Противоопухолевые эффекты внеклеточного ощелачивания: гидрокарбонат натрия модулирует функцию митохондрий, динамику лизосом и миграционную активность опухолевых клеток
Автор: Богданов А.А., Богданов А.А., Бурдаков В.С., Митусова К.А., Моисеенко В.М.
Журнал: Сибирский онкологический журнал @siboncoj
Рубрика: Лабораторные и экспериментальные исследования
Статья в выпуске: 1 т.25, 2026 года.
Бесплатный доступ
Кислотность микроокружения опухоли представляет собой фундаментальный признак рака, способствующий прогрессированию опухоли, инвазии, уклонению от иммунного ответа и устойчивости к лечению. Цель исследования – изучение противоопухолевых механизмов ощелачивания гидрокарбонатом натрия на клетках аденокарциномы толстой кишки мыши CT26 и меланомы человека. Материал и методы. Клетки подвергали воздействию различных повышенных концентраций гидрокарбоната натрия (50–119 мМ) с целью оценки его влияния на жизнеспособность, метаболизм, миграцию и пути клеточной гибели. Результаты. Результаты показали немедленное повышение значений внеклеточного pH, зависящее от концентрации гидрокарбоната натрия, с последующим их понижением через 24 ч вследствие метаболической адаптации. Обе линии клеток демонстрировали дозозависимую цитотоксичность с концентрацией полумаксимального ингибирования примерно 80–90 мМ, при этом уровень апоптоза, определённый с помощью окрашивания Annexin V/PI, был минимален, что указывает на альтернативные механизмы клеточной гибели. Гидрокарбонат натрия значительно нарушал миграцию клеток в тестах по восстановлению монослоя и вызывал деполяризацию митохондрий, подтверждённую снижением флуоресценции зонда Mito Red. Метаболический анализ выявил повышенное потребление глюкозы и глутамина на фоне увеличенного образования лактата, что свидетельствует о метаболическом перепрограммировании в ответ на стресс ощелачивания. Несмотря на увеличение накопления лизосом, определенное с помощью зонда Lyso Green, классические маркеры аутофагии (LC3B и p62) не показали значимых изменений, свидетельствуя о том, что традиционные пути аутофагии не играют ведущей роли. Заключение. Полученные данные указывают на то, что ощелачивание гидрокарбонатом натрия индуцирует гибель опухолевых клеток посредством механизмов, выходящих за рамки классического апоптоза и аутофагии, возможно включая лизосом-зависимую смерть или алкалиптоз. Исследование дает механистическое обоснование потенциала гидрокарбоната натрия в качестве потенциальной адъювантной терапии, направленной на кислотность микроокружения опухоли, с перспективой повышения эффективности традиционных методов лечения рака через модуляцию pH. Для полного понимания задействованных путей клеточной гибели необходимы дальнейшие исследования.
Гидрокарбонат натрия, опухолевые клетки, деполяризация митохондрий, миграция, лизосомальное накопление, гибель клеток
Короткий адрес: https://sciup.org/140314348
IDR: 140314348 | УДК: 616-006.6:576.3:549.746.12]-092.9 | DOI: 10.21294/1814-4861-2026-25-1-62-73
Antitumor effects of extracellular alkalization: sodium bicarbonate modulates mitochondrial function, lysosomal dynamics and migration activity of cancer cells
Tumor microenvironment acidity represents a fundamental hallmark of cancer that promotes tumor progression, invasion, immune evasion, and treatment resistance. Objective. This study investigates the antitumor mechanisms of sodium bicarbonate-induced alkalization on mouse colon adenocarcinoma CT26 cells and human melanoma cells. Material and Methods. Cells were treated with varying sodium bicarbonate concentrations (50–119 mM) to assess effects on viability, metabolism, migration, and cell death pathways. Results. Results demonstrated immediate concentration-dependent extracellular pH elevation that decreased after 24 hours due to metabolic adaptation. Both cell lines exhibited dose-dependent cytotoxicity with an IC50 of approximately 80–90 mM, yet minimal apoptosis was detected via Annexin V/PI staining, suggesting alternative cell death mechanisms. Sodium bicarbonate signifcantly impaired cellular migration in wound healing assays, coinciding with mitochondrial depolarization as evidenced by reduced Mito Red fuorescence. Metabolic analysis revealed increased consumption of glucose and glutamine alongside elevated lactate production, indicating metabolic reprogramming in response to alkalization stress. While lysosomal accumulation increased with treatment (measured by Lyso Green), canonical autophagy markers (LC3B and p62) showed no signifcant changes, suggesting classical autophagy pathways are not primarily involved. Conclusion. These fndings indicate that sodium bicarbonate-induced alkalization triggers tumor cell death through mechanisms beyond conventional apoptosis and autophagy, potentially involving lysosome-mediated cell death or alkaliptosis. The study provides mechanistic insights supporting sodium bicarbonate as a potential adjuvant therapy that targets the tumor microenvironment’s acidity, with implications for enhancing conventional cancer treatments through pH modulation. Further research is needed to fully elucidate the precise cell death pathways involved.
Текст научной статьи Противоопухолевые эффекты внеклеточного ощелачивания: гидрокарбонат натрия модулирует функцию митохондрий, динамику лизосом и миграционную активность опухолевых клеток
Among the various biochemical characteristics of the tumor microenvironment (TME), acidity has emerged as a fundamental hallmark of cancer that significantly influences tumor biology and treatment outcomes [1, 2]. Tumor cells exhibit the inverted pH gradient phenomenon, where the extracellular space becomes acidic (pHe ~6.4–7.1) while maintaining a relatively alkaline intracellular pH (pHi ~7.1–7.8), in contrast to normal tissues [3, 4]. The acidic TME arises primarily due to metabolic reprogramming, notably the Warburg effect, whereby cancer cells preferentially utilize glycolysis leading to excess lactic acid and proton accumulation despite adequate oxygen supply [1, 5]. The acidic TME promotes tumor cell proliferation, invasion, immune evasion, and resistance to chemo- therapy and radiotherapy, thus posing a significant challenge in cancer treatment [1–3, 6].
Targeting tumor acidity through alkalization therapy has emerged as a promising complementary strategy to conventional cancer therapies. Buffering extracellular acidity can restore pH homeostasis and impair tumor progression by modulating cancer cell metabolism and improving the efficacy of chemotherapeutic agents, especially weakly basic drugs whose intracellular uptake is hindered by ion trapping in acidic conditions [1, 7–11]. Sodium bicarbonate (NaHCO3), a clinically established and cost-effective buffering agent, has demonstrated potential in preclinical models and clinical settings to elevate tumor extracellular pH (pHe), reduce metastasis, enhance tumor sensitivity to therapy, and improve survival outcomes [7–9, 12–24].
For instance, intraperitoneal perfusion with sodium bicarbonate significantly prolonged survival in murine Ehrlich ascites carcinoma models and showed promising clinical outcomes in malignant ascites associated with ovarian cancer resistant to chemotherapy [7]. Also the clinical study ChiCTR-IOR-14005319 found that adding local 5 % sodium bicarbonate to transarterial chemoembolization (TACE) in large-focal hepatocellular carcinoma increased the objective response rate to 100 %, compared to 44.4 % in a nonrandomized and 63.6 % in a randomized conventional TACE cohort [13]. In the recently published real-world study (ChiCTR-ONC-17013416) involving 413 patients with hepatocellular carcinoma (9.7 % early-stage, 7.0 % intermediate-stage, and 83.3 % advanced-stage), bicarbonate-integrated TACE achieved an objective response rate of 99.01 %, including complete responses in 72.77 % of cases [25].
The mechanisms underlying the antitumor effects of sodium bicarbonate appear multifaceted. It is hypothesized that beyond neutralizing extracellular acidity, sodium bicarbonate affects intracellular pH dynamics, interferes with lactate-proton cotransport via monocarboxylate transporters, and potentially induces a novel form of cell death known as “alkalip-tosis” [1, 7]. Additionally, by reversing the inhibitory effects of lactic acid on T-cells, sodium bicarbonate can potentially re-energize metabolically impaired immune cells within the TME [26]. Moreover, despite promising preclinical and clinical findings, the direct effects of sodium bicarbonate on specific cancer cell lines in vitro have not yet been fully elucidated. A recent study demonstrates that sodium bicarbonate induces cytotoxicity in ovarian cancer cells and inhibits their migration and invasion in vitro . When combined with Olaparib, sodium bicarbonate significantly potentiates the anticancer effects [27]. Another study demonstrated that sodium bicarbonate-induced alkalinization disrupted mitochondrial function and elevated adenosine monophosphate (AMP) levels, initially triggering autophagy but ultimately inhibiting it under alkaline conditions, thus promoting cell death [28].
In this study, we examined the effects of sodium bicarbonate on mouse colon adenocarcinoma CT26 cells and human melanoma cells cultured in vitro to assess its impact on critical cancer cell behaviors. Specifically, we aimed to characterize how extracellular alkalization influenced cell viability, proliferation, apoptosis, glucose and migration, thereby elucidating potential mechanisms through which tumor alkalization may exert anti-cancer effects. The findings are intended to inform the development of novel pharmacological approaches targeting tumor acidity within the tumor microenvironment to enhance cancer treatment efficacy.
Material and Methods
Reagents and Cell Culture
Mouse adenocarcinoma CT26 cells (ATCC Number: CRL-2638) and a primary adhesive melanoma cell line derived from a patient tumor (own collection) were used in this study. Both cell lines were maintained in a humidified incubator at 37 °C with 5 % CO2. CT26 cells were cultured in RPMI-1640 medium (PanEco, Russia or Servicebio, China), while melanoma cells were cultured in DMEM/F12 medium (PanEco, Russia). Both media were supplemented with 10 % fetal bovine serum (FBS) and 50 µg/mL gentamicin (PanEco, Russia). To adjust the pH and alkalize the culture medium, a 4 % sodium bicarbonate (NaHCO3) solution (Sigma-Aldrich, Germany) was added to the complete medium to achieve the desired sodium bicarbonate concentration, taking into account the baseline bicarbonate content present in RPMI-1640 (0.024 M) and DMEM/F12 (0.028 M).
Culture Medium pH Measurements
The pH of the culture medium was determined indirectly by analyzing changes in the spectral properties of phenol red, the pH indicator present in the medium. Phenol red exhibits distinct absorption maxima at wavelengths of 435 nm, 450 nm, and 560 nm, which vary depending on the pH. The absorption ratios at these wavelengths were used to generate a calibration curve for titration and to calculate the precise pH of the culture medium. For calibration, the pH of the medium was directly measured using an AB33PH-F pH meter equipped with an ST310 electrode (Ohaus, USA) or an FP20-TRIS pH meter fitted with an LE420 electrode (Mettler Toledo, Switzerland). Optical density readings were obtained using either an EnSpire Multilabel Plate Reader (PerkinElmer, USA) or a NanoPhotometer NP80 spectrophotometer (Implen, Germany).
Cytotoxicity Assay
To assess cell viability, cells were seeded at a density of 1 × 10⁵ cells per well in 24-well plates (SPL, Korea) two days prior to the start of the experiment. At the start of the experiment, the culture medium in each well was completely replaced with fresh medium containing varying concentrations of sodium bicarbonate. Cell viability and death pathways were analyzed by flow cytometry using a Longcyte cytometer (Challenbio, China). Total cell concentration was determined by dividing the total number of recorded events by the sample volume. The number of viable cells was calculated by multiplying the total cell concentration by the percentage of cells negative for PI and annexin V-FITC staining. Apoptosis and necrosis were evaluated using the Annexin V-FITC/PI apoptosis detection kit (Servicebio, China) following the manufacturer’s protocol. Flow cytometry results were analyzed using FlowJo X 10.0.7r2 software (BD Biosciences, USA).
Wound Healing Assay
The wound healing assay was performed to evaluate the migratory capacity of cells and their ability to repair a scratched monolayer. Cells were seeded in 24-well plates and cultured until reaching full confluence
(100 %). A uniform linear scratch (“wound”) was then created in the center of each well’s monolayer using a sterile 100-µL pipette tip. To remove detached cells and debris resulting from the scratch, wells were gently washed twice with phosphate-buffered saline (PBS). Fresh culture medium containing the specified sodium bicarbonate concentrations was added to experimental wells, while control wells received medium with baseline bicarbonate levels corresponding to RPMI-1640 (0.024 M) or DMEM/F12 (0.028 M). Images of the wound area were captured immediately after scratching (0 h) and at subsequent time points up to 48 h using the JuLI Stage automated live-cell imaging system (NanoEntek, South Korea). Cell migration was quantified by measuring the wound area at each time point using ImageJ software. Results were expressed as the percentage of wound closure relative to the initial wound area at 0 h.
Metabolite Consumptionand Production Measurements
To quantify glucose, glutamine, and lactate concentrations in the culture medium, cells were seeded at a density of 1×105 cells per well in 24-well plates and cultured for 48 hours. Prior to the experiment, the culture medium was completely replaced with fresh medium containing the specified sodium bicarbonate concentrations. Metabolite concentrations in the medium were measured using an M-1000 Bioprocess Biochemistry Analyzer (Siemanbio, China) after 24 hours of incubation. Consumption or production rates of metabolites were calculated per 106 viable cells by determining the difference in metabolite concentration between two time points normalized to the average number of live cells during that interval. Viable cell counts were obtained using a Luna-II automated cell counter (Logos Biosystems, South Korea).
Autophagy flow cytometry studies
For flow cytometric analysis of autophagy, cells were seeded in 24-well plates at a density of 3×104 to 5×104 cells per well, 48 hours prior to the experiment, and maintained under the previously described culture conditions. On the day of the experiment, the culture medium was completely replaced with fresh medium containing the designated concentrations of sodium bicarbonate. Cells were then incubated for 24 hours. Autophagy activity was assessed by intracellular staining of the autophagy-related proteins LC3B and SQSTM1/p62. Cells were fixed, permeabilized, and incubated with primary antibodies targeting LC3B (AF4650, Affinity Bioscience, China) and SQSTM1/ p62 (AF5384, Affinity Bioscience, China), followed by incubation with corresponding fluorophore-conjugated secondary antibodies (FNSA-0031, Fine Biotech Co., Ltd., China) according to the manufacturers’ protocols. Flow cytometry data acquisition was performed, and subsequent analysis was carried out using FlowJo software version X 10.0.7r2 (BD Biosciences, USA).
Autophagy markers were quantified based on fluorescence intensity profiles to evaluate autophagic activity under different experimental conditions.
Mitochondrial Polarizationand Lysosome Studies
Fluorescence microscopy was employed to assess changes in mitochondrial membrane potential and lysosomal fluorescence intensity in response to medium alkalization in cell lines. Cells were seeded in 24-well plates at a density of 5×104 cells per well and cultured for 48 hours under standard conditions. Prior to the experiment, the culture medium was completely replaced with freshly prepared medium containing the designated sodium bicarbonate concentrations, and the cells were incubated for 4 hours to allow equilibration. Subsequently, cells were stained with fluorescent probes according to the respective manufacturers’ protocols: Lyso Green (Lumiprobe, Russia) for lysosomal labeling, Mito Red CMXRos (Lumiprobe, Russia) for assessing mitochondrial membrane potential, and Hoechst 33342 (Lumiprobe, Russia) for nuclear staining as a reference. As a positive control for mitochondrial membrane depolarization, cells were treated with carbonyl cyanide m-chlorophenyl hydrazone (CCCP) (Elabscience, USA) following the manufacturer’s instructions. CCCP disrupts mitochondrial membrane potential, serving to validate the sensitivity and specificity of the mitochondrial assay. Fluorescence imaging was performed using the JuLI Stage automated live-cell imaging system (NanoEntek, South Korea). Quantitative analysis of fluorescence intensity from mitochondrial and lysosomal dyes was conducted using ImageJ software, with signal normalization applied as necessary to facilitate comparison across conditions.
Statistical Analysis
All experiments were conducted with a minimum of three independent biological replicates to ensure reproducibility and reliability of the results. Each measurement within the experiments was performed in triplicate or more to account for technical variability. Statistical analyses were performed using a combination of software tools, including Excel2016 (Microsoft Corporation, USA) and OriginLab2019b (OriginLab Corporation, USA), as well as other specialized software packages referenced earlier in the Methods section. Quantitative data are presented as mean ± standard deviation (SD) unless otherwise specified. Group comparisons were performed using statistical tests selected based on data distribution and experimental design. Normality was assessed with the Shapiro–Wilk test. Parametric tests, such as the Student’s t-test, were applied to normally distributed data, while the nonparametric Mann–Whitney U test was used for non-normally distributed data. A p-value<0.01 was considered statistically significant.
Results
Our experimental results demonstrated a clear, concentration-dependent alkalization of the culture medium following sodium bicarbonate treatment. Specifically, we observed a progressive increase in extracellular pH correlating with rising sodium bicarbonate concentrations, measured immediately after treatment (Figure 1 A, B). However, after 24 hours, the pH of the culture medium decreased slightly more pronouncedly than in control samples of cells cultured in unmodified medium. This pH reduction is attributed to dynamic shifts in cellular metabolic activity in response to alkalization-induced stress.
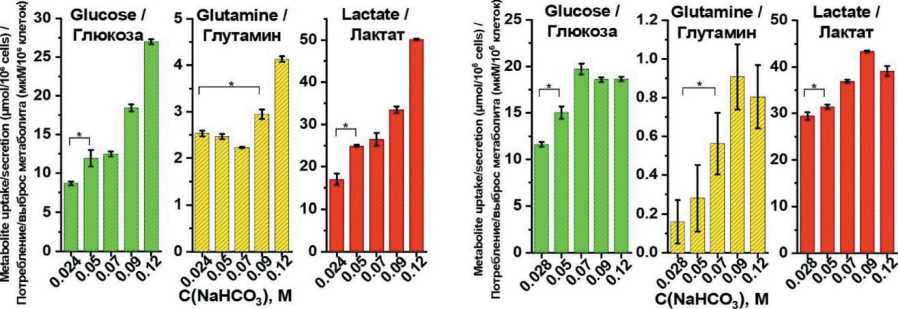
Under elevated pH conditions, both CT26 and human melanoma cells exhibited altered metabolic profiles characterized by increased consumption of key nutrients, including glucose and glutamine, alongside enhanced lactate production (Figure 1 C, D). These metabolic changes likely reflect an adaptive cellular response aimed at maintaining energy production and redox homeostasis in the response to the elevated extracellular pH. The increased lactate secretion con- tributes substantially to the acidification of the medium observed at 24 hours, effectively counteracting the initial bicarbonate-induced alkalinization.
In accordance with these metabolic alterations, increased concentrations of sodium bicarbonate induced dose-dependent cytotoxic and/or antiproliferative effects in both CT26 and human melanoma cell lines. It should be noted that the addition of sodium chloride to the culture medium to achieve an equivalent increase in theoretical osmolarity as those induced by sodium bicarbonate – but without altering pH – did not result in any significant cytotoxic effect (data not shown). The half-maximal sodium bicarbonate inhibitory concentration (IC50) was estimated at approximately 80–90 mM for both cell types, indicating a comparable sensitivity to alkalization stress (Figure 2 A, B). Notably, flow cytometric analysis of apoptotic markers revealed that the proportion of cells positive for Annexin V alone or in combination with propidium iodide (Annexin V/PI) remained minimal at both early (4-hour) and later (24-hour) timepoints (Figure 2 C, D). These findings suggest that the cytotoxic response
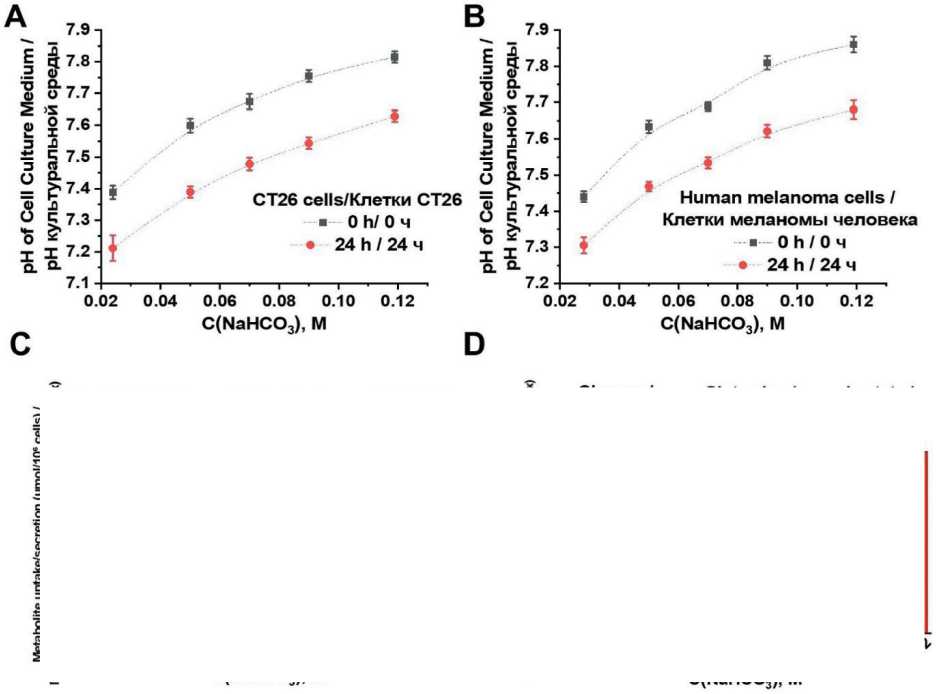
Fig. 1. Effects of sodium bicarbonate treatment on culture medium pH and cellular metabolism in CT26 and melanoma cells. Changes in culture medium pH for CT26 (A) and melanoma cells (B) measured immediately (0 h) and after 24 hours of incubation with varying concentrations of sodium bicarbonate. Quantification of glucose and glutamine uptake, as well as lactate secretion (µmol/106 cells), in CT26 (C) and melanoma cells (D) following 24-hour exposure to different sodium bicarbonate concentrations.
Notes: data are presented as mean ± standard deviation; * – p<0.01 versus corresponding control group; created by the authors Рис. 1. Влияние гидрокарбоната натрия на pH культуральной среды и клеточный метаболизм в клетках CT26 и меланомы. Изменения pH культуральной среды для клеток CT26 (A) и меланомы (B), измеренные сразу (0 ч) и после 24 ч инкубации с различными концентрациями гидрокарбоната натрия (C, D). Количественная оценка потребления глюкозы и глутамина, а также секреции лактата (мкмоль/106 клеток) в клетках CT26 (C) и меланомы (D) после 24-часового воздействия различных концентраций гидрокарбоната натрия. Примечания: данные представлены как среднее значение ± стандартное отклонение; * – различия значимы по сравнению с соответствующей контрольной группой (p<0,01); рисунок выполнен авторами
is primarily driven by mechanisms other than classical apoptosis, such as proliferation arrest or necrosis, under sustained alkalized conditions.
Wound healing assays demonstrated that increasing sodium bicarbonate concentrations in the culture medium caused a pronounced and statistically significant reduction in the migratory capacity of both CT26 and human melanoma cells (Figure 3 A, B). Quantitative image analysis revealed a clear dose-dependent inhibition of wound closure throughout the assay, indicating that alkalization substantially impairs cellular motility mechanisms critical for migration and tissue repair. Considering the concurrent metabolic changes and reduced viability under alkaline conditions, we hypothesized that disturbances in cellular energy homeostasis may underlie these migratory defects.
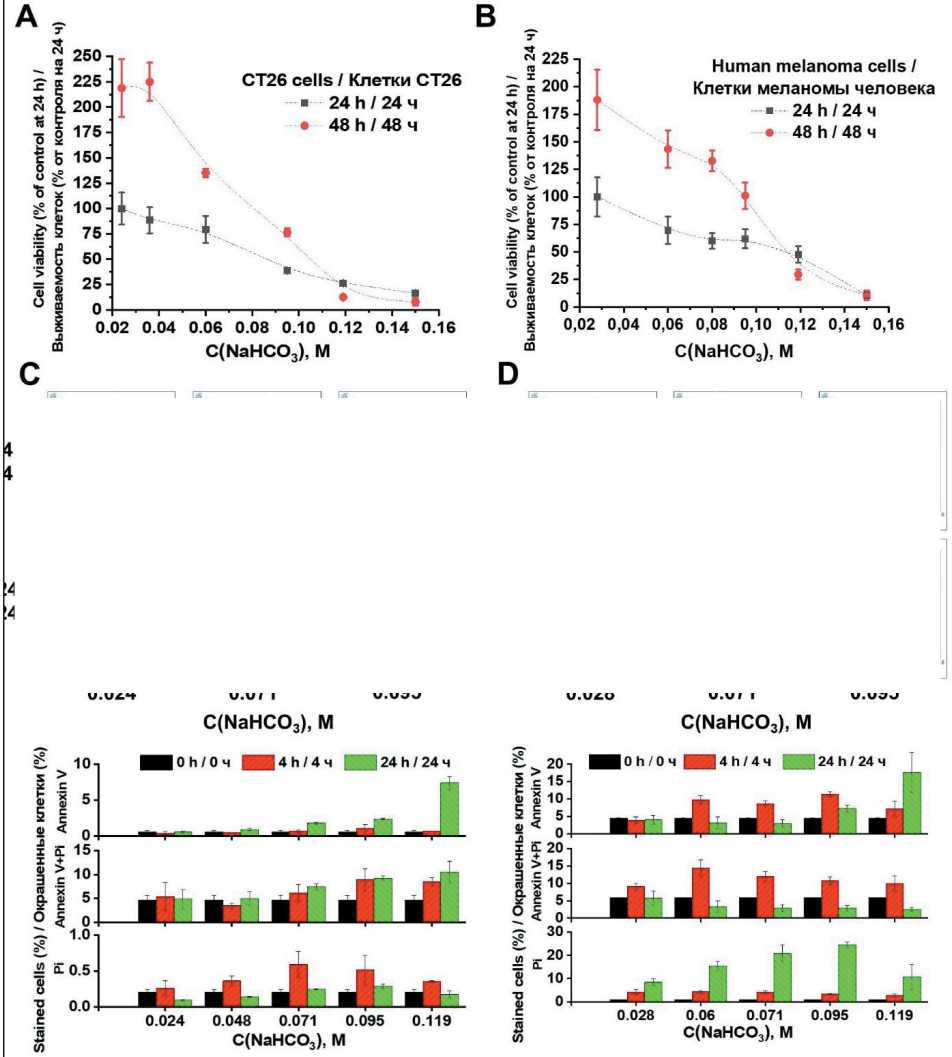
Fig. 2. Effects of sodium bicarbonate on cell viability and apoptosis in CT26 and melanoma cells. Cell viability of CT26 (A) and melanoma cells (B) following treatment with varying concentrations of sodium bicarbonate, assessed by flow cytometry (Control group- cell in unmodified medium at 24 h). Flow cytometric analysis of apoptotic markers in CT26 (C) and melanoma cells (D) using Annexin V staining alone and in combination with propidium iodide (Annexin V/PI) (Control group – cells in unmodified medium at 0 h).
Notes: data are presented as mean ± standard deviation; * – p<0.01 versus corresponding control group; created by the authors Рис. 2. Влияние гидрокарбоната натрия на жизнеспособность клеток и апоптоз в клетках CT26 и меланомы. Жизнеспособность клеток CT26 (A) и меланомы (B) после обработки различными концентрациями гидрокарбоната натрия, оцененная методом проточной цитометрии (Контрольная группа – клетки в немодифицированной среде через 24 ч). Анализ маркеров апоптоза методом проточной цитометрии в клетках CT26 (C) и меланомы (D) с использованием окрашивания аннексином V в отдельности и в комбинации с йодидом пропидия (аннексин V/PI) (контрольная группа – клетки в немодифицированной среде на 0 ч). Примечания: данные представлены как среднее значение ± стандартное отклонение; * – различия значимы по сравнению с соответствующей контрольной группой (p<0,01); рисунок выполнен авторами
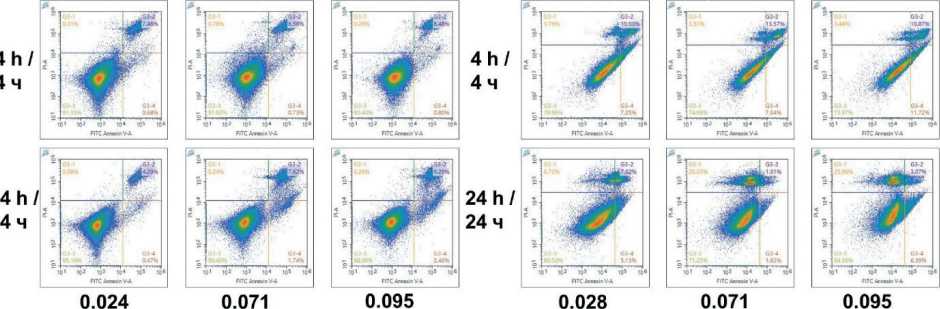
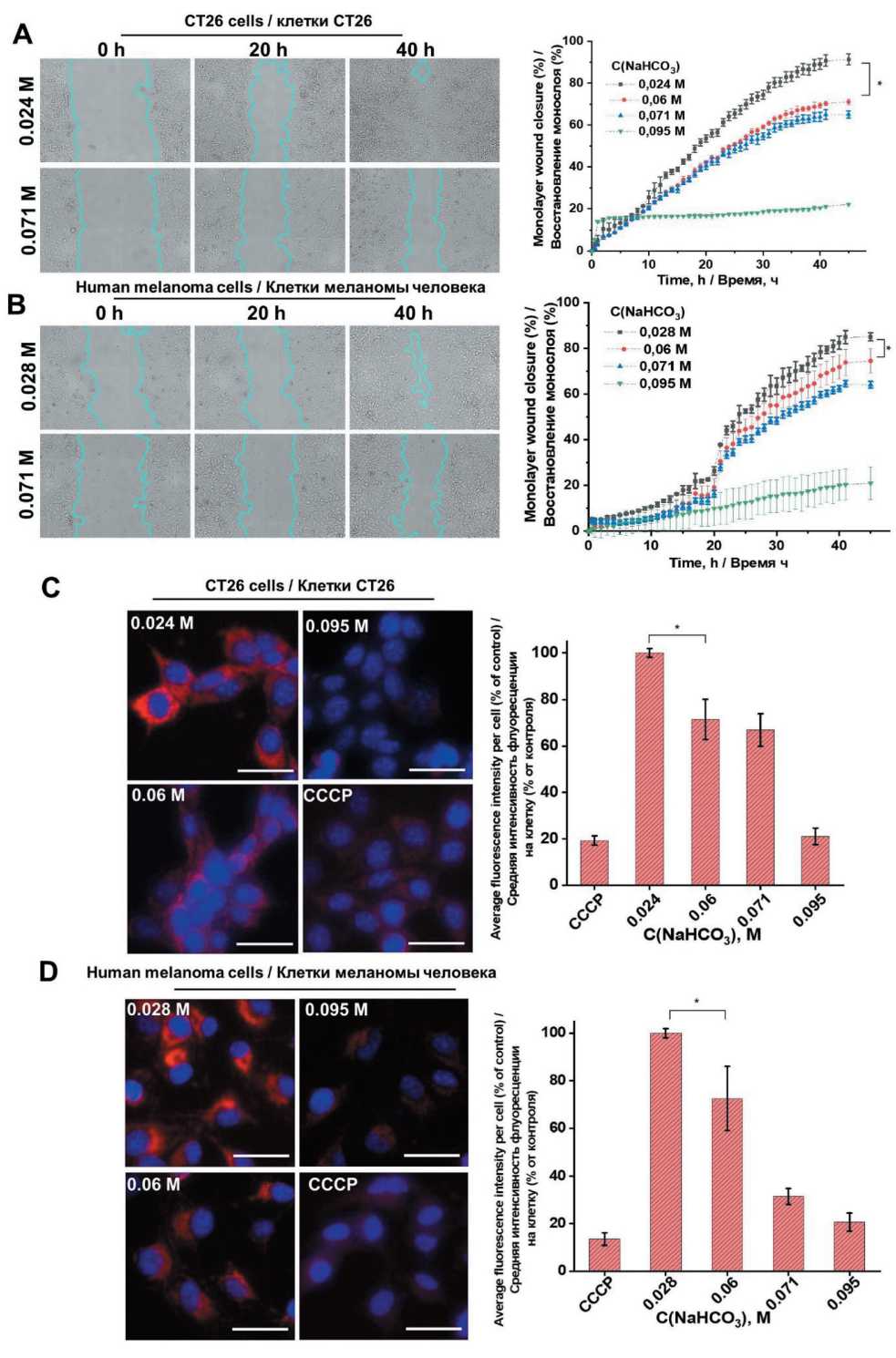
Fig. 3. Wound healing and mitochondrial polarization assays in CT26 and melanoma cells treated with sodium bicarbonate. Wound healing assays showing the migratory capacity of CT26 (A) and melanoma cells (B) after 48 hours incubation with sodium bicarbonate at concentrations ranging from 0.024 to 0.119 M. Fluorescence intensity of MitoRed CMXRos staining in CT26 (C) and melanoma cells (D) following 4-hour exposure to sodium bicarbonate (0.024 to 0.095 M). Mitochondria are shown in red and cell nuclei stained with Hoechst 33342 are shown in blue. Carbonyl cyanide m-chlorophenyl hydrazone (CCCP) served as a positive control for mitochondrial membrane depolarization. Notes: scale bar = 50 μm; data are presented as mean ± standard deviation; * – p<0.01 versus corresponding control group; created by the authors
Рис. 3. Анализ восстановления монослоя и поляризации митохондрий в клетках CT26 и меланомы, обработанных гидрокарбонатом натрия. Результаты анализа восстановления монослоя, демонстрирующие миграционную способность клеток CT26 (A) и меланомы (B) после 48-часовой инкубации с гидрокарбонатом натрия в концентрациях от 0,024 до 0,119 М. Интенсивность флуоресценции окрашивания MitoRed CMXRos в клетках CT26 (C) и меланомы (D) после 4-часового воздействия гидрокарбоната натрия (0,024–0,095 М). Митохондрии показаны красным цветом, ядра клеток, окрашенные Hoechst 33342, – синим. Карбонилцианид м-хлорфенилгидразон (CCCP) использовался в качестве положительного контроля деполяризации митохондриальной мембраны. Примечания: масштабная линейка = 50 мкм; данные представлены как среднее значение ± стандартное отклонение; * – различия значимы по сравнению с соответствующей контрольной группой (p<0,01);
рисунок выполнен авторами
To investigate this, we assessed mitochondrial membrane potential ‒ a key indicator of mitochondrial health and cellular energetic status ‒ using Mito Red staining combined with fluorescence microscopy. Per-cell fluorescence intensity was measured across multiple fields of view, providing single-cell resolution of mitochondrial polarization. This analysis revealed a robust, concentration-dependent decrease in Mito Red fluorescence intensity in both cell lines after sodium bicarbonate treatment (Figure 3 C, D), indicative of mitochondrial depolarization. This depolarization suggests compromised mitochondrial membrane integrity and potential dysfunction in the electron transport chain, likely resulting in impaired ATP synthesis. Impaired energy production may adversely affect cytoskeletal organization and signaling pathways necessary for cell motility, thereby explaining the diminished migration observed in the wound healing assays under alkalized conditions.
Building on these findings and in accordance with previous reports indicating that extracellular alkalization can disrupt mitochondrial function and induce autophagic responses [28], we further explored autophagic activity in treated cells. Using fluorescence microscopy with the lysosome-specific dye Lyso Green, we detected a marked, concentration-dependent increase in lysosomal accumulation within both CT26 and human melanoma cells exposed to sodium bicarbonate (Figure 4 A, B). The enhanced Lyso Green fluorescence likely reflects increased lysosome biogenesis or accumulation, phenomena commonly associated with autophagy activation or lysosomal stress responses.
Contrary to expectations, however, flow cytometric analysis of canonical autophagy markers LC3B and SQSTM1/p62 showed no significant alteration in their expression levels following sodium bicarbonate treatment (Figure 4 C, D). LC3B is a critical component of autophagosome membranes, while p62 serves as a selective autophagy receptor typically degraded during autophagic flux. The unchanged levels of these proteins suggest that canonical autophagy induction and progression may not be occurring despite the increased lysosomal content. This discrepancy may indicate a complex regulatory response to alkalization wherein lysosomal biogenesis and autophagy marker expression are uncoupled, or autophagy might be impaired or stalled at certain stages.
Alternatively, the observed lysosomal changes could reflect activation of other cellular stress response mechanisms distinct from classical autophagy, such as lysosomal stabilization, altered endocytic trafficking, or non-canonical autophagy pathways. Collectively, these results demonstrate that sodium bicarbonate-induced extracellular alkalization triggers significant lysosomal remodeling but does not unequivocally activate canonical autophagy in CT26 and human melanoma cells.
Discussion
Our experimental results demonstrated a logical concentration-dependent increase in extracellular pH following sodium bicarbonate supplementation in the culture medium of CT26 and human melanoma cells (Figure 1 A, B). This alkalization was associated with a significant reduction in cell viability and/or proliferative capacity, as reflected by lower total live cell counts compared to untreated controls (Figure 2 A, B). Notably, cells remaining after exposure to elevated sodium bicarbonate concentrations exhibited only weak annexin V and PI staining, suggesting that classical apoptotic and necrotic pathways do not fully explain the observed cytotoxic effects (Figure 2 C, D). This suggests alternative cell death mechanisms that merit further investigation.
Sodium bicarbonate cytotoxicity findings are consistent with previously reported studies in other cancer models, including liver cancer-derived SK-HEP-1 [28] and ovarian cancer-derived A2780 and SKOV3 [27] cell lines, where increasing sodium bicarbonate concentrations similarly reduced cell viability. The reproducibility of these effects across diverse tumor types underscores the role of pH modulation ‒ both extracellularly and intracellularly ‒ in disrupting critical cellular processes such as metabolism, ionic homeostasis, and enzyme function, thereby undermining tumor cell survival. However, we must note that not only pH changes, but also the concomitant alterations in bicarbonate ion concentration and increased osmolarity may contribute to the effects of sodium bicarbonate, warranting further investigation.
In addition to cytotoxicity, elevated sodium bicarbonate impaired cellular migratory capacity, as evidenced by wound healing assays (Figure 3 A, B). This observation aligns with in vivo studies reporting anti-invasive and antimetastatic effects of oral sodium bicarbonate administration [22, 23]. Tumor microenvironment alkalization likely interferes with the activity of proteases involved in invasion, including cathepsins and matrix metalloproteinases, by altering their optimal physicochemical milieu [23]. Our prior studies using isolated perfusion models of malignant ascites and limb tumors further support these findings, with sodium bicarbonate treatment correlating with prolonged survival [7, 29].
Moreover, our metabolic profiling demonstrated increased uptake of glucose and glutamine, coupled with heightened lactate production (Figure 1 C, D). This metabolic shift likely reflects an adaptive response to mitochondrial impairment (Figure 3 C, D), whereby enhanced glycolytic flux supports cellular energy demand and contributes to intracellular pH regulation through lactic acid generation. Conversely, the enhanced uptake of glucose and other energetic substrates in the presence of sodium bicarbonate mitigates lactate-induced acidosis by promoting the formation of lactate salts ‒ predominantly lactic acid
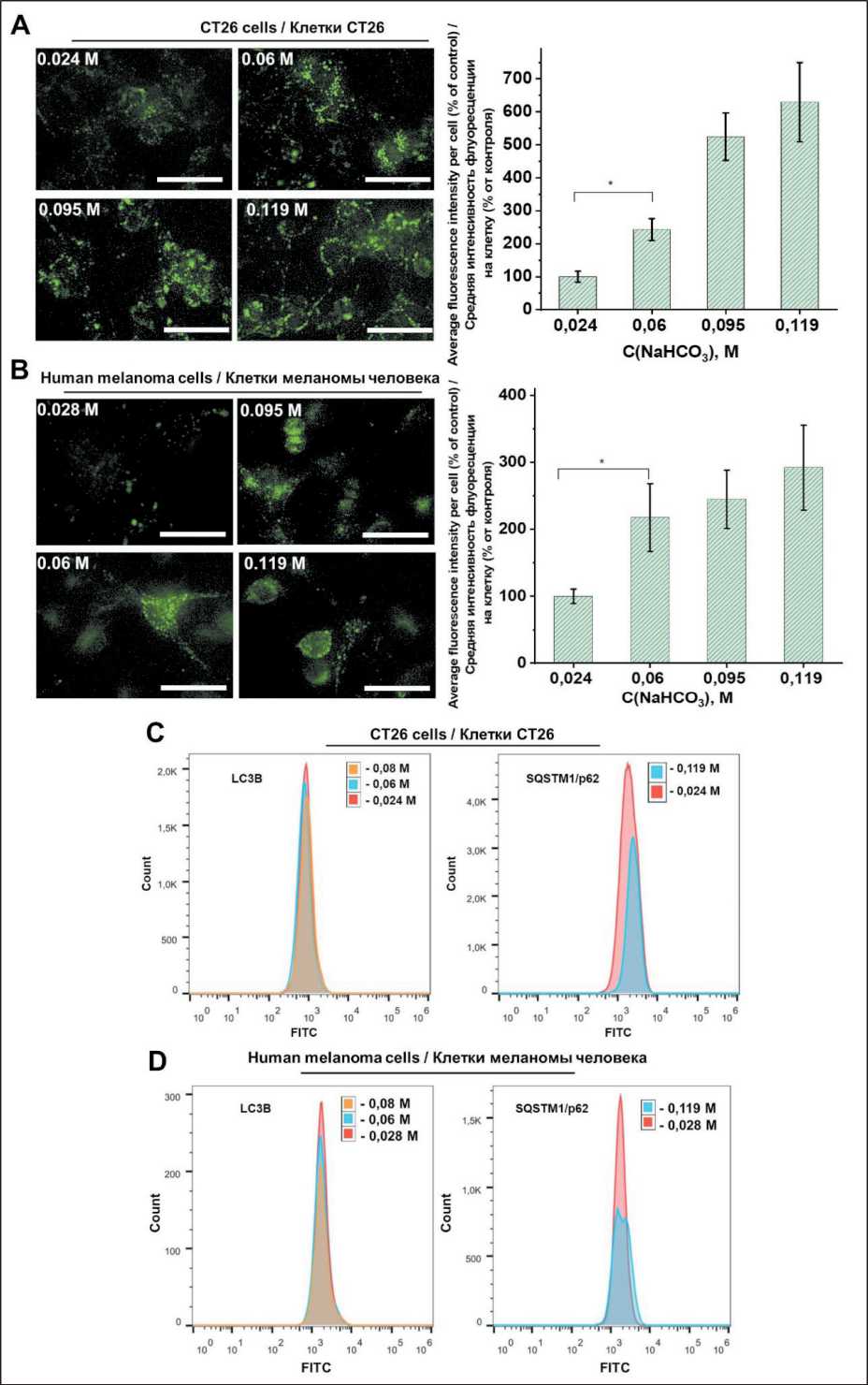
Fig. 4. Effects of sodium bicarbonate on lysosomal fluorescence and autophagy markers in CT26 and melanoma cells. Changes in
LysoGreen fluorescence intensity in CT26 (A) and melanoma cells (B) exposed to increasing concentrations of sodium bicarbonate (0.024–0.119 M). Flow cytometric analysis of autophagy-related proteins LC3B and SQSTM1/p62 in CT26 (C) and melanoma cells (D) following treatment with sodium bicarbonate (0.024–0.095 M). Notes: scale bar = 50 μm; data are presented as mean ± standard deviation; * – p<0.01 versus corresponding control group; created by the authors
Рис. 4. Влияние гидрокарбоната натрия на лизосомальную флуоресценцию и маркеры аутофагии в клетках CT26 и меланомы. Изменения интенсивности флуоресценции LysoGreen в клетках CT26 (A) и меланомы (B) при воздействии возрастающих концентраций гидрокарбоната натрия (0,024–0,119 М). Проточная цитометрия маркеров аутофагии LC3B и SQSTM1/p62 в клетках CT26 (C) и меланомы (D) после обработки гидрокарбонатом натрия (0,024–0,095 М).


