Ozone decomposition on the surface of metal oxide catalyst
Author: Batakliev Todor Todorov, Georgiev Vladimir Ferdinandov, Anachkov Metodi Parvanov, Rakovsky Slavcho Kirilov, Zaikov Gennadiy Efremovich
Journal: НБИ технологии @nbi-technologies
Section: Технико-технологические инновации
Article in issue: 6 (15), 2014.
Free access
The catalytic decomposition of ozone to molecular oxygen over catalytic mixture containing manganese, copper and nickel oxides was investigated in the present work. The catalytic activity was evaluated on the basis of the decomposition coefficient which is proportional to ozone decomposition rate, and it has been already used in other studies for catalytic activity estimation. The reaction was studied in the presence of thermally modified catalytic samples operating at different temperatures and ozone flow rates. The catalyst changes were followed by kinetic methods, surface measurements, temperature programmed reduction and IR-spectroscopy. The phase composition of the metal oxide catalyst was determined by X-ray diffraction. The catalyst mixture has shown high activity in ozone decomposition at wet and dry O3/O2 gas mixtures. The mechanism of catalytic ozone degradation was suggested.
Ozone, catalyst, decomposition, synthesis, kinetics, mechanism
Short address: https://sciup.org/14968368
IDR: 14968368 | UDC: 541.1 | DOI: 10.15688/jvolsu10.2014.6.4
Разложение озона на поверхности катализатора оксида металла
В настоящей работебыл исследован механизм каталитического разложения озона на молекулярный кислород из каталитической смеси, содержащей марганец, медь и никель оксиды. Реакция была изучена в присутствии термически модифицированных каталитических образцов, работающих при различных температурах и расходах озона. Фазовый состав металла оксидного катализатора определялся методом рентгеновской дифракции. Катализатор смеси показал высокую активность разложения озона на мокрой и сухой O3/O2 смеси газов. Был предложен механизм каталитической деградации озона.
Text of the scientific article Ozone decomposition on the surface of metal oxide catalyst
DOI:
Ozone finds wide application in such important industrial processes like: purification of drinking water, bleaching of textiles, oxidation of sulfurous gas, complete oxidation of exhaust gases from production of nitric acid and production of many organic compounds [21]. Ozone in the atmosphere protects the Earth’s surface against UV radiation, but on the ground level it is an air contaminant [7; 20; 21]. At this level ozone can be removed by adsorption, absorption, thermal and catalytic decomposition. The most effective catalysts for ozone decomposition are based on manganese oxide [10; 12; 18; 19]. The main method for purification of waste gases containing residual ozone is the heterogeneous catalytic decomposition. Noble metals like Pt, Ag, Pd and transition metal oxides including Co, Cu, and Ni supported on γ -Al2O3, SiO2 and TiO2 also are effective catalysts in this reaction [3; 4; 9; 11; 16; 24; 26], as it can be mentioned for activated carbon fibers [25].
The decomposition of ozone is a thermodynamically favored process with a heat of reaction of Δ H0298= –138 kJ/mol and free energy of reaction of Δ G0298= –163 kJ/mol [17]. The ozone structure is resonance stabilized that is a reason for its relative stability. The coefficient of ozone decomposition γ was used in other studies for investigation of NiO addition influence over cement-containing catalysts activity [15] and for study of thermal treatment influence over oxide catalyst activity [27].
The aim of present study is to apply mixed metal oxide catalyst for ozone decomposition, to investigate its behavior at different conditions and to determine its composition and surface properties using different physical methods for analysis.
Experimental
The basic copper, manganese, nickel carbonates and clay-bearing cement are milled in advance, then carefully mixed, crushed and compressed under pressure 4 t/cm2. The resulting tablets were treated hydrothermally at temperature of 80°C for 6 hours, dried at 120 °C for 6 hours and calcinated at 420 °C for 6 hours. The metal oxide catalyst based on the mixture of manganese oxide (20 wt %), copper oxide (10 wt %), nickel oxide (30 wt %) and claybearing cement (40 wt %) was thermally modified at 500 oC for 2 h and finally was applied in our investigation as catalyst for ozone decomposition in dry and water enriched gas flows. The catalyst was granulated and contained cylindrical grains with a diameter of about 5 mm and thickness of 3 mm.
The reactor for kinetic measurements was a glass tube (6 × 150 mm) filled in with 0.08-0.12 g of catalyst. Fig. 1 shows the schematic of the experimental set-up for all kinetics.
The kinetic measurements of ozone degradation were performed at flow rates ranging from 6.0 to 24 l h-1 and ozone concentration – from 1.0 to 1.2 mM. Ozone was generated by passing dry oxygen through a high-voltage silent-discharge ozone generator. At 15-20 kV was achieved ozone concentration about 1 mM. The inlet and outlet ozone concentrations were monitored using an UV absorption-type ozone analyzer at 300 nm.
The specific surface area of the catalyst (72 m2/g) was measured by N2 adsorptiondesorption isotherms at 77 K using BET method in a FlowSorb 2300 instrument (Micromeritics Instrument Corporation). IR studies were performed in the transmittance mode using a Nicolet 6700 FT-IR spectrometer (Thermo
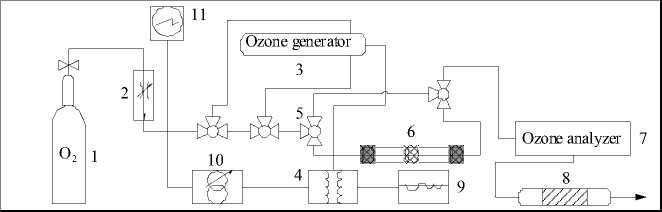
Fig. 1. Experimental set-up of reaction system for catalytic decomposition of ozone:
1 – oxygen; 2 – flow controller; 3 – ozone generator; 4 – transformer; 5 – three way turn cock;
6 –reactor charged with catalyst sample; 7 – ozone analyzer; 8 – reactor for decomposition of residual ozone;
9 – current stabilizer; 10 – autotransformer; 11 – voltmeter
Electron Corporation). A mixture of KBr and manganese oxide catalyst (100:1) was milled in an agate mortar manually before the preparation of pellets. The spectra were obtained by averaging 50 scans with 0.4 cm-1 resolution.
A typical TPR experiment is done by passing a H2 stream over a catalyst while it is heated linearly and monitoring the consumption of H2 with a thermal conductivity detector or mass spectrometer. In our study a 10 % H2/Ar mixture was used and the consumption of H2 was monitored using a thermal conductivity detector. A linear heating rate of 0.17 K s-1 was used for the experiment. X-ray diffraction (XRD) analysis was used to determine the crystalline metal oxide phases for the supported catalyst. A Bruker D8 Advance powder diffractometer with Cu K a radiation source and SolX detector was used. The samples were scanned from 2q angles of 10° to 80° at a rate of 0.04° s-1. The X-ray power operated with a current of 40 mA and a voltage of 45 kV.
Results and Discussion
The catalytic activity was evaluated on the basis of the coefficient g [14] that is proportional to ozone decomposition rate and to catalyst efficiency. It has been already used in other studies [15; 27]:
4ю , Y =--- In
V t S
[ О з ] o [ О з ]
where ю is the flow rate, Vt - specific heat rate of ozone molecules, S – geometrical surface of catalyst sample and [O3]0 and [O3] – inlet and outlet ozone concentrations, respectively.
In general, there is no precise estimation of Y by solving the diffusion-kinetic equation. This is possible in some special cases, for example to find y using the approximate method of Frank-Kamenetsky (method of equally accessible surface) [5]. Equally accessible surface is that surface where in each section the molecules fall with equal probability. The rate of the chemical reaction on the surface is expressed by concentration of reacting molecules in the volume near the surface. For reactions of first order:
ws = k s c s = kc '.
The parameters dimension is as follows: ws – (molecules/cm2. s ), кs and к – ( s -1) and (cm/ s ), cs и c - (molecules/cm2) и (molecules/cm3). It has been suggested that the molecular flow from volume to surface does not depend on the reaction rate, and with approximation it is defined of the equation:
j = p ( с - c ' )
where: в - coefficient of mass transfer, having dimension as the rate constant k, equal to cm s -1, c and c - concentration in the regions of the volume, where the flow is passing trough.
The distance between the surface and the region with concentration c ‘ the ozone molecules pass without collisions with average specific heat rate vT . The number of hits on unit of surface per unit of time z = vT c ‘ and taking into account the definition of coefficient of ozone decomposition it has been found that:
kc' 4 k
Y = = .
z vT
Thus, the coefficient у is related to the rate constant k . Now we could consider the case when the surface, where the reaction takes place, is located in an unlimited volume of gas. In stationary conditions the molecular flow toward the surface is equal to the chemical reaction rate:
ws
p kc k + p
= k eff c
where c – concentration of actives molecules standing to great distance from the catalytic surface.
Thus, the rate of reaction on the surface is expressed by the concentration in the volume and the effective rate constant that depends on the rate constant k and the coefficient of mass transfer p . Obviously:
1 11
----=-- + .
k eff k p
If p >> k, then c' = c and k eff= k : the total reaction rate is limited by the no hits stage with constant k . In this case the reaction proceeds in the kinetic region. If p << k , c' << c and k eff = p the reaction rate is determined by the rate of mass transfer and the reaction occurs in the diffusion region.
On the other hand [14], when operating in stationary conditions the relationship between the constant rate of the reaction of heterogeneous ozone decomposition and the coefficient of ozone decomposition у is given by the formula:
where: V – volume of reactor, S – geometric surface of catalyst, vT – specific heat rate of ozone molecules.
Taking into account the dependence of the gas flow rate from the reactor radius, and considering the concentration of active ozone molecules and the reaction time of ozone decomposition, after complex mathematical transformations the latter formula passes into the expression:
[ O 3 ] o [ O 3 ]
4ю , у =--- ln
SvT where: ю - gas flow rate, [O3]0 и [O3] - inlet and outlet ozone concentrations.
This formula is applicable for tubular type reactor, when the catalyst is supported as thin layer on the walls in the inner side of the tube. The expression is also convenient for calculation of catalytic activity in the case when the reactor is filled with granulated catalytic samples having specific geometric surface. Therefore, this method has been used by us for calculation of the catalysts activity in the process of ozone decomposition.
Results and Discussion
Fig. 2 shows changing catalytic activity of the cement containing catalyst when the calcination temperature of the samples is different. All the experiments were made in dry conditions and ozone flow rate of 6 l h-1. The MnOx/CuO/NiO catalyst has catalytic activity in ozone decomposition that y = 4kheV
SvT
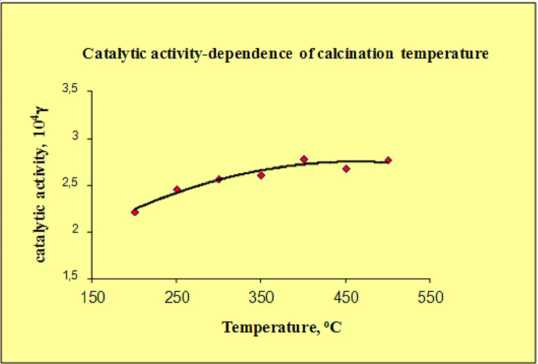
Fig. 2. Dependence of the catalytic activity in ozone decomposition on calcination temperature
does not change dramatically with calcination temperatures increasing. However, we can see that the catalyst is more active when the calcination temperature is in the range of 400-500 °C. On Fig. 3 the changes of γ at two different flow rates in temperature range of 258-323 K are shown. The calculated activation energy is 5 kJ/mol. In these experiments the reactor was kept up at constant temperature enough time to get the necessary value. The duration of the decomposition reaction and the reaction time were much smaller than the time of cooling or heating the reactor. That means that the temperature inside the reactor was maintained constant during the measurement of ozone decomposition rate. The difference between the values of γ at different flow rates is due to the low loading of catalyst. The low values of Ea are directly connected with the fact that limited stage of reaction is the adsorption of ozone on catalytic surface.
On Fig. 4 are presented the changes of γ depending on temperature and humidity of the gas flow. The measured activation energy was also 5 kJ/mol. It was found out that the humidity of the gas flow decreases the catalytic activity by 10 %, but the catalyst does not disturb its stability in the time of ozonation.
Fig. 5 presents the changes of γ at different flow rates at two temperatures -258 and 298 K. It can be seen that γ is proportional to the flow rate at both temperatures. The coefficient of
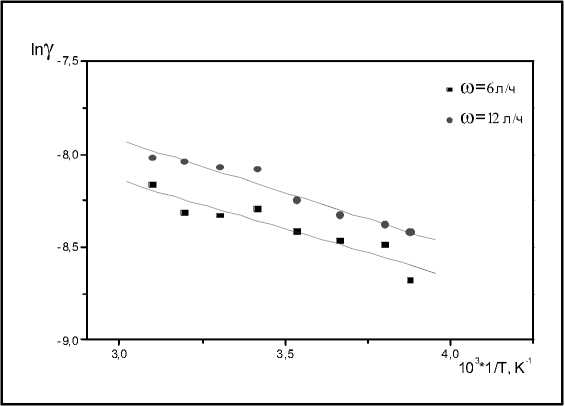
Fig. 3. Dependence of the catalytic activity in ozone decomposition on reaction temperature at two different flow rates
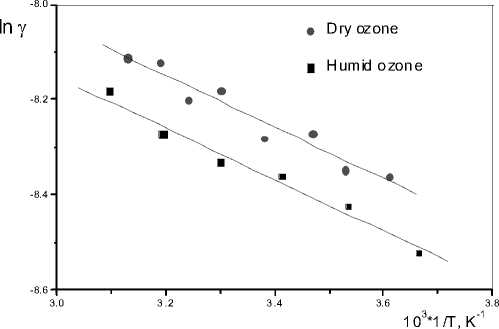
Fig. 4. Temperature dependence of catalytic activity at dry and humid conditions, temperature range 273-323 K, ozone flow rate 8 l h-1
decomposition depends on temperature, but at 258 K the steady state of the curve is reached faster. The obtained values of γ are close to the coefficients reported in literature [15]. Therefore, it could be concluded that at low values of gas flow rate, a reason for the dependence of the catalytic activity from gas flow rate is the influence of the external diffusion over the kinetics of the process of heterogeneous catalytic ozone decomposition on catalyst surface, i.e. the process takes place mainly in the outer diffusion region or in the transition diffusion-kinetic region.
Fig. 6 shows the dependence of γ on gas flow humidity at 298 K. The values of γ decrease with increasing the humidity, but nevertheless these values remain relatively high. The effect of water vapor may be result of thin film formation on the catalytic surface that makes the diffusion of ozone to catalytic centers more difficult. The humidity of O3/O2 gas flow was measured to be 50 %.
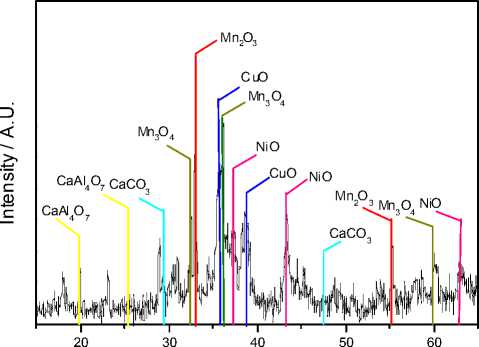
A popular technique used to characterize manganese oxide containing catalyst and to determine the identity of the manganese oxide phase at high loadings (> 6 %) has been X-ray diffraction (XRD) [2; 13]. The X-ray diffraction results for the cement-containing metal oxide catalyst are presented on Fig. 7. The diffractogram for the catalyst sample showed peaks with a certain number of large intensities at different 2 θ values. The peaks at 39° and 35.5° correspond to copper oxide (CuO). The diffraction features for the catalyst at 33° and 55.1° are indicative of bixbyite-o
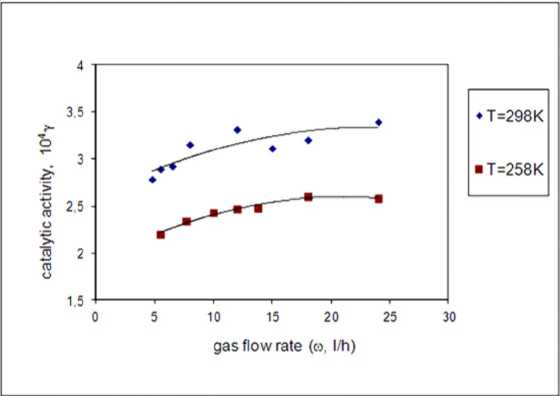
Fig. 5. Flow rate dependence of catalytic activity at 258 and 298 K, ozone flow rate range 5-25 l h-1
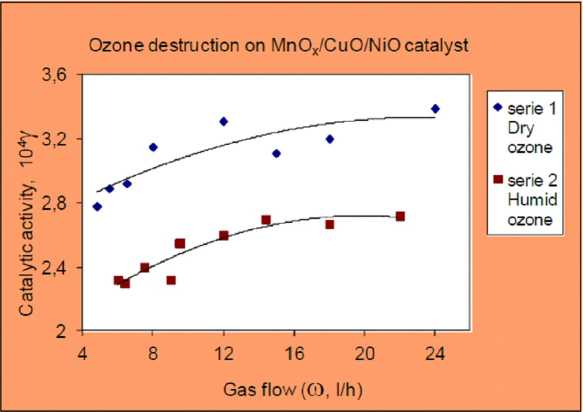
Fig. 6. Flow rate dependence of catalytic activity at dry and humid conditions, ozone flow rate range 4-26 l h-1
(Mn2O3). The metal oxide catalyst sample peaks at 43°, 37.2° and 62.9° are due to nickel oxide (NiO). The catalyst diffraction peaks at 36.1°, 32.4° and 59.9° correspond to hausmannite (Mn3O4). Finally, the cement diffraction peaks at 20°, 25.5° and 29.5°, 47.5° are due respectively to grossite (CaAl4O7) and calcite (CaCO3). In conclusion, the information which can be deduced from the X-ray diffractogram for this catalyst is that there are three metal oxides as the manganese oxide is present in two forms - Mn2O3 and Mn3O4.
It could also be seen that the cement support of the catalyst is build mainly by two components -CaAl4O7 and CaCO3.
The TPR experiment was carried out for the supported on cement metal oxide catalyst (Fig. 8). The H2 consumption was monitored by thermal conductivity detector in the course of time. Manganese-containing catalyst was already studied using TPR [8]. The peak temperatures of reduction on Fig. 8 are 527 K, 596 K, 643 K and 976 K or the reduction
29 / Degrees
Fig. 7. XRD of MnOx/CuO/NiO catalyst supported on cement
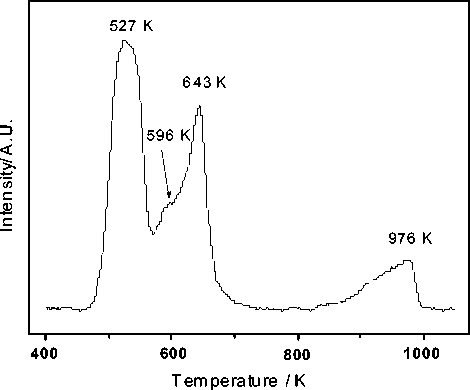
Fig. 8. TPR of MnOx/CuO/NiO catalyst supported on cement
temperature of the catalyst was in the range 527976 K. The bulk reduction peaks at 596 K and 643 K can be identified for the manganese oxide in the cement-containing metal oxide mixture while the peak at 976 K can be related with reduction of the nickel oxide [3; 8].
The most possible mechanism [10; 12] of catalytic ozone decomposition can be presented as follows:
O 3 + * ^ O 2 + O * , (1)
O 3 + O ^ O2+ O2 , (2)
O2 ^ O2 + * , (3)
where the symbol * was used to denote surface sites. In step (1) ozone decays and the finding that the adsorbed ozone does not desorbs ascertains the irreversibility of steps (1) and (2). Further peroxide particles are formed in accordance with step (2) and then oxygen is desorbed from the catalytic surface – step (3). The finding that the peroxide species could not be formed from molecular oxygen at any conditions shows the irreversibility of step (3).
The FT-IR spectra of the catalyst before and after ozone decomposition are shown in Fig. 9. The two similar spectra indicate that the catalyst does not change practically during the reaction. A broad band at 3415-3425 cm-1 and also the band at 1410-1430 cm-1 are associated with the vibrations of water molecules [6; 25]. The intensive bands at 5 15-530 cm-1 in accordance with literature [1], were assigned to the stretching vibration of the surface metal-oxygen bond.
The FT-IR spectra of cement-oxide catalyst after dry ozone decomposition ( a ) and after humid ozone decomposition ( b ) are presented in Fig. 10. The spectra are almost identical, showing that the catalyst structure is not altered during the humid catalytic reaction. The broad adsorption band at 3430 cm-1 appears from the stretching vibration of hydrogen bonded hydroxyl groups [25]. The adsorption band at 1635 cm-1 is due to vibrations of water molecules [24]. The intensive band at 520530 cm-1 appears at higher manganese concentrations and, in accordance with literature, can be attributed to well-defined Mn2O3 phase [2].
Conclusions
-
1. The catalyst based on MnOx/CuO/NiO has high efficiency in the reaction of ozone decomposition both at dry and wet conditions.
-
2. The catalyst work is stable and its activity does not change dramatically by varying temperature, O3/O2 flow rate and humidity.
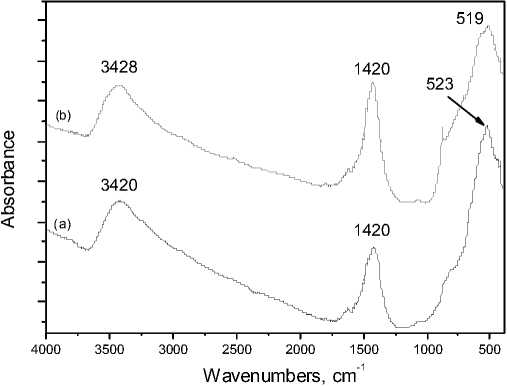
Fig. 9. FT-IR spectra of MnOx/CuO/NiO catalytic samples obtained before ozone decomposition ( a ) and after ozone decomposition for 8 h ( b )
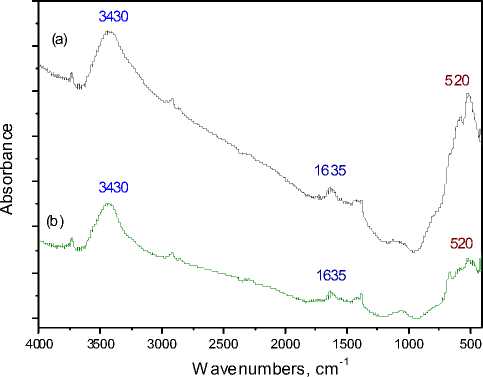
Fig. 10. FT-IR spectra of cement-oxide catalyst after dry ozone decomposition ( a ) and after humid ozone decomposition ( b )
-
3. XRD analysis has proven to be a useful tool for the identification of all metal oxide phases in catalyst mixture. The temperature programmed reduction of the catalyst denotes its high reducibility.
-
4. The FT-IR spectral band at 526 cm-1 shows that the catalyst stays unchangeable during the ozone decomposition for about 8 h.
-
5. The FT-IR spectra after humid ozone decomposition indicates that even decomposing ozone in humid conditions the catalyst structure does not change practically during the reaction.
References Ozone decomposition on the surface of metal oxide catalyst
- Bielanski A., Haber J. Oxygen in Catalysis. Marcel Dekker Inc., New York, 1991.
- Buciuman F., Patcas F., Craciun R., Zhan D.R.T. Vibrational Spectroscopy of Bulk and Supported Manganese Oxides. Phys. Chem., 1998, no. 1, p. 185.
- Dhandapani B., Oyama S.T. Gas Phase Ozone Decomposition Catalysts. J. Appl. Catal. B: Environmental, 1997, no. 11, p. 129.
- Einaga H., Futamura S. Comparative Study on the Catalytic Activities of Alumina-Supported Metal Oxides for Oxidation of Benzene and Cyclohexane With Ozone. React. Kinet. Catal. Lett., 2004, no. 81, p. 121.
- Frank-Кamenetskii D.А. Diffusion and Heat Transfer in Chemical Kinetics. Moscow, 1987. (in Russian).
- Gomez-Serrano V., Alvarez P. M., Jaramillo J., Beltran F.J. Formation of Oxygen Structures by Ozonation of Carbonaceous Materials Prepared from Cherry Stones-II, Kinetic Study. Carbon, 2002, no. 40, p. 513.
- Heisig C., Zhang W., Oyama S.T. Decomposition of Ozone Using Carbon Supported Metal Oxide Catalysts. Appl. Catal. B: Environ., 1997, no. 14, p. 117.
- Kapteijn F., Van Langeveld A.D., Moulijn J.A., Andreini A., Vuurman M.A., Turek M.A., Jehng J.M., Wachs I.E. Alumina-Supported Manganese Oxide Catalysts. J. Catal., 1994, vol. 150, p. 94.
- Konova P., Stoyanova M., Naydenov A., Christoskova St., Mehandjiev D. Catalytic oxidation of VOCs and CO by ozone over alumina supported cobalt oxide. J. Appl. Catal. A: Gen., 2006, vol. 298, p. 109.
- Li W., Gibbs G.V., Oyama S.T. Mechanism of Ozone Decomposition on Manganese Oxide: 1. In Situ Laser Raman Spectroscopy and ab Initio Molecular Orbital Calculations. J. Am. Chem. Soc., 1998, vol. 120, p. 9041.
- Li W., Oyama S. T. Absolute Determination of Reaction Mechanisms by in Situ Measurements of Reaction Intermediates. Top. Catal., 1999, vol. 8, p. 75.
- Li W., Oyama S.T. The Mechanism of Ozone Decomposition on Manganese Oxide: 2. Steady-state and Transient Kinetic Studies. J. Am. Chem. Soc., 1998, vol. 120, p. 9047.
- Lo Jacono M., Schiavello M. The Influence of Preparation Methods on Structural and Catalytic Properties of Transition Metal Ions Supported on Alumina. Delmon B., Jacobs P., Poncelet G., eds. Preparation of Catalysts. New York, 1976, p. 473.
- Lunin V.V., Popovich M.P., Tkachenko S.N. Physical Chemistry of Ozone. Moscow, Moscow University Publ. House, 1998, pp. 377-444. (in Russian).
- Martinov I.V., Tkachenko S.N., Demidyuk V.I., Egorova G.V., Lunin V.V. Addition Influence Over Cement-Containing Catalysts Activity in Ozone Decomposition. J. of Moscow Univ., Ser. 2, Chemistry, 1999, vol. 40, p. 355. (in Russian).
- Oyama S.T. Chemical and Catalytic Properties of Ozone. Catal. Rev. Sci. Eng., 2000, vol. 42, p. 279.
- Perry R.H., Green D. Perry's Chemical Engineer's Handbook. New York, McGraw-Hill, 1989, pp. 3-147.
- Radhakrishnan R., Oyama S.T., Chen J., Asakura A. Electron Transfer Effects in Ozone Decomposition on Supported Manganese Oxide..J. Phys. Chem. B, 2001, vol. 105 (19), p. 4245.
- Radhakrishnan R., Oyama S.T., Ohminami Y., Asakura K. Structure of MnOx/Al2O3 Catalyst: A Study Using EXAFS, In Situ Laser Raman Spectroscopy and Ab Initio Calculations. J. Phys. Chem., 2001, vol. 105, p. 9067.
- Rakitskaya T.L., Bandurko A.Yu., Ennan A.A., Paina V.Ya., Rakitskiy A.S. Carbon-Fibrous-Material-Supported Base Catalysts of Ozone Decomposition. Micro. Meso. Mater., 2001, vol. 43, p. 153.
- Rakovsky S., Zaikov G. Kinetics and Mechanism of Ozone Reactions with Organic and Polymeric Compounds in Liquid Phase. 2nd ed. New York, Nova Science Publishers Inc., 2007, pp. 1-340.
- Semenova L.M., Bakhracheva Yu.S., Semenov S.V. Laws of Formation of Diffusion Layers and Solution of the Diffusion Problem in Temperature-Cycle Carbonitriding of Steel. Metal Science and Heat Treatment, 2013, vol. 55, no. 1-2, pp. 34-37.
- Shapochkin V.I., Semenova L.M., Bakhracheva Yu.S., Gyulikhandanov E.L., Semenov S.V. Effect of Nitrogen Content on the Structure and Properties of Nitrocarburized Steel. Metal Science and Heat Treatment, 2011, vol. 52, no. 9-10, pp. 413-419.
- Stoyanova M., Konova P., Nikolov P., Naydenov A., Christoskova S., Mehandjiev D. Alumina-Supported Nickel Oxide for Ozone Decomposition and Catalytic Ozonation of CO and VOCs. Chem. Eng. Journal, 2006, vol. 122, p. 41.
- Subrahmanyam C., Bulushev D., Kiwi-Minsker L. Dynamic Behaviour of Activated Carbon Catalysts During Ozone Decomposition at Room Temperature. J. Appl. Catal. B: Environmental, 2005, vol. 61, p. 98.
- Tong S., Liu W., Leng W., Zhang Q. Characteristics of MnO2 Catalytic Ozonation of Sulfosalicylic Acid Propionic Acid in Water. Chemosphere, 2003, vol. 50, p. 1359.
- Zavadskii A.V., Kireev S.G., Muhin V.M., Tkachenko S.N., Chebkin V.V., Klushin V.N., Teplyakov D.E. Thermal Treatment Influence Over Hopcalite Activity in Ozone Decomposition. J. of Phys. Chem., 2002, vol. 76, p. 2278. (in Russian).


