Physiochemical characterization and molecular phylogeny of drought responsive proteins in Finger Millet (Eleusine coracana L.)
Author: Hosita Gupta, Sugandha Tiwari
Journal: Журнал стресс-физиологии и биохимии @jspb
Article in issue: 1 т.22, 2026.
Free access
Background. Millets are considered climate-resilient crops because they require minimal inputs, grow in poor soils, and tolerate abiotic stresses such as drought and high temperature. As global water availability becomes more unpredictable, drought is emerging as a major factor reducing agricultural productivity. Understanding how millet plants respond biochemically to drought conditions provides valuable insight for developing stress-resilient varieties. Drought affects plants at multiple levels physiological, biochemical, and molecular. Drought stress is a major abiotic factor limiting crop productivity worldwide. Finger millet (Eleusine coracana) is renowned for drought and salinity resilience. This study involves in -silico physiochemical characterization of drought-responsive proteins and their molecular phylogeny to understand evolutionary relationships with other cereal species. Results. Proteins associated with drought stress exhibited diverse molecular characteristics. Genotypes possessing proteins with: lower instability index (<40), higher aliphatic index, and negative GRAVY values were predicted to exhibit greater structural stability and hydrophilicity traits commonly associated with stress resilience. Some of the highly expressed proteins involved in stress tolerance in Eleusine coracana retrieved from UNIPROT (UniProt Consortium ,2025) in FASTA format are -ACL97372.1 truncated calmodulin, partial [Eleusine coracana], -ADB43602.1 prolamin, partial [Eleusine coracana], -ADC44447.1 monodehydroascorbate reductase, partial [Eleusine coracana],-AEF58885.1 NADH dehydrogenase subunit F, partial (chloroplast) [Eleusine coracana],-AEH04409.1 hydroxymethylglutaryl-CoA synthase, partial [Eleusine coracana],etc. Their physicochemical properties like stability, charge, hydrophobicity, and solubility help to determine their function adaptive for survival under stress. These proteins represent key components of stress signaling, antioxidant defense, mitochondrial energy metabolism, and nutrient transport, which collectively contribute to drought resilience. These proteins subcellular organization and gene ontology justifies their role in stress tolerance. Molecular phylogeny reveals both conserved and uniquely expanded gene families, reflecting evolutionary adaptation. Conclusion. This in silico investigation identifies and characterizes a suite of highly expressed climate-resilience proteins in E. coracana. Molecular phylogeny reveals both conserved and uniquely expanded gene families, reflecting evolutionary adaptation. These results lay the groundwork for targeted functional genomics to harness stress tolerance for finger millet improvement and translational breeding. Our findings are vital for breeding and selecting drought tolerant varieties of finger millet. Further, genomic and molecular investigations need to be undertaken to gain a deeper insight into the detailed mechanisms of drought tolerance in finger millet.
Drought stress, In silico analysis, Physiochemical characterization, instability index, molecular phylogeny, gene ontology, Protein modeling
Short address: https://sciup.org/143185425
IDR: 143185425
Text of the scientific article Physiochemical characterization and molecular phylogeny of drought responsive proteins in Finger Millet (Eleusine coracana L.)
A total of 729 drought-responsive genes previously reported in six crop species Oryza sativa, Triticum aestivum, Zea mays, Hordeum vulgare, Sorghum bicolor, Pennisetum glaucum and the model plant Arabidopsis thaliana were compiled and analyzed. Among these, 20 genes exhibited strong enrichment for drought-associated Gene Ontology (GO) terms and were significantly overrepresented in multiple biological process and molecular function categories. Functional annotation indicated that these genes participate in diverse stress-adaptive pathways, including water and solute transport, tolerance to salinity, cold, and drought, abscisic acid (ABA)-mediated signaling, de novo NA methylation, and transcriptional regulation in tef and other crops. Comparative domain analysis revealed substantial divergence in conserved domains between several tef genes and their well-characterized rice orthologs. Furthermore, the expression patterns of sixteen tef orthologs were examined using quantitative RT-PCR under polyethylene glycol (PEG)-induced osmotic stress. (Bekele et al., 2023) Pearl millet (Pennisetum glaucum L.) is a key cereal crop cultivated in arid and semi-arid regions, where it plays a critical role in ensuring food and fodder security. Its inherent tolerance to drought stress provides an opportunity to elucidate underlying cellular and molecular mechanisms using functional genomics approaches aimed at improving grain yield. In the present study, drought responses were investigated in 48 inbred pearl millet lines representing four distinct maturity groups at the flowering stage. A total of 74 drought-responsive genes were identified and classified into five major phylogenetic clades and eight functional categories, including abscisic acid (ABA) signaling, hormone signaling, ion and osmotic homeostasis, transcription factor (TF)-mediated regulation, molecular adaptation, signal transduction, physiological adaptation, and detoxification. Conserved motif analysis revealed that protein kinases and MYB domain–containing proteins were among the most highly conserved. Comparative in silico analysis across millet species indicated that foxtail millet shared the highest number of orthologs with pearl millet. Haplotype analysis across millet crops identified 698 haplotypes, with MyC2 and Myb4 exhibiting the greatest haplotype diversity. Protein–protein interaction network analysis highlighted ABI2, P5CS, C PK, REB, MYB, and CYP707A3 as key hub genes. Expression profiling demonstrated both shared and maturity group– specific drought-responsive genes, enabling the identification of drought-tolerant genotypes within each maturity group. Among the gene families analyzed, ABA signaling components, transcription factors, and signaling proteins emerged as major contributors to drought tolerance across maturity groups. These functionally characterized genes represent promising candidates for deployment in backcross breeding, genomic selection, and gene-editing strategies to enhance drought resilience in pearl millet and related millet crops grown in water-limited environments (Chakraborty et al., 2022).In a related in silico study, the 1.5 kb upstream promoter regions of 40 rice germin-like protein (OsGLP) genes were analyzed to identify cis-regulatory elements corresponding to NAC, WRKY, bHLH, bZIP, MYB, and AP2/ERF transcription factor binding sites (TFbs). Among the analyzed promoters, OsGLP8-11 contained the highest number of predicted TFbs, whereas OsGLP5-1 exhibited the lowest. Phylogenetic analysis of OsGLP promoter sequences grouped them into four distinct clades, reflecting evolutionary diversification. These findings highlight the evolutionary significance of promoter variation among OsGLP genes. Consistent with previous reports, gene duplication events followed by diversification of cis-regulatory elements likely contributed to neofunctionalization of OsGLP genes and their promoters, enabling adaptive responses to both biotic and abiotic stresses in rice. ( as et al., 2019) Hydrogen peroxide (H₂O₂) is a stable reactive oxygen species that functions as a signaling molecule at low concentrations but causes oxidative stress and cell death when accumulated excessively.
Catalases (CATs) are key enzymes involved in H₂O₂ detoxification and plant stress tolerance. In this study, seven CAT genes in Triticum dicoccoides , four in Triticum urartu , and eight in Triticum spelta were observed. Gene structure, phylogenetic, protein domain, and subcellular localization analyses confirmed the reliability of the identified CAT members, all of which contained conserved catalase (pfam00199) and catalase-related (pfam06628) domains. Phylogenetic analysis indicated conservation of CAT proteins across ancient and modern wheat species, with most proteins predicted to localize to peroxisomes. Promoter analysis revealed diverse cis-regulatory elements associated with development and stress responses. RT-qPCR analysis showed tissue-specific and stress-responsive expression patterns under salt, heat, cold, mannitol, and ABA treatments. Overall, this comprehensive analysis provides insights into CAT gene evolution and function in Triticeae and establishes a foundation for future functional studies. (Ghorbel et al. , 2023) SO s are defense associated proteins that detoxify ROS and primarily serve as scavengers. Genome wide study of SO gene family revealed the conserved character of BrSO s in Brassica rapa that can be potentially utilised in breeding program for resilience to climate change and abiotic stresses tolerance. (Iqbal et al. , 2021)
Phosphorus (P) availability and water supply are fundamental determinants of plant growth and productivity, strongly affecting global agricultural output. Phosphate transporter (PHT) proteins are central to phosphate (Pi) uptake, internal transport, and maintenance of Pi homeostasis under varying environmental conditions. A genome-wide survey of the PHT gene family in Avena sativa revealed that the functional diversity of the AsPHT gene family and enhance the understanding of the molecular basis of phosphate and water utilization in oats, providing a foundation for breeding strategies aimed at improving phosphorus use efficiency and drought resilience. (Li et al., 2025) Glucose-6-phosphate dehydrogenase (G6P H) regulates redox homeostasis and drought tolerance in soybean under polyethylene glycol 6000 (PEG6000)-induced drought stress showed higher activities of SO , CAT, PO , GR, HAR, and M HAR, and increased glutathione (GSH) and ascorbate (Asc) levels. (Liu et al., 2013) Phosphorus (P), primarily absorbed as inorganic phosphate (Pi), is a critical limiting nutrient for plant growth. 12 PHosphate Transporter 1 (PHT1) family genes (SiPHT1;1–1;12) in 20 foxtail millet (Setaria italica genes revealed their role in stress tolerance and provide potential targets for improving phosphorus-use efficiency in millet and other cereals. (Roch et al., 2020) Members of the phosphate transporter 1 (PHT1) gene family play crucial roles in plant growth regulation and stress responses. In this study, eight PHT1 family genes were identified from the potato genome using a combination of molecular and bioinformatic approaches. Expression analysis revealed that StPHT1;7 was highly expressed across the whole potato plant. (Cao et al., 2020) Peanut seed prolamins are traditionally recognized as nitrogen storage proteins and the study indicated their potential role in defense against fungal pathogens. (Senakoon et al. 2015) Finger millet (Eleusine coracana L.), a resilient C4 cereal crop, is vital in semi-arid regions due to its adaptability to poor soils and minimal water requirements. However, with increasing climate variability, understanding drought tolerance mechanisms in finger millet is essential for sustainable agriculture.
MATERIALS AND METHOD
Database search and sequence retrieval:
The corresponding stress responsive proteins were retrieved from the publicly available database UNIPROT. Retrieved the E. coracana stress responsive proteins from UNIPROT (UniProt Consortium, 2025) in FASTA format. Selected some candidate proteins like Ec calmodulin, Ec prolamin, Ec monodehydroascorbate reductase, Ec ascorbate reductase and Ec phosphate transporter. Protein sequences retrieved from public databases were analyzed using ProtParam, and SWISS-MO EL to determine molecular weight, isoelectric point, instability index, aliphatic index, hydropathicity, and structural stability.
The physiochemical Characterization:
The physiochemical characterization of stress responsive proteins that is determination of number of amino acids, molecular weight, iso-electric point (pI), aliphatic index (AI), instability index (II), the grand average of hydropathy (GRAVY) with the help of ProtParam available in ExPASy The ExPASy ProtParam tool was used to estimate isoelectric point (pI), protein length (aa), and molecular weight (k a) .
Subcellular distribution:
The subcellular localisation of the corresponding stress responsive proteins and their gene ontology were analysed using CELLO, UNIPROT, WOLFPSORT.
Structure modeling:
Retrieved stress responsive protein sequences were uploaded for protein homologous modelling using Swiss model in ExPASy and corresponding Ramachandran Plot was constructed. The models showing maximum sequence identity were selected and corresponding Ramachandran plot were assessed.
Phylogenetic Analysis
Blasted each candidate vs homologous proteins in related millets likewise Eleusine coracana, Setaria italica, Sorghum bicolor, Paspalum scrobiculatum, Cenchrus americanus, grasses like Zea mays, Oryza sativa and Arabidopsis thalliana via NCBI protein BLAST using non-redundant protein sequences with parameters: threshold 0.05, maximum target sequence 100, matrix BLOSUM 62. BLAST (Basic Local Alignment Search Tool) helped to align & identify homologous sequences to query sequences. Ran the multiple sequence alignment (MSA) algorithm and saved the resulting file. MSA identified conserved regions, variation and phylogenetic relationships. A high percent identity with low E-value suggested a strong evolutionary relationship. COBALT (CONSTRAINT BASE MULTIPLE ALIGNMENT TOOL) was used for constructing phylogenetic trees. For analyzing a phylogenetic tree using BLAST results, homologous sequences were aligned using the Neighbor Joining evolutionary model with 1000 bootstraps. Conserved domains were observed using NCBI C SEARCH.
RESULTS
Proteins associated with drought stress exhibited diverse molecular characteristics. Genotypes possessing proteins with: lower instability index (<40), higher aliphatic index, and negative GRAVY values were predicted to exhibit greater structural stability and hydrophilicity traits commonly associated with stress resilience.
Database search and sequence retrieval:
Some of the highly expressed proteins involved in stress tolerance in Eleusine coracana retrieved from UNIPROT (Uniport Consortium, 2025) in FASTA format are >Q Z71191.1 catalase A1 ( Eleusine coracana ], >ACC59765.1 of-type zinc finger protein, partial [ Eleusine coracana ], >ACC77653.1 NAC transcription factor [ Eleusine coracana ], >ACF06728.1 cold shock protein, partial [ Eleusine coracana ], >ACF06727.1 ascorbate peroxidase 1, partial [ Eleusine coracana ], >ACL97372.1 truncated calmodulin, partial [ Eleusine coracana ], >A B43602.1 prolamin, partial [ Eleusine coracana ], >A C44447.1 monodehydroascorbate reductase, partial [ Eleusine coracana ],>AEF58885.1 NA H dehydrogenase subunit F, partial (chloroplast) [ Eleusine coracana ],>AEH04409.1
hydroxymethylglutaryl-CoA synthase, partial [ Eleusine coracana ],etc.
Table 1: summarizes the identified drought-responsive proteins, including their predicted subcellular localization, gene ontology annotations, amino acid length, molecular weight, isoelectric point (pI), instability index (II), aliphatic index (AI), and GRAVY values. These parameters highlight the functional diversity and structural properties of proteins involved in calcium-mediated signalling, post-transcriptional regulation, mitochondrial energy metabolism, antioxidant defense, and phosphate transport, collectively contributing to plant drought resilience.
THE PHYSIOCHEMICAL CHARACTERIZATION:
Identification of Proteins Associated with Drought Stress Response
Five drought-associated proteins were identified and functionally annotated based on subcellular localization, gene ontology, and physicochemical properties (Table 1). These proteins represent key components of stress signaling, antioxidant defense, mitochondrial energy metabolism, and nutrient transport, which collectively contribute to drought resilience. Calmodulin was identified as a cytoplasmic calcium-binding protein with enzyme regulator activity. Its hydrophilic nature (GRAVY -0.740) and acidic pI (4.39) support dynamic protein– protein interactions required for calcium-mediated signal transduction. The presence of calmodulin indicates activation of early drought stress signaling pathways involved in downstream transcriptional and physiological responses. Prolamin, localized in the cytoplasm and associated with the spliceosomal complex, was linked to positive regulation of water stress responses. Its low instability index (32.31) and strong hydrophilicity (GRAVY -1.008) suggest structural stability under dehydration, supporting a role in post-transcriptional regulation and cellular protection during drought stress. A mitochondrial inner membrane NA H dehydrogenase exhibiting oxidoreductase activity was detected, indicating maintenance of respiratory electron transport under drought conditions. The high aliphatic index (101.29) suggests enhanced stability, supporting sustained ATP production and mitigation of stress-induced metabolic disruption. Ascorbate peroxidase, a cytoplasmic protein involved in the cellular response to oxidative stress, was identified. Its physicochemical properties indicate solubility and functional efficiency in detoxifying hydrogen peroxide, highlighting activation of antioxidant defenses to counter drought-induced reactive oxygen species accumulation. A multi-pass phosphate transporter was identified, characterised by a positive GRAVY value (0.430) consistent with membrane localisation. Its involvement in phosphate ion transport suggests an adaptive mechanism to maintain phosphate availability and energy metabolism during water-limited conditions. Together, these proteins define a coordinated drought response involving calcium signalling, RNA-level regulation, mitochondrial energy maintenance, antioxidant protection, and nutrient transport, underscoring their collective contribution to drought resilience.
SUBCELLULAR DISTRIBUTION:
Calmodulin, prolamin, and ascorbate peroxidase are localised in the cytoplasm and are involved in stress signalling, post-transcriptional regulation, and antioxidant defence, respectively. NA H dehydrogenase is localised to the mitochondrial inner membrane, supporting energy metabolism under drought conditions. A multi-pass phosphate transporter is localised to cellular membranes, contributing to phosphate homeostasis during water deficit. (Table 1).
STRUCTURE MODELING:
Calmodulin contributes to drought tolerance by sensing drought-induced calcium signals in plant cells. Upon calcium binding, it changes shape and activates stress-response proteins that regulate stomatal closure, gene expression, and antioxidant defences, helping plants conserve water and survive dehydration stress. The Ramachandran plot shows that calmodulin is predominantly α-helical and structurally stable. It showed 97.40% favoured region. This well-ordered yet flexible structure allows calmodulin to undergo calcium-induced conformational changes, enabling efficient sensing of drought-induced Ca² ⁺ signals and activation of stress-response pathways that promote drought tolerance in plants (Figure 1).
The structure of prolamin favours stress resistance and storage stability, enabling plants to withstand drought by protecting nutrient reserves rather than directly signalling stress responses. Ramachandran plot analysis revealed that most prolamin residues occupy favoured α-helical and β-sheet regions (85.48%), indicating a stable backbone conformation. The prolinerich, hydrophobic structure supports conformational stability during dehydration, contributing to protein resilience and enhanced drought tolerance in plants (Figure 2).
The structure of M HAR reveals a conserved flavoprotein fold with strategically arranged FA - and NA (P)H-binding domains that enable efficient electron transfer and stabilisation of monodehydroascorbate during antioxidant recycling. The predominance of M HAR residues in favoured regions of the
Ramachandran plot indicates a high-quality and stable protein structure. Structural stability is particularly important under drought stress, where dehydration and oxidative conditions can compromise protein conformation and enzymatic activity. A well-folded M HAR ensures efficient regeneration of ascorbate, thereby sustaining the ascorbate–glutathione cycle and reducing ROS accumulation. This contributes to the protection of cellular components such as membranes, proteins, and the photosynthetic apparatus. The presence of limited outlier residues may confer necessary conformational flexibility, enabling M HAR to function effectively under fluctuating stress conditions. The Ramachandran plot analysis confirms that M HAR possesses 95.79% favourable stereochemical properties and structural stability. This structural robustness is essential for maintaining M HAR activity during drought-induced oxidative stress. Consequently, M HAR contributes to enhanced drought tolerance by sustaining redox homeostasis and protecting plant cells from oxidative damage (Figure 3).
The structural analysis of ascorbate peroxidase (APX) revealed a well-folded protein dominated by α-helical regions interconnected by flexible loops, indicating high structural stability. Such stability is critical under drought stress, where dehydration and elevated reactive oxygen species (ROS) levels can compromise protein integrity. The conserved folding pattern of APX supports proper positioning of the heme prosthetic group and the ascorbate-binding site, ensuring efficient catalytic reduction of hydrogen peroxide (H₂O₂) to water. Enhanced structural stability of APX enables sustained enzymatic activity during drought-induced oxidative stress, thereby preventing excessive accumulation of H₂O₂ and protecting cellular components from oxidative damage. Furthermore, the functional efficiency of APX contributes to the maintenance of redox homeostasis as part of the ascorbate–glutathione cycle, supporting membrane stability and photosynthetic performance under water-deficit conditions. These results suggest that structurally robust APX plays a significant role in enhancing drought tolerance by strengthening the plant antioxidant defence system. The analysis showed that the majority of amino acid residues were distributed within the most favoured regions, corresponding mainly to α-helical and β-sheet conformations. A smaller proportion of residues occupied the additionally allowed regions, while only a negligible number of residues were observed in the disallowed region, represented by a single outlier. The high clustering of residues in favoured regions indicates proper backbone geometry and confirms the reliability of the predicted three-dimensional structure. The limited presence of outlier residues suggests that these positions may correspond to flexible loop regions or functionally important sites rather than structural anomalies. Overall, the Ramachandran plot validates the structural integrity and acceptable stereochemical quality of the protein model, supporting its suitability for further functional interpretation in the context of stress tolerance studies (Figure 4).
Structural modelling revealed that the phosphate transporter is an integral membrane protein composed predominantly of α-helical structures, with 12 transmembrane helices characteristic of the PHT1 family. Hydrophobic residues were mainly localised within the membrane-spanning regions, supporting proper membrane insertion and functional stability. Ramachandran plot analysis demonstrated that the majority of amino acid residues (99.04%) were located in the most favoured regions, primarily within the α-helix region, while only a minimal number of residues occurred in disallowed regions. This indicates correct backbone geometry and reliable structural quality of the predicted model. The stable α-helical architecture and favourable stereochemical properties suggest efficient phosphate transport under drought stress. Maintenance of phosphate uptake is likely to support energy metabolism and stress-responsive pathways, contributing to improved drought tolerance in plants (Figure 5).
PHYLOGENETIC RELATIONSHIPS:
Blast tree helps in evolutionary studies of the genes in family. Identifies orthologs and paralogs across related species. The branches represent evolutionary divergence. Proteins that are closer together on the tree share a more recent common ancestor and are more closely related. Some proteins appear to be only distantly related, indicated by longer branches.
The evolutionary relationship indicates conservation of stress responsiveness in Ec CALMO ULIN PROTEIN across related species such as Arabidopsis , maize, sorghum, fox millet, and rice (Figure 6).
The evolutionary relationship indicates conservation of stress responsive function across related species such as Arabidopsis , rice, maize, and finger millet. Prolamin sequences from monocot cereals cluster closely together, indicating high sequence conservation and a shared evolutionary origin. The placement of Eleusine coracana prolamin within the cereal clade suggests functional similarity with maize and rice prolamins, despite species-specific diversification. In contrast, non-cereal or dicot sequences are absent or distantly related, highlighting the lineage-specific expansion of prolamins in monocots. Overall, the tree supports the idea that prolamins evolved primarily within grasses and diversified to support grain development and stress adaptation (Figure 7).
The BLAST tree view suggests that query protein Ec monodehydroascorbate reductase is likely homologous to related proteins in grass species like maize, rice, and sorghum. It shares evolutionary ancestry with these proteins in stress tolerance. Eleusine coracana sequence clusters within a well-supported monocot clade of monodehydroascorbate reductase (M HAR) proteins, showing close evolutionary relationships with homologs from Oryza sativa, Zea mays, Setaria italica, and Sorghum bicolor. The short branch lengths indicate high sequence conservation, supporting functional preservation across cereal crops. M HAR is a key component of the ascorbate–glutathione cycle, where it catalyzes the regeneration of ascorbate and thereby mitigates drought-induced oxidative stress. uring water deficit, enhanced M HAR activity contributes to efficient reactive oxygen species scavenging, protection of photosynthetic machinery, and maintenance of cellular redox balance. The conserved phylogenetic placement of the E. coracana M HAR suggests an essential role in drought tolerance and underscores its potential importance in improving stress resilience in cereals through targeted breeding or biotechnological approaches (Figure 8).
The BLAST tree view suggests that query protein Ec ascorbate peroxidase is likely homologous to proteins in species like Arabidopsis , Digitalis , maize, rice, and sorghum. It shares evolutionary ancestry with these proteins in stress tolerance. The protein ascorbate peroxidase is highly conserved across angiosperms (Figure 9).
Table 1. Functional and physicochemical characteristics of drought-responsive proteins [CELLO:Subcellular Localisation Predictive System; Expasy – ProtParam; UniProt; WoLF PSORT: Protein Subcellular Localisation Prediction]
|
PROTEIN NAME |
SUBCELLULAR LOCATION |
GENE ONTOLOGY |
AA |
MW |
pI |
II |
AI |
GRAVY |
|
CALMO ULIN |
CYTOPLASMIC |
CALCIUM ION BIN ING ENZYME REGULATOR ACTIVITY |
82 |
9326.31 |
4.39 |
43.53 |
64.27 |
-0.740 |
|
PROLAMIN |
CYTOPLASMIC |
SPLICEOSOMAL COMPLEX POSITIVE REGULATION OF RESPONSE TO WATER STRESS |
130 |
14513.09 |
5.30 |
32.31 |
42.08 |
-1.008 |
|
M A |
MITOCHON RION INNER MEMBRANE |
NA H EHY ROGENASE ACTIVITY OXI O- RE UCTASE ACTIVITY |
101 |
10919.58 |
9.69 |
32.59 |
101.29 |
-0.003 |
|
ASCORBATE PEROXI ASE |
CYTOPLASM |
CELLULAR RESPONSE TO OXI ATIVE STRESS |
53 |
5698.40 |
11.29 |
94.16 |
77.36 |
-0.589 |
|
PHOSPHATE TRANSPORTER |
MULTI-PASS MEMBRANE |
PHOSPHATE ION TRANSPORT |
490 |
53400.07 |
9.01 |
31.70 |
90.69 |
0.430 |
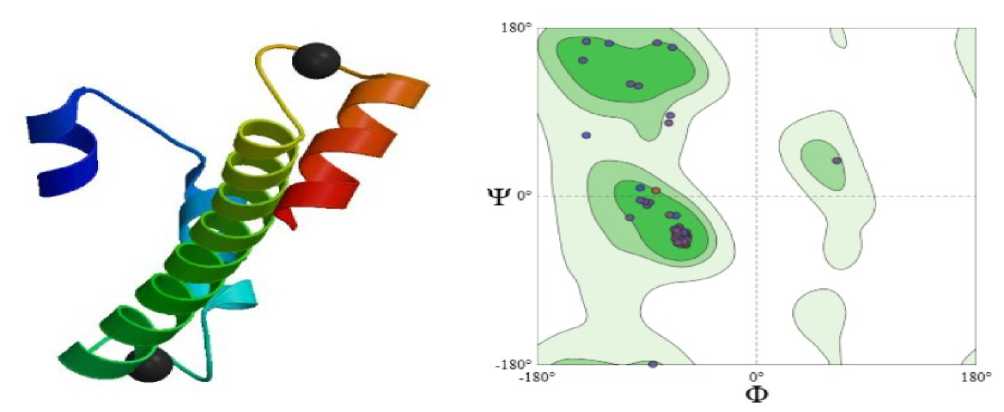
Figure 1 SHOWING STRUCTURE & RAMACHAN RAN PLOT OF Ec CALMO ULIN (SWISS-MO EL; Untitled Project | Structure Assessment)
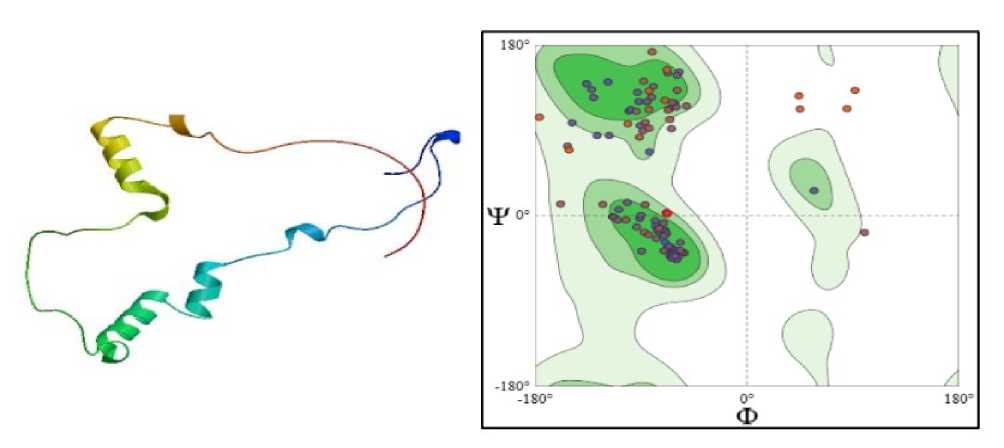
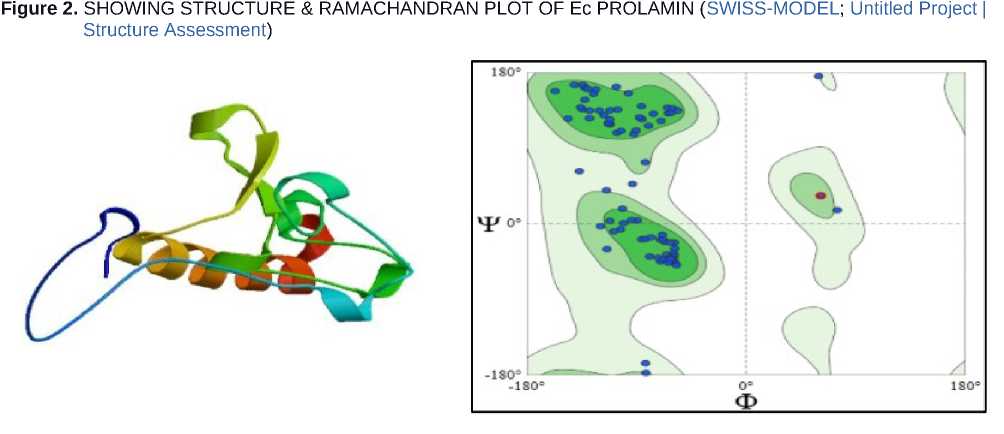
Figure 3. SHOWING STRUCTURE & RAMACHAN RAN PLOT OF Ec MONO EHY ROASCORBATE RE UCTASE (SWISS-MO EL; Untitled Project | Structure Assessment)
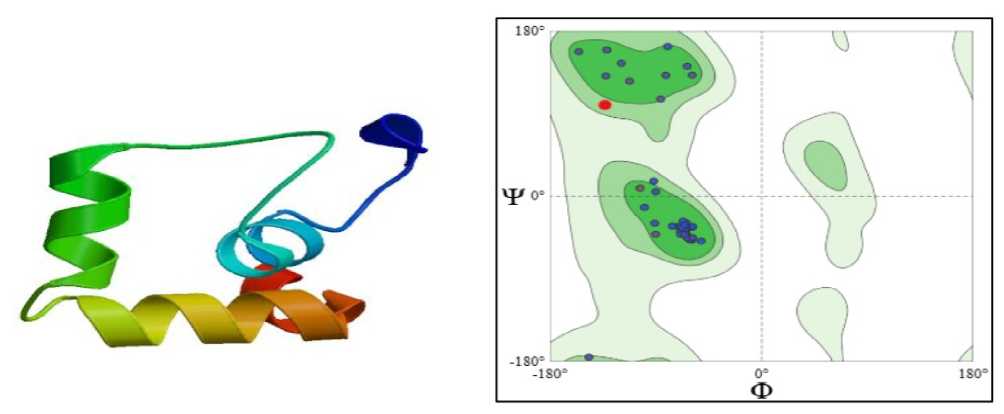
Figure 4. SHOWING STRUCTURE & RAMACHAN RAN PLOT OF Ec ASCORBATE PEROXI ASE (SWISS-MO EL; Untitled Project | Structure Assessment)
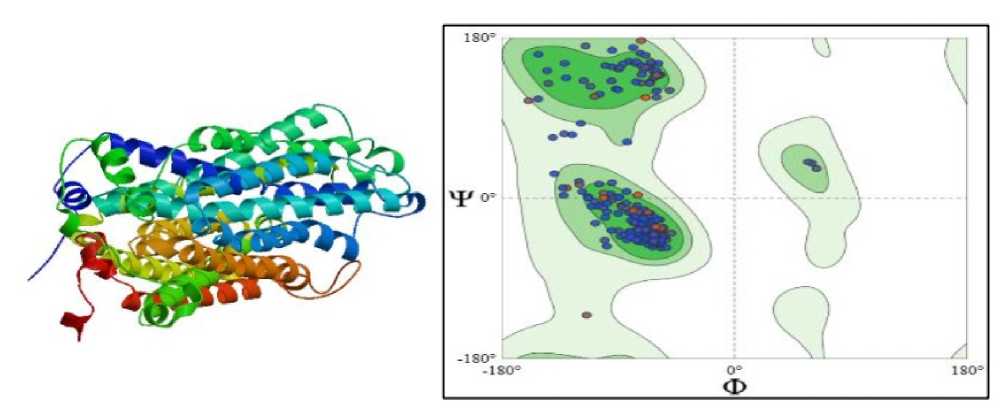
Figure 5. SHOWING STRUCTURE & RAMACHAN RAN PLOT OF Ec PHOSPHATE TRANSPORTER (SWISS-MO EL; Untitled Project | Structure Assessment)
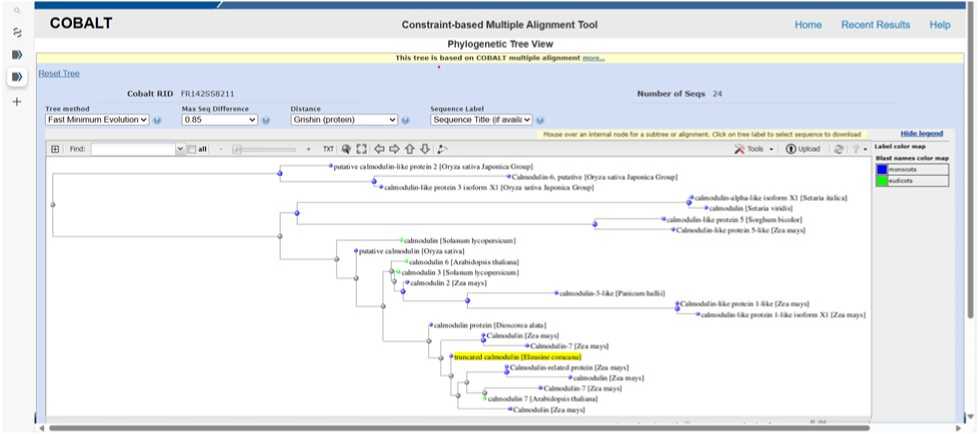
Figure 6. PHYLOGENETIC TREE SHOWING EVOLUTIONARY RELATIONSHIP OF Ec CALMO ULIN (Blast Tree View Widget; BLAST: Basic Local Alignment Search Tool; COBALT:Multiple Alignment Tool)
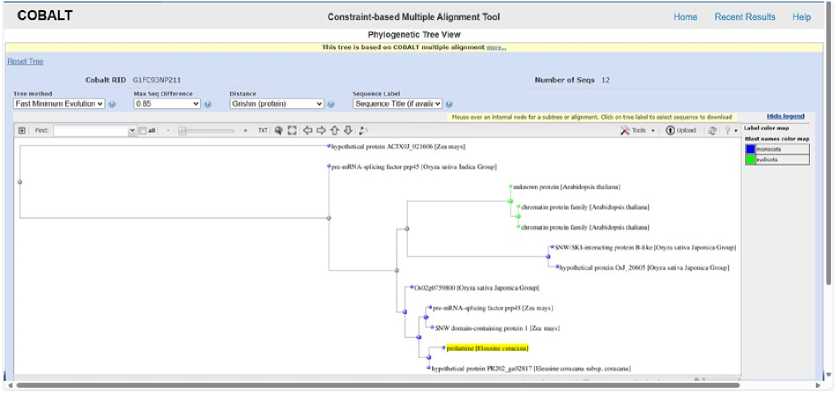
Figure 7. PHYLOGENETIC TREE SHOWING EVOLUTIONARY RELATIONSHIP OF Ec PROLAMIN (Blast Tree View Widget; BLAST: Basic Local Alignment Search Tool; COBALT:Multiple Alignment Tool)
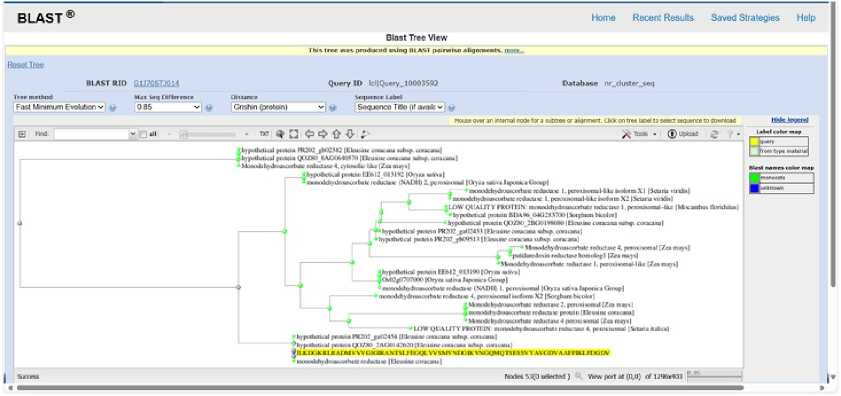
Figure 8. PHYLOGENETIC TREE SHOWING EVOLUTIONARY RELATIONSHIP OF Ec monodehydroascorbate reductase (Blast Tree View Widget; BLAST: Basic Local Alignment Search Tool; COBALT:Multiple Alignment Tool)
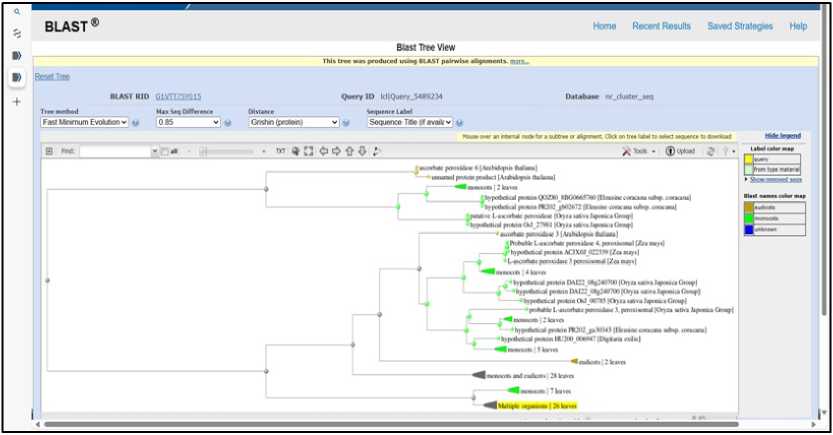
Figure 9. PHYLOGENETIC TREE SHOWING EVOLUTIONARY RELATIONSHIP OF Ec ASCORBATE PEROXI ASE (Blast Tree View Widget; BLAST: Basic Local Alignment Search Tool; COBALT:Multiple Alignment Tool)
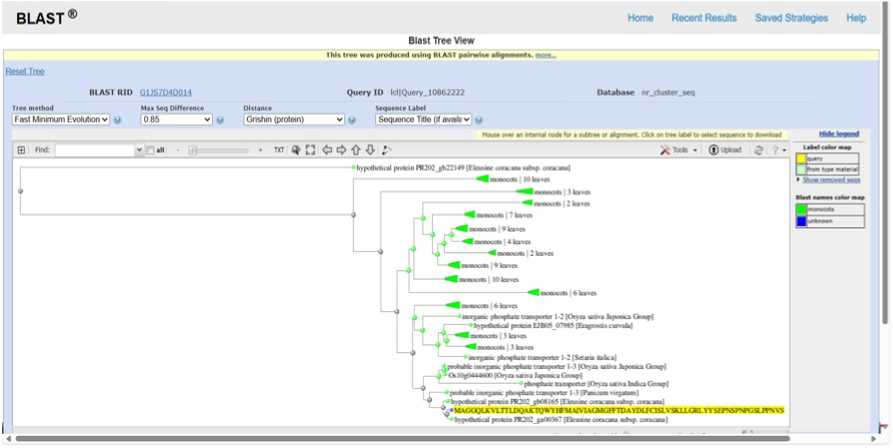
Figure 10. PHYLOGENETIC TREE SHOWING EVOLUTIONARY RELATIONSHIP OF Ec phosphate transporter (Blast Tree View Widget; BLAST: Basic Local Alignment Search Tool)
Phylogenetic analysis based on BLAST pairwise alignments placed the Eleusine coracana protein within a well-supported clade of monocot inorganic phosphate transporter (PHT1) proteins, closely related to homologs from Oryza sativa , Setaria italica , and Panicum virgatum . The tight clustering and short branch lengths indicate high sequence conservation, suggesting preservation of core transporter structure and function. Phosphate transporters are critical under drought stress, as reduced soil moisture severely limits phosphate diffusion and availability. Conservation of PHT1 proteins in drought-adapted grasses implies an essential role in sustaining phosphate uptake, ATP production, membrane integrity, and stress-responsive signaling during water deficit. The evolutionary proximity of the E. coracana transporter supports its involvement in drought resilience and underscores its potential utility as a candidate gene for enhancing phosphorus-use efficiency and drought tolerance in cereal crops (Figure 10).
DISCUSSION
Highly expressed proteins in finger millet represent classic stress-responsive classes (TFs, enzymes, chaperones). Some highly expressed proteins involved in stress tolerance in Eleusine coracana retrieved from
UNIPROT (Uniport Consortium, 2025) in FASTA format are >Q Z71191.1 catalase A1 Eleusine coracana ], >ACC59765.1 of-type zinc finger protein, partial Eleusine coracana ], >ACC77653.1 NAC transcription factor Eleusine coracana ], >ACF06728.1 cold shock protein, partial [ Eleusine coracana ], >ACF06727.1 ascorbate peroxidase 1, partial Eleusine coracana ], >ACL97372.1 truncated calmodulin, partial [ Eleusine coracana ], >A B43602.1 prolamin, partial [ Eleusine coracana ], >A C44447.1 monodehydroascorbate reductase, partial Eleusine coracana ],>AEF58885.1 NA H dehydrogenase subunit F, partial (chloroplast) Eleusine coracana ],>AEH04409.1 hydroxymethylglutaryl-CoA synthase, partial [ Eleusine coracana ], etc. Phylogenetic analyses indicate gene family expansions in C4 pathway and stress regulators, potentially underpinning finger millet’s resilience. In silico findings can guide reverse-genetic validation (e.g. CRISPR, overexpression, promoter assays) for climatesmart crop development. This study demonstrates a strong role played by these proteins in drought tolerance. Proteins with high predicted aliphatic index and low instability index are likely more resistant to denaturation under stress, which correlates with better physiological performance in the plant.
CONCLUSION
This in silico investigation identifies and characterizes a suite of highly expressed climateresilience proteins in E. coracana . Molecular phylogeny reveals both conserved and uniquely expanded gene families, reflecting evolutionary adaptation. These results lay the groundwork for targeted functional genomics to harness stress tolerance for finger millet improvement and translational breeding.
CONFLICTS OF INTEREST
The authors declares that they have no potential conflicts of interest.


