918-015-0145-3
- Комарова Е.В., Минкна Г.Н., Гаврикова М.В., Храмова О.К. Вирус папилломы человека -тестирование и генотипирование в диагностике цервикальных интраэпителиальных неоплазий. Медицина критических состояний. 2010; 1 (1): 54-61.
- Marks M., Gravitt P.E., Utaipat U., Gupta S.B., Liaw K., Kim E., Tadesse A., Phongnarisorn C., Wootipoom V., Yuenyao P., Vipupinyo C., Rugpao S., Sriplienchan S., Celentano D.D. Kinetics of DNA load predict HPV 16 viral clearance. J Clin Virol. 2011 May;51(1):44-9. doi: 10.1016/j. jcv.2011.01.011.
- Колесников Н.Н., Титов С.Е., Веряскина Ю.А., Карпинская Е.В., Шевченко С.П., Ахмерова Л.Г., Иванов М.К., Козлов В.В., Елисафенко Е.А., Гуляева Л.Ф., Жимулев И.Ф. МикроРНК, эволюция и рак. Цитология. 2013; 55 (3): 159-164.
- Di Leva G., Garofalo M., Croce C.M. MicroRNAs in cancer. Annu Rev Pathol. 2014;9:287-314 DOI: 10.1146/annurev-pathol-012513104715
- Pedroza-Torres A., Lopez-Urrutia E., Garcia-Castillo V., Jacobo-Herrera N., Herrera L.A., Peralta-Zaragoza O., Lopez-Camarillo C., De Leon D.C., Fernandez-Retana J., Cerna-Cortes J.F., Perez-Plasencia C. MicroRNAs in cervical cancer: evidences for a miRNAprofile deregulated by HPV and its impact on radio-resistance. Molecules. 2014 May 16; 19 (5): 6263-81 DOI: 10.3390/molecules19056263
- Au Yeung C.L., Tsang T.Y., Yau P.L., Kwok T.T. Human papilloma-virus type 16 E6 induces cervical cancer cell migration through the p53/microRNA-23b/urokinase-type plasminogen activator pathway. Oncogene. 2011 May 26; 30 (21): 2401-10 DOI: 10.1038/onc.2010.613
- Greco D., Kivi N., Qian K., Leivonen S.K., Auvinen P., Auvinen E. Human papillomavirus 16 E5 modulates the expression of host microRNAs. PLoS One. 2011; 6 (7): e21646 DOI: 10.1371/journal.pone.0021646
- Bernard X., Robinson P., Nomine Y., Masson M., Charbonnier S., Ramirez-Ramos J.R., Deryckere F., Trave G., Orfanoudakis G. Proteasomal degradation of p53 by human papillomavirus E6 oncoprotein relies on the structural integrity of p53 core domain. PLoS One. 2011; 6 (10): e25981 DOI: 10.1371/journal.pone.0025981
- Nomine Y., Masson M., Charbonnier S., Zanier K., Ristriani T., Deryckere F., Sibler A. P., Desplancq D., Atkinson R. A., Weiss E., Orfanoudakis G., Kieffer B., Trave G. Structural and functional analysis of E6 oncoprotein: insights in the molecular pathways of human papillomavirusmediated pathogenesis. Mol Cell. 2006 Mar 3; 21 (5): 665-78.
- Wang X., Wang H.K., McCoy J.P., Banerjee N.S., Rader J.S., Broker T.R., Meyers C., Chow L.T., Zheng Z.M. Oncogenic HPV infection interrupts the expression of tumor-suppressive miR-34a through viral oncoprotein E6. RNA. 2009 Apr; 15 (4): 637-47 DOI: 10.1261/rna.1442309
- Turner M.A., Palefsky J.M. Urokinase plasminogen activator expression by primary and HPV 16-transformed keratinocytes. Clin Exp Metastasis. 1995 Jul; 13 (4): 260-8.
- Martinez I., Gardiner A.S., Board K.F., Monzon F.A., Edwards R.P., Khan S.A. Human papillomavirus type 16 reduces the expression of microRNA-218 in cervical carcinoma cells. Oncogene. 2008 Apr 17; 27 (18): 2575-82.
- Myklebust M.P., Bruland O., Fluge O., Skarstein A., Balteskard L., Dahl O. MicroRNA-15b is induced with E2F-controlled genes in HPV-related cancer. Br J Cancer. 2011 Nov 22; 105 (11): 1719-25 DOI: 10.1038/bjc.2011.457
- Yamamoto N., Kinoshita T., Nohata N., Itesako T., Yoshino H., Enokida H., Nakagawa M., Shozu M., Seki N. Tumor suppressive microRNA-218 inhibits cancer cell migration and invasion by targeting focal adhesion pathways in cervical squamous cell carcinoma. Int J Oncol. 2013 May; 42(5): 1523-32 DOI: 10.3892/ijo.2013.1851
- Banno K., Iida M., Yanokura M., Kisu I., Iwata T., Tominaga E., Tanaka K., Aoki D. MicroRNA in cervical cancer: OncomiRs and tumor suppressor miRs in diagnosis and treatment. Scientific World J. 2014 Jan 2; 2014: 178075 DOI: 10.1155/2014/178075
- Gocze K., Gombos K., Kovacs K., Juhasz K., Gocze P., Kiss I. MicroRNA expressions in HPV-induced cervical dysplasia and cancer. Anticancer Res. 2015 Jan; 35 (1): 523-30.
- Kogo R., How C., Chaudary N., Bruce J., Shi W., Hill R.P., Zahedi P., Yip K.W., Liu F.F. The microRNA-218~Survivin axis regulates migration, invasion, and lymph node metastasis in cervical cancer. Oncotarget. 2015 Jan 20; 6 (2): 1090-100.
- Campos-Viguri G.E., Jimenez-Wences H., Peralta-Zaragoza O., Torres-Altamirano G., Soto-Flores D.G., Hernandez-Sotelo D., Alarcon-Romero Ldel C., Jimenez-Lopez M.A., Illades-Aguiar B., Fernandez-Tilapa G.miR-23b as a potential tumor suppressor and its regulation by DNAmethylation in cervical cancer. Infect Agent Cancer. 2015; 10: 42 DOI: 10.1186/s13027-015-0037-6
- Honegger A., Schilling D., Bastian S., Sponagel J., Kuryshev V., Sultmann H., Scheffner M., Hoppe-Seyler K., Hoppe-Seyler F. Dependence of intracellular and exosomal microRNAs on viral E6/E7 oncogene expression in HPV-positive tumor cells. PLoS Pathog. 2015 Mar 11; 11 (3): e1004712 DOI: 10.1371/journal.ppat.1004712
- Zheng Z.M., Wang X. Regulation of cellular miRNA expression by human papillomaviruses. Biochim Biophys Acta. 2011 Nov-Dec; 1809 (11-12): 668-77 DOI: 10.1016/j.bbagrm.2011.05.005
- Ben W., Yang Y., Yuan J., Sun J., Huang M., Zhang D., Zheng J. Human papillomavirus 16 E6 modulates the expression of host microRNAs in cervical cancer. Taiwan J Obstet Gynecol. 2015 Aug; 54 (4): 364-70 DOI: 10.1016/j.tjog.2014.06.007
- Wang X., Wang H.K., Li Y., Hafner M., Banerjee N.S., Tang S., Briskin D., Meyers C., Chow L.T., Xie X., Tuschl T., Zheng Z.M. micro-RNAs are biomarkers of oncogenic human papillomavirus infections.Proc Natl Acad Sci USA. 2014 Mar 18; 111 (11): 4262-7 DOI: 10.1073/pnas.1401430111
- Wang L., Yu J., Xu J., Zheng C., Li X., Du J. The analysis of microRNA-34 family expression in human cancer studies comparing cancer tissues with corresponding pericarcinous tissues. Curr Pharm Des. 2014; 20 (11): 1639-46.
- Yuan F., Sun R., Chen P., Liang Y., Ni S., Quan Y., Huang J., Zhang L., Gao L. Combined analysis of pri-miR-34b/c rs4938723 and TP53 Arg72Pro with cervical cancer risk. Tumour Biol. 2016 May; 37 (5): 6267-73 DOI: 10.1007/s13277-015-4467-y
- Chen Y., Ma C., Zhang W., Chen Z., Ma L. Down regulation of miR-143 is related with tumor size, lymph node metastasis and HPV16 infection in cervical squamous cancer. Diagn Pathol. 2014 Apr 28; 9: 88 DOI: 10.1186/1746-1596-9-88
- Liu L., Wang Y.L., Wang J.F. Differential expression of miR-21, miR-126, miR-143, miR-373 in normal cervical tissue, cervical cancer tissue and Hela cell. Sichuan Da Xue Xue Bao Yi Xue Ban. 2012 Jul; 43 (4): 536-9.
- Yuan W., Xiaoyun H., Haifeng Q., Jing L., Weixu H., Ruofan D., Jinjin Y., Zongji S. MicroRNA-218 enhances the radiosensitivity of human cervical cancer via promoting radiation induced apoptosis. Int J Med Sci. 2014 May 6; 11 (7): 691-6 DOI: 10.7150/ijms.8880
- ZHu Y.K., Cheng N., Hu Y., Cen Y.Z. The role of microRNAs in the pathogenesis of cervical cancer and its relationship to HPV. Sheng Li Ke Xue Jin Zhan. 2012 Aug; 43 (4): 251-6.
- Botezatu A., Goia-Rusanu C.D., Iancu I.V., Huica I., Plesa A., Socolov D., Ungureanu C., Anton G. Quantitative analysis of the relationship between microRNA124a, -34b and -203 gene methylation and cervical oncogenesis. Mol Med Rep. 2011 Jan-Feb;4 (1): 121-8 DOI: 10.3892/mmr.2010.394
- Seifoleslami M., Khameneie M.K., Mashayekhi F., Sedaghati F., Ziari K., Mansouri K., Safari A. Identification of microRNAs (miR-203/miR-7) as potential markers for the early detection of lymph node metastases in patients with cervical cancer. Tumour Biol. 2015 Oct 22.
- Zhu X., Er K., Mao C., Yan Q., Xu H., Zhang Y., Zhu J., Cui F., Zhao W., Shi H. miR-203 suppresses tumor growth and angiogenesis by targeting VEGFA in cervical cancer. Cell Physiol Biochem. 2013; 32 (1): 64-73 DOI: 10.1159/000350125
- Lee J.W., Choi C.H., Choi J.J., Park Y.A., Kim S.J., Hwang S.Y., Kim W.Y., Kim T.J., Lee J.H., Kim B.G., Bae D.S. Altered MicroRNA expression in cervical carcinomas. Clin Cancer Res. 2008 May 1; 14 (9): 2535-42 DOI: 10.1158/1078-0432.CCR-07-1231
- You W., Wang Y., Zheng J. Plasma miR-127 and miR-218 Might Serve as Potential Biomarkers for Cervical Cancer. Reprod Sci. 2015Aug; 22 (8): 1037-41 DOI: 10.1177/1933719115570902
- Tian Q., Li Y., Wang F., Li Y., Xu J., Shen Y., Ye F., Wang X., Cheng X., Chen Y., Wan X., Lu W., Xie X. MicroRNA detection in cervical exfoliated cells as a triage for human papillomavirus-positive women. J Natl Cancer Inst. 2014 Sep 4; 106 (9). pii: dju241 DOI: 10.1093/jnci/dju241
- Han Y., Xu G.X., Lu H., Yu D.H., Ren Y., Wang L., Huang X.H., Hou W.J., Wei Z.H., Chen Y.P., Cao Y.G., Zhang R. Dysregulation of miRNA-21 and their potential as biomarkers for the diagnosis of cervical cancer. Int J Clin Exp Pathol. 2015 Jun 1; 8 (6): 7131-9.
- Shishodia G., Shukla S., Srivastava Y., Masaldan S., Mehta S., Bhambhani S., Sharma S., Mehrotra R., Das B.C., Bharti A.C. Alterations in microRNAs miR-21 and let-7a correlate with aberrant STAT3 signaling and downstream effects during cervical carcinogenesis. Mol Cancer. 2015 Jun 9; 14: 116 DOI: 10.1186/s12943-015-0385-2
- Leonard S.M., Wei W., Collins S.I., Pereira M., Diyaf A., Constandinou-Williams C., Young L.S., Roberts S., Woodman C.B. Oncogenichuman papillomavirus imposes an instructive pattern of DNA methylation changes which parallel the natural history of cervical HPV infection in young women. Carcinogenesis. 2012 Jul; 33 (7): 1286-93. doi: 10.1093/carcin/bgs157.
- Burgers W.A., Blanchon L., Pradhan S., de Launoit Y., Kouzarides T., Fuks F. Viral oncoproteins target the DNA methyltransferases. Oncogene. 2007 Mar 8; 26 (11): 1650-5.
- Jimenez-Wences H., Peralta-Zaragoza O., Fernandez-Tilapa G. Human papilloma virus, DNA methylation and microRNA expression in cervical cancer (Review). Oncol Rep. 2014 Jun; 31 (6): 2467-76 DOI: 10.3892/or.2014.3142
- Wilting S.M., van Boerdonk R.A., Henken F.E., Meijer C.J., Dios-dado B., Meijer G.A., le Sage C., Agami R., Snijders P.J., Steenbergen R.D. Methylation-mediated silencing and tumour suppressive function of hsa-miR-124 in cervical cancer. Mol Cancer. 2010 Jun 26; 9: 167 DOI: 10.1186/1476-4598-9-167
- Wang J.T., Ding L., Jiang S.W., Hao J., Zhao W.M., Zhou Q., Yang Z.K., Zhang L. Folate deficiency and aberrant expression of DNA methyltransferase 1 were associated with cervical cancerization. Curr Pharm Des. 2014; 20 (11): 1639-46.
- Hu Z., Zhu D., Wang W., Li W., Jia W., Zeng X., Ding W., Yu L., Wang X., Wang L., Shen H., Zhang C., Liu H., Liu X., Zhao Y., Fang X., Li S., Chen W., Tang T., Fu A., Wang Z., Chen G., Gao Q., Li S., Xi L., Wang C., Liao S., Ma X., Wu P., Li K., Wang S., Zhou J., Wang J., Xu X., Wang H., Ma D. Genome-wide profiling of HPV integration in cervical cancer identifies clustered genomic hot spots and a potential microhomologymediated integration mechanism. Nat Genet. 2015 Feb; 47 (2): 158-63 DOI: 10.1038/ng.3178
- Calin G.A., Sevignani C., Dumitru C.D., Hyslop T., Noch E., Yendamuri S., Shimizu M., Rattan S., Bullrich F., Negrini M., Croce C.M.Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc Natl Acad Sci USA. 2004 Mar 2; 101 (9): 2999-3004.
- Wentzensen N., Vinokurova S., von Knebel Doeberitz M. Systematic review of genomic integration sites of human papillomavirus genomes in epithelial dysplasia and invasive cancer of the female lower genital tract.Cancer Res. 2004 Jun 1; 64 (11): 3878-84.
- Zubillaga-Guerrero M.I., Alarcon-Romero L de lC., Illades-AguiarB., Flores-Alfaro E., Bermudez-Morales V.H., Deas J., Peralta-Zaragoza O. MicroRNA miR-16-1 regulates CCNE1 (cyclin E1) gene expression in human cervical cancer cells. Int J Clin Exp Med. 2015 Sep 15; 8 (9): 15999-6006.
- Gunasekharan V., Laimins L.A. Human papillomaviruses modulate microRNA145 expression to directly control genome amplification. J Virol. 2013 May; 87 (10): 6037-43 DOI: 10.1128/JVI.00153-13
- Tian Q., Li Y., Wang F., Li Y., Xu J., Shen Y., Ye F., Wang X., Cheng X., Chen Y., Wan X., Lu W., Xie X. MicroRNA detection in cervical exfoliated cells as a triage for human papillomavirus-positive women. J Natl Cancer Inst. 2014 Sep 4; 106 (9). pii: dju241 DOI: 10.1093/jnci/dju241
- Ribeiro J., Marinho-Dias J., Monteiro P., Loureiro J., Baldaque I., Medeiros R., Sousa H. miR-34a and miR-125b Expression in HPV Infection and Cervical Cancer Development Biomed Res Int. 2015; 2015: 304584 DOI: 10.1155/2015/304584
- He Y., Lin J., Ding Y., Liu G., Luo Y., Huang M., Xu C., Kim T.K., Etheridge A., Lin M., Kong D., Wang K. Asystematic study on dysregulated microRNAs in cervical cancer development. Int J Cancer. 2016 Mar 15; 138 (6): 1312-27 DOI: 10.1002/ijc.29618
- Mo W., Tong C., Zhang Y., Lu H. microRNAs’ differential regulations mediate the progress of Human Papillomavirus (HPV)-induced Cervical Intraepithelial Neoplasia (CIN). BMC Syst Biol. 2015 Feb 7; 9: 4 DOI: 10.1186/s12918-015-0145-3


