Роль кишечной микробиоты при иммунотерапии рака толстой кишки
Автор: Хакимова Г.Г., Трякин А.А., Заботина Т.Н., Цуканов А.С., Алиев В.А., Гуторов С.Л.
Журнал: Злокачественные опухоли @malignanttumors
Рубрика: Обзоры и аналитика
Статья в выпуске: 2 т.9, 2019 года.
Бесплатный доступ
Считается, что генетические факторы, дисфункция иммунной системы, хроническое воспаление и дисбиоз кишечной микробиоты (КМ) являются частью патогенеза колоректального рака (КРР). Положительная роль регуляции КМ при лечении воспалительных заболеваний кишечника определяется снижением роста патогенных бактерий и увеличением продукции противовоспалительных факторов. На сегодняшний день, современные данные свидетельствуют о том, что КM дисрегулирует иммунный ответ против опухоли в ее микроокружении, тем самым замедляя либо ускоряя эффективность противоопухолевой терапии. В клинических исследованиях сообщается о преимуществах терапии КРР с учетом состава КM, в отношении улучшения иммунного гомеостаза кишечника, функции эпителиального барьера и качества жизни. Между тем, специфическая сигнатура КМ может модулировать чувствительность к химио- и/или лучевой терапии и прогноз пациентов раком толстой кишки. В данной статье, мы представили общие проблемы терапии КРР, основанной на данных по КМ в сочетании с иммунотерапией, а также описали направления будущих перспектив.
Кишечная микробиота, иммунотерапия
Короткий адрес: https://sciup.org/140243822
IDR: 140243822 | DOI: 10.18027/2224-5057-2019-9-2-5-11
The role of the intestinal microbiome in the immunotherapy of colon cancer
Genetic factors, immune dysfunction, chronic inflammation, and dysbiosis of the intestinal microbiome (IM) are believed to participate in the pathogenesis of colorectal cancer (CRC). The positive role of IM regulation in the treatment of inflammatory bowel disorders is determined by a reduction in the growth of pathogenic bacteria and an increase in the production of anti-inflammatory factors. Currently, the available data suggests that the IM dysregulates the immune response against the tumor in its microenvironment, thus either slowing down or accelerating the efficacy of antitumor therapy. Clinical studies have reported benefits of CRC therapy selected based on IM in improving immune intestinal homeostasis, epithelial barrier functions, and quality of life. Moreover, the specific IM signature may modulate the sensitivity to chemo- and/or radiotherapy, as well as the prognosis in patients with colorectal cancer. In this article, we presented the general challenges of the CRC therapy based on IM data in combination with immunotherapy, and described the future prospects of this approach.
Текст научной статьи Роль кишечной микробиоты при иммунотерапии рака толстой кишки
Колоректальный рак (КРР) является одной из ведущих причин смертности от онкологических заболеваний в мире [1]. В зависимости от этиологии и патогенеза, КРР классифицируется на спорадический, наследственный и воспалительный. Считается, что в рамках многоэтапного процесса, включающего генетические факторы, образ жизни, особенности пищевого поведения и хроническое воспаление путем накопления множества генетических и эпигенетических изменений происходит возникновение и прогрессирование рака толстой кишки [2–6]. Хроническое воспаление является важным фактором риска, ряд исследований показали, что 5-летний накопительный риск возникновения воспалительного заболевания кишечника (ВЗК) составляет 33%–54% [7–9]. Кроме того, у пациентов с ВЗК, риск развития КРР в 2–4 раза выше, чем у пациентов без воспалительного агента [10].
Хотя множественные мутации необходимы как для воспалительного, так и для спорадического КРР, рак толстой кишки сопряженный с воспалительным процессом может ускорить гиперметилирование, хромосомную и микросателлитную нестабильность и изменить постоянство и разнообразие КМ [11,12,13]. В свою очередь, дисбактериоз кишечных бактерий связан с потерей функции эпителиального барьера, патогенезом ВЗК и колит-ассоциированным КРР [14,15]. Таким образом, эффективная профилактика и лечение ВЗК может значительно снизить частоту колит-ассоциированного КРР.
Из этого следует, что КМ человека, будучи гетерогенной и содержащей по меньшей мере 1000 разновидностей микроорганизмов, необходима для переваривания пищи, контроля кишечного эпителиального гомеостаза и здоровья человека в целом [14].
Симбиотические бактерии слизистой оболочки кишечника повышают его гомеостаз и ингибируют кишечную колонизацию патогенами. При нарушении баланса КM нарушается и слизистая оболочка кишечника, и функция врожденного иммунитета, а количество относительных патогенных факторов растет, что вызывает хронические воспалительные и инфекционные заболевания.
Первоначально, при исследовании мышей с карциномами толстой кишки была выявлена важная модуляционная роль КМ в канцерогенезе, индуцированном воспалением, в замедлении роста опухоли [16,17]. Например, есть данные что у особей, имевших признаки дезорганизации КМ, вызванной приемом антибиотиков, наблюдалось снижение выработки фактора некроза опухоли и, как следствие, снижение эффективности противоопухолевой терапии [18]. Кроме этого, многочисленные виды бактерий, такие как Bacteroides fragilis, Fusobacterium nucleatum и Peptostreptococcus stomatis имеют высокую степень сродства с КРР [19]. Между тем, некоторые кишечные бактерии могут влиять на чувствительность к химио- и лучевой терапии у пациентов КРР [20,21]. Исследования Vétizou M, PittJM et al. показали, что КM также играет роль в ответе, опосредованном действием ингибиторов иммунных контрольных точек [22,23].
В настоящее время хирургия, химиотерапия и лучевая терапия улучшили выживаемость больных раком толстой кишки и, в определенной степени, снизили частоту рецидивов [24]. При этом 35% пациентов КРР диагностируются уже с имеющимися отдаленными метастазами, что является основной причиной смертности [25]. Очевидно, что профилактика и ранняя диагностика имеют большое значение для прогноза и лечения больных раком толстой кишки.
Таким образом, КМ является обнадеживающим звеном в клиническом применении и играет многообещающую роль в терапии КРР.
Из анализа данных следует, что изменение КМ при химио- и иммуннотерапии рака толстой кишки, определение профиля безопасности и эффективности является крайне необходимым.
Взаимосвязь КМ и рака толстой кишки
Анализ ряда зарубежных исследований показал, что КМ способствует развитию КРР посредством изменения бактерио-кишечных биопленок, гомеостаза микроокружения и иммунной реакции. Бактериальные биопленки состоят из высокоорганизованных многоорганических структур в микробных сообществах слизистой оболочки в кишечнике человека и выступают в качестве первой линии защиты от инвазивных индуцированных микробами воспалительных реакций и продуцирования генотоксических соединений, полученных из бактерий [26–28]. Недавно было выявлено, что уменьшение биологического разнообразия и богатства микробной составляющей с увеличением видов Fusobacterium, Peptostreptococcus, Bacteroides, Eubacterium, Proteobacteria, Prevotella и Clostridium наблюдалось у пациентов с КРР [29,30–34]. У них F. nucleatum способствует переходу предракового состояния в онкологический процесс, а также прогрессированию рака толстой кишки [35,36]. Дальнейшие исследования показали, что F. nucleatum содействует пролиферации клеток КРР посредством индуцирования воспалительной реакции и увеличения количества миелоидных иммунных клеток, подавляющих пролиферацию Т-клеток, тем самым индуцируя Т-клеточный апоптоз [37,38]. Следовательно, КМ является важным аспектом в инициировании и прогрессировании рака толстой кишки.
Взаимосвязь КМ и иммуноонкологии
На сегодняшний день известно, что иммунотерапия способна стимулировать и мобилизировать иммунную систему человека, а также усиливать противоопухолевый иммунный ответ в опухолевом микроокружении, что в конечном итоге вызывает апоптоз раковых клеток и ингибирует рост опухоли. В то же время, отрицательная регуляция опухолевого иммунного ответа является наиболее важной причиной ускользания от противоопухолевой иммуно- терапии, преимущественно за счет рекрутирования или индуцирования воспалительных клеток, включая регуляторные Т- клетки (Treg), миелоидные супрессорные клетки (MDSC) и макрофаги [39]. Кроме этого, цитотоксический Т- лимфоцит-ассоциированный антиген 4 (CTLA-4), рецептор программируемой смерти (PD-1) и лиганд программируемой смерти (PD–L1), иммуносупрессивные цитокины противоопухолевого иммунного ответа, такие как TGF-β, интерлейкин-10 (IL-10), IL-17 и IL-6, также участвуют в отрицательной регуляции опухолевого иммунитета [40–43]. Несколько работ показали, что КМ имеет большую прогностическую ценность при ответе на лечение ингибиторами контрольных точек (check-points inhibitors-ICK) [44–46].
Имеются данные, что Treg могут ингибировать функцию CD4+ T-клеток, CD8+ Т-клеток, дендритных клеток (DC) и естественных киллеров (NK), способствуя формированию иммуносупрессивного микроокружения опухоли [47]. По мнению Geis AL, Fan H et al., амплификация Treg тесно связана с КМ, Enterotoxigenic Bacteroides fragilis (ETBF) — ассоциированным хроническим воспалением и канцерогенезом толстой кишки у мышей посредством усиления пролиферации Treg и продуцирования IL-17 [48]. Соответственно, дисбаланс КМ может приводить к усилению регуляции IL-17 в эпителиальных клетках кишечника посредством TLR/MyD88-зависимого сигнального пути в процессе канцерогенеза [49]. Эти исследования подтверждают идею о том, что подавление отрицательных регуляторно-ассоциированных воспалительных клеток и иммуносупрессивных цитокинов путем улучшения КM могут быть перспективной стратегией противоопухолевой иммунотерапии (рис. 1).
Анализ ряда зарубежных исследований показал, что клетки CD4+ CD25+ Treg могут ингибировать иммунную функцию активированных Т-клеток, усиливая экспрессию CTLA-4, которая объединяет B7 на антиген-презентативной клетке (АПК) и затем антагонизирует активацию передачи CD28/B7 и противоопухолевого иммунного ответа [50,51]. По данным исследований на опухолевых образцах животных и людей, одновременное применение антител против CTLA-4 и Bacteroides благоприятно влияет на иммунный ответ DC и Т-клеток с противоопухолевыми свойствами [52]. Кроме того, было обнаружено, что количественное увеличение Bacteroides коррелирует с резистентностью к развитию CTLA-4 –индуцированного колита [53].
Помимо CTLA-4, другой ингибитор иммунной контрольной точки — PD-1 — может связываться с рецептором на поверхности опухолевой клетки PD–L1, сенсибилизировать PD-1/PD–L1 сигнальный путь и стимулировать опухолевые клетки к ускользанию от иммунного надзора и апоптозу [54]. В недавнем исследовании Gopalakrishnan V. et al. утверждалось, что бактерии Clostridiales обнаружены в микробиоме кишечника больных меланомой, подвергавшихся анти-PD-1 иммунотерапии, где основополагающим механизмом может быть усиление пролиферации эффекторных Т-клеток и ингибирование уровня иммуносупрес-
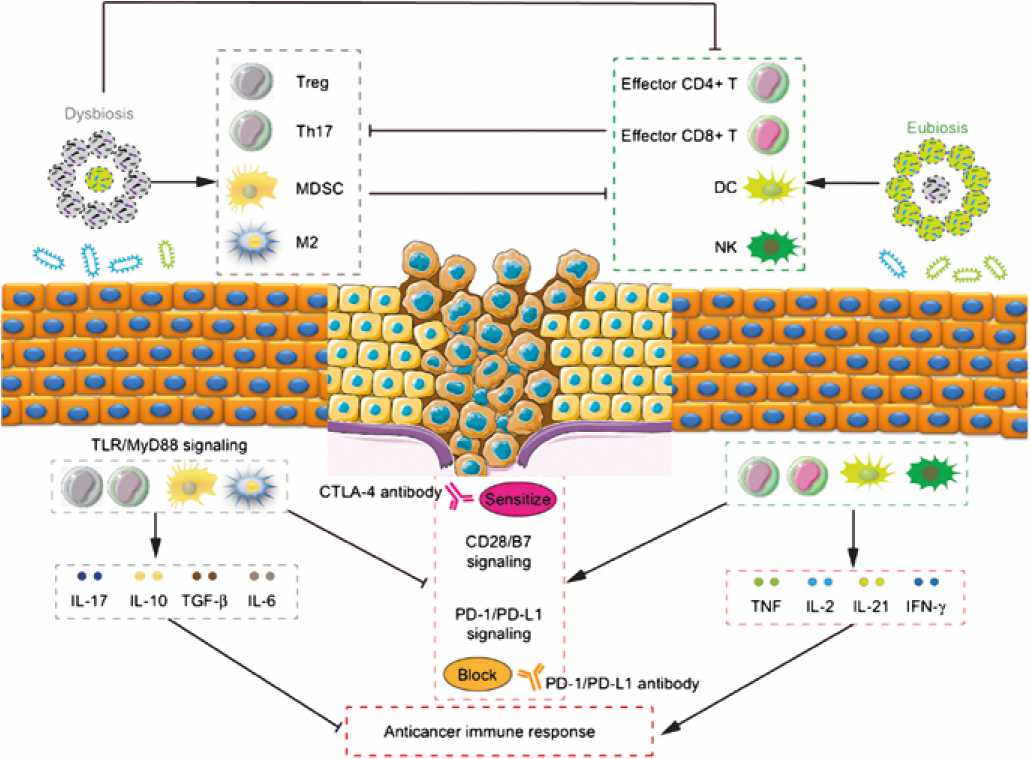
Рисунок 1. Роли кишечной микробиоты в регуляции опухолевого иммунитета.
Список литературы Роль кишечной микробиоты при иммунотерапии рака толстой кишки
- Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, JemalA. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65 (2):87-108.
- Fearon ER. Molecular genetics of colorectal cancer. Ann Rev Pathol. 2011;6 (1):479-507.
- Jafri SH, Mills G. Lifestyle modification in colorectal cancer patients: an integrative oncology approach. Future Oncol. 2013;9 (2):207-218.
- Okugawa Y, Grady WM, Goel A. Epigenetic alterations in colorectal cancer: emerging biomarkers. Gastroenterology. 2015;149 (5):1204-1225.
- Lasry A, Zinger A, Ben-Neriah Y. Inflammatory networks underlying colorectal cancer. Nat Immunol. 2016;17 (3):230-240.
- O'Keefe SJ. Diet, microorganisms and their metabolites, and colon cancer. Nat Rev Gastroenterol Hepatol. 2016;13 (12):691-706.
- Jess T, Simonsen J, Jørgensen KT, Pedersen BV, Nielsen NM, Frisch M. Decreasing risk of colorectal cancer in patients with inflammatory bowel disease over 30 years. Gastroenterology. 2012;143 (2):375-381.
- Jess T, Horváth-Puhó E, Fallingborg J, Rasmussen HH, Jacobsen BA. Cancer risk in inflammatory bowel disease according to patient phenotype and treatment: a Danish population-based cohort study. Am J Gastroenterol. 2013;108 (12):1869-1876.
- Johnson CM, Wei C, Ensor JE, et al. Meta-analyses of colorectal cancer risk factors. Cancer Causes Control. 2013;24 (6):1207-1222.
- Farraye FA, Odze RD, Eaden J, Itzkowitz SH. AGA medical position statement on the diagnosis and management of colorectal neoplasia in inflammatory bowel disease. Gastroenterology. 2010;138 (2):738-745.
- Lasry A, Zinger A, Ben-Neriah Y. Inflammatory networks underlying colorectal cancer. Nat Immunol. 2016;17 (3):230-240.
- Johnson CM, Wei C, Ensor JE, et al. Meta-analyses of colorectal cancer risk factors. Cancer Causes Control. 2013;24 (6):1207-1222.
- Kostic AD, Chun E, Meyerson M, Garrett WS. Microbes and inflamma-tion in colorectal cancer. Cancer Immunol Res. 2013;1 (3):150-157.
- Bruner SD, Jobin C. Intestinal microbiota in inflammatory bowel disease and carcinogenesis: implication for therapeutics. Clin Pharmacol Ther. 2016;99 (6):585-587.
- Ijssennagger N, van der Meer R, van Mil SWC. Sulfide as a mucus barrier-breaker in inflammatory bowel disease? Trends Mol Med. 2016; 22 (3):190-199.
- Uronis JM, Mühlbauer M, Herfarth HH, Rubinas TC, Jones GS, Jobin C. Modulation of the intestinal microbiota alters colitis-associated colorectal cancer susceptibility. PLoS One. 2009;4 (6):e6026.
- Li Y, Kundu P, Seow SW, et al. Gut microbiota accelerate tumor growth via c-jun and STAT3 phosphorylation in APC Min/+ mice. Carcino-genesis. 2012;33 (6):1231-1238.
- Васильев А.Н. Трансплантация фекальной микробиоты: возможные терапевтические подходы и вопросы правового регулирования/А.Н. Васильев, Д.В. Горячев, Е.В. Гавришина и др.//Биопрепараты. Рецензируемый научно-практический журнал. -2015. -No 2 (54) -С. 15-23.
- Yu J, Feng Q, Wong SH, et al. Metagenomic analysis of faecal micro-biome as a tool towards targeted non-invasive biomarkers for colorectal cancer. Gut. 2017;66 (1):70-78.
- Crawford PA, Gordon JI. From the cover: microbial regulation of intestinal radiosensitivity. Proc Natl Acad Sci. 2005;102 (37): 13254-13259.
- Yu T, Guo F, Yu Y, et al. Fusobacterium nucleatum promotes chemoresistance to colorectal cancer by modulating autophagy. Cell. 2017; 170 (3):548-563.
- Vétizou M, Pitt JM, Daillère R, et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science. 2015; 350 (6264):1079-1084.
- Routy B, Le Chatelier E, Derosa L, et al. Gut microbiome influences efficacy of PD-1 -based immunotherapy against epithelial tumors. Science. 2018;359 (6371):91-97.
- Ahmed S, Johnson K, Ahmed O, Iqbal N. Advances in the management of colorectal cancer: from biology to treatment. Int J Colorectal Dis. 2014;29 (9):1031-1042.
- Field K, Lipton L. Metastatic colorectal cancer-past, progress and future. World J Gastroenterol. 2007;13 (28):3806-3815.
- Dejea CM, Wick EC, Hechenbleikner EM, et al. Microbiota organization is a distinct feature of proximal colorectal cancers. Proc Natl Acad Sci. 2014;111 (51):18321-18326.
- Li S, Konstantinov SR, Smits R, Peppelenbosch MP. Bacterial biofilms in colorectal cancer initiation and progression. Trends Mol Med. 2017; 23 (1):18-30.
- Sicard J-F, Le Bihan G, Vogeleer P, Jacques M, Harel J. Interactions of intestinal bacteria with components of the intestinal mucus. Front Cell Infect Microbiol. 2017;7:387.
- Yu J, Feng Q, Wong SH, et al. Metagenomic analysis of faecal micro-biome as a tool towards targeted non-invasive biomarkers for colorectal cancer. Gut. 2017;66 (1):70-78.
- Nakatsu G, Li X, Zhou H, et al. Gut mucosal microbiome across stages of colorectal carcinogenesis. Nat Commun. 2015;6 (1):8727.
- Huipeng W, Lifeng G, Chuang G, Jiaying Z, Yuankun C. The differences in colonic mucosal microbiota between normal individual and colon cancer patients by polymerase chain reaction-denaturing gradient gel electrophoresis. J Clin Gastroenterol. 2014;48 (2):138-144.
- Warren RL, Freeman DJ, Pleasance S, et al. Co-occurrence of anaerobic bacteria in colorectal carcinomas. Microbiome. 2013;1 (1):16.
- Wu N, Yang X, Zhang R, et al. Dysbiosis signature of fecal microbiota in colorectal cancer patients. Microb Ecol. 2013;66 (2):462-470.
- Kahouli I, Tomaro-Duchesneau C, Prakash S. Probiotics in colorectal cancer (CRC) with emphasis on mechanisms of action and current perspectives. J Med Microbiol. 2013;62 (Pt_8):1107-1123.
- Ito M, Kanno S, Nosho K, et al. Association of Fusobacterium nucleatum with clinical and molecular features in colorectal serrated pathway. Int J Cancer. 2015;137 (6):1258-1268.
- Park CH, Han DS, Oh Y-H, Lee A-Reum, Lee Y-Ra, Eun CS. Role of Fusobacteria in the serrated pathway of colorectal carcinogenesis. Sci Rep. 2016;6 (1):25271.
- Ye X, Wang R, Bhattacharya R, et al. Fusobacterium nucleatum sub-species Animalis influences proinflammatory cytokine expression and monocyte activation in human colorectal tumors. Cancer Prev Res. 2017;10 (7):398-409.
- Nosho K, Sukawa Y, Adachi Y, et al. Association of Fusobacterium nucleatum with immunity and molecular alterations in colorectal cancer. World J Gastroenterol. 2016;22 (2):557-566.
- de Vries NL, Swets M, Vahrmeijer AL, Hokland M, Kuppen PJ. The immunogenicity of colorectal cancer in relation to tumor development and treatment. Int J Mol Sci. 2016;17 (7):E1030.
- Nishino M, Ramaiya NH, Hatabu H, Hodi FS. Monitoring immune-checkpoint blockade: response evaluation and biomarker development. Nat Rev Clin Oncol. 2017;14 (11):655-668.
- Stewart CA, Metheny H, Iida N, et al. Interferon-dependent IL-10 production by Tregs limits tumor Th17 inflammation. J Clin Invest. 2013;123 (11):4859-4874.
- Pang Y, Gara SK, Achyut BR, et al. TGF-β signaling in myeloid cells is required for tumor metastasis. Cancer Discov. 2013;3 (8):936-951.
- Qian X, Chen H, Wu X, Hu L, Huang Q, Jin Y. Interleukin-17 acts as double-edged sword in anti-tumor immunity and tumorigenesis. Cytokine. 2017;89:34-44.
- Pitt JM, Vétizou M, Waldschmitt N, et al. Fine-tuning cancer immunotherapy: optimizing the gut microbiome. Cancer Res. 2016; 76 (16):4602-4607.
- West NR, Powrie F. Immunotherapy not working? Check your micro-biota. Cancer Cell. 2015;28 (6):687-689.
- Snyder A, Pamer E, Wolchok J. Immunotherapy. Could microbial therapy boost cancer immunotherapy? Science. 2015;350 (6264):1031-1032.
- Tanaka A, Sakaguchi S. Regulatory T cells in cancer immunotherapy. Cell Res. 2017;27 (1):109-118.
- Geis AL, Fan H, Wu X, et al. Regulatory T-cell response to entero-toxigenic Bacteroides fragilis colonization triggers IL17-dependent colon carcinogenesis. Cancer Discov. 2015;5 (10):1098-1109.
- Viaud S, Saccheri F, Mignot G, et al. The intestinal microbiota modulates the anticancer immune effects of cyclophosphamide. Science. 2013;342 (6161):971-976.
- Baecher-Allan C, Viglietta V, Hafler DA. Human CD4+CD25+ regulatory T cells. Semin Immunol. 2004;16 (2):89-98.
- Egen JG, Kuhns MS, Allison JP. CTLA-4: new insights into its biological function and use in tumor immunotherapy. Nat Immunol. 2002; 3 (7):611-618.
- Vétizou M, Pitt JM, Daillère R, et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science. 2015; 350 (6264):1079-1084.
- Dubin K, Callahan MK, Ren B, et al. Intestinal microbiome analyses identify melanoma patients at risk for checkpointblockade-induced colitis. Nat Commun. 2016;7:10391.
- Nishino M, Ramaiya NH, Hatabu H, Hodi FS. Monitoring immune-checkpoint blockade: response evaluation and biomarker development. Nat Rev Clin Oncol. 2017;14 (11):655-668.
- Gopalakrishnan V, Spencer CN, Nezi L, et al. Gut microbiome modulates response to anti-PD-1 immunotherapy in melanoma patients. Science. 2018;359 (6371):97-103.
- Sivan A, Corrales L, Hubert N, et al. Commensal Bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science. 2015;350 (6264):1084-1089.
- Matson V, Fessler J, Bao R, et al. The commensal microbiome is associated with anti-PD-1 efficacy in metastatic melanoma patients. Science. 2018;359 (6371):104-108.
- Jin P, Wang K, Huang C, Nice EC. Mining the fecal proteome: from biomarkers to personalised medicine. Expert Rev Proteomics. 2017; 14 (5):445-459.
- Ai L, Tian H, Chen Z, Chen H, Xu J, Fang JY. Systematic evaluation of supervised classifiers for fecal microbiotabased prediction of colorectal cancer. Oncotarget. 2017;8 (6):9546-9556.
- Zhang MM, Cheng JQ, Xia L, et al. Monitoring intestinal microbiota profile: a promising method for the ultraearly detection of colorectal cancer. Med Hypotheses. 2011;76 (5):670-672.
- Shah MS, Desantis TZ, Weinmaier T, et al. Leveraging sequence-based faecal microbial community survey data to identify a composite biomarker for colorectal cancer. Gut. 2018;67 (5):882-891.
- Enq RR. How's your microbiota? Let's check your urine. Blood. 2015; 126 (14):1641-1642.
- Ferreira RM, Pereira-Marques J, Pinto-Ribeiro I, et al. Gastric microbial community profiling reveals a dysbiotic cancer-associated microbiota. Gut. 2018;67 (2):226-236.
- Panteli JT, Forkus BA, van Dessel N, Forbes NS. Genetically modified bacteria as a tool to detect microscopic solid tumor masses with triggered release of a recombinant biomarker. Integr Biol. 2015;7 (4):423-434.
- Gardlik R, Fruehauf JH. Bacterial vectors and delivery systems in cancer therapy. IDrugs. 010;13(10):701-706.


