Single vein resection in surgery for pancreatic ductal carcinoma as a criterion of resectability
Автор: Chichevatov D.A., Kalentjev V.V., Glukhov A.E.
Журнал: Сибирский онкологический журнал @siboncoj
Рубрика: Клинические исследования
Статья в выпуске: 4 т.21, 2022 года.
Бесплатный доступ
Background. pancreatic ductal carcinoma (pdc) with involvement of the superior mesenteric vein (smV) or/and portal vein (pV) remains a discussible subject. We have evaluated vein invasion as a criterion of borderline resectability and long-term outcome. material and methods. in our center, 68 patients underwent either 65 standard pancreatoduodenal resections or 3 pancreatoduodenectomies for pdc. Resection of smV/pV was performed in 18 cases (26.5 %). three patients received neoadjuvant chemotherapy (Nact), and adjuvant chemotherapy (act) was assigned to 37 patients (54.4 %). Results. morbidity (42.0 vs 50.0 %, р=0.590) and mortality rates (4.0 vs 16.7 %, р=0.111) had no significant differences in groups of standard and angioplasty operations respectively. act was completed in 10 (16.7 %) patients only. there was true vein invasion in 12 of 18 patients with vein resection. рN+ (р=0.012) and angioplasty by itself (р
Carcinoma, pancreatic ductal, portal vein, angioplasty
Короткий адрес: https://sciup.org/140295748
IDR: 140295748 | УДК: 616.37-006.6-089 | DOI: 10.21294/1814-4861-2022-21-4-25-31
Изолированная резекция вены в хирургическом лечении протоковой карциномы поджелудочной железы как фактор резектабельности
Лечение протоковой карциномы поджелудочной железы (ПКПЖ) с вовлечением верхней брыжеечной и/или воротной вены остается предметом дискуссий. Мы оценили венозную инвазию как критерий пограничной резектабельности и отдаленного прогноза заболевания. Материал и методы. В нашем центре 68 пациентов оперированы в объеме стандартной панкреатодуоденальной резекции (n=65) или панкреатодуоденэктомии (n=3) по поводу ПКПЖ. В 18 (26,5 %) наблюдениях выполнена резек-ция верхней брыжеечной и/или воротной вены. Неоадъювантную химиотерапию (НАХТ) получили 3 пациента, адъювантная химиотерапия (АХТ) начата у 37 (54,4 %) пациентов. Результаты. Частота послеоперационных осложнений в группе стандартных операций и ангиопластических операций (42,0 против 50,0 %, р=0,590), частота летальных исходов (4 против 16,7 %, р=0,111) значимо не отлича-лись. АХТ закончена полностью у 10 (16,7 %) пациентов. Истинная инвазия опухоли отмечена у 12 из 18 пациентов с резекцией вены. Независимыми предикторами общей выживаемости (ОВ) были рN+ (р=0,012) и наличие ангиопластики (р=0,000). У пациентов с резекцией вены медиана ОВ составила 9,4 мес. В группе стандартных ПДР медиана ОВ была 26,9 мес (р=0,000). У пациентов с резекцией вены медиана ОВ при законченной химиотерапии составила 17,7 мес, без химиотерапии - 8,9 мес (р=0,439). Заключение. ПДР с резекцией вены и отсутствием или незавершенной химиотерапией может рассматриваться как неоправданная операция. Данную операцию целесообразно выполнять после эффективной НАХТ.
Текст научной статьи Single vein resection in surgery for pancreatic ductal carcinoma as a criterion of resectability
So far treatment of pancreatic ductal carcinoma (PDC) has been discussible [1, 2]. While the best results have been shown in patients undergoing surgery with chemotherapy, the choice of the adjuvant (ACT) or neoadjuvant (NACT) regime remains unclear. Many investigators demonstrate advantages of NACT such as marginal but significant improvement of the overall survival and increased rate of R0, N0 resections [3–5]. On the other hand, according to several reports these benefits have been achieved in “per protocol” analyses only, whereas there were no significant differences in the frame of the “intention-to-treat” concept. It could be because up to 27 % of patients did not enter the surgical protocol due to either tumor progression against the background of chemotherapy or intolerable adverse effects [1, 6, 7]. Nevertheless, the latter evidence cannot be a reason for “up-front” surgery since as few as 55–57 % of patients who underwent the “surgery first” protocol obtain complete ACT due to early progression, postsurgical complications, and deaths [8, 9]. Besides, current meta-analyses focused on this problem do not avoid biases most of the time and their authors accentuate high difficulty in retrieving data that strictly meet inclusion criteria [1, 10, 11].
Assessment of the tumor extent is another important aspect that affects the choice of treatment. While either primary resectable or locally advanced tumors are believed to be quite determined groups, the so-called borderline resectable malignancies cannot be defined properly. Anderson's criteria proposed in 2009 are based on radiological findings only. Some authors mention the uncertainty of borderline resectability which can depend not only on anatomical but also on subjective and technical predictors (surgical modality, experience, equipment, etc.) [12, 13]. All this provides “migration” of potentially resectable tumors to the group of unresectable locally advanced ones, thus surgical exploration as a final diagnostic procedure becomes warranted [13, 14].
Nowadays “up-front” surgery is regarded as an acceptable procedure in case of isolated involvement of the vein [2, 15]. Necessity and availability of arterial resection are expected to be utterly rare in pancreatic surgery [16]. On the other hand, we believe that isolated vein involvement should not be a contraindica- tion to surgery at any stage of treatment. The present paper is a retrospective analysis of immediate and follow-up results of PDC treatment which required vein resection on account of surgically proven vein involvement.
Material and Methods
In our department, 120 patients underwent pancreatoduodenal resection (PDR) or pancreatoduodenectomy (PDE) within a period from 2014 to 2021. According to inclusion criteria we picked up patients with PDC only who underwent PDR or PDE with/with-out resection of the superior mesenteric or/and portal vein. All arterial resections were excluded. Finally, the total investigated group included 68 patients, 34 males and 34 females (50.0 %). The age range was 43–79 years with an average of 63.3 ± 8.1 years. pT1 tumor was observed in 5 patients (7.4 %), рТ2 – in 2 (2.9 %) and рТ3 – in 61 (89.7 %). Regional lymph node metastases (pN+) were found in 29 cases (42.6 %), all of them were N1. There was the following stage distribution: IA – 5 (7.4 %), IB – 1 (1.5 %), IIA – 33 (48.5 %), IIB – 23 (33.8 %), III – 1 (1.5 %), IV – 5 (7.4 %). Stage IV was determined by paraaortic metastases in 3 patients and by the solitary liver metastasis – in 2 ones.
PDR and PDE were performed as a standard procedure with lymph node dissection including groups 3b, 4d, 5, 6, 8, 9, 12V, 12P, 12D, 13, 14V, 14A, 17 according to the modified classification of Japanese Research Society for Gastric Cancer (1962). Paraaortic lymph nodes (group 16) were excised in case of proven metastases only (3 patients). The restoring stage of the surgery was executed by pancreatojejunostomy, hepaticojejunostomy, and gastrojejunostomy in this sequence using the single intestinal loop.
In the case of the dilated pancreatic duct, we performed two-layer anastomosis by sewing the duct up separately with interrupted sutures (polypropylene 6–0 or 7–0). Such a technique was employed in 55 patients (80.9 %). In the case of the non-dilated pancreatic duct, we performed sleeve-like pancreatojejunostomy without a separate connection of the duct (14.7 % of patients). There were three PDEs. Hepaticojejunostomy was accomplished by running suture (polypropylene 5–0 or 6–0). One metastasectomy and one S2,3-liver resection were concomitants in two patients with stage IV, respectively.
Patients with superior mesenteric (SMV) or/and portal (PV) vein involvement were considered to be candidates for vein resection if it did not require multiple vascular anastomoses of SMV confluents. PV resection was performable most of the time. Lateral SMV/PV resection with closing the defect by running suture (polypropylene 6–0) was made in 4 patients. Sleeve SMV/PV resection (Fig. 1) with direct suturing of vein cuffs was made in 7 patients and with prosthesis replacement (Fig. 2) in 6 cases. Closure of the vein defect with a patch (Fig. 3) was performed in one patient. There were 18 (26.5 %) vascular resections altogether. In all cases, we used polytetrafluoroethylene prostheses and the running suture (polypropylene 6–0).
The performance status of patients was assessed according to the ECOG scale. Postoperative pancreatic fistulas were defined in accordance with International Study Group for Pancreatic Surgery (ISGPS) classification, 2016. Data were processed and calculated with the statistical package SPSS Statistics v. 17.0. Descriptive statistics, cross-tabulation, two-tailed Fisher's exact test, Cox regression analysis, Kaplan-Meier analysis were employed.
Results
Postsurgical complications were observed in 30 (44.1 %) patients. There were 19 (27.9 %) compli- cations in grade II, 5 (7.4 %) – in grade IIIb, 1 IV (1.5 %) – in grade, and 5 – in grade V (7.4 %) according to the Clavien–Dindo classification (Table 1). Causes of death were as diverse as the myocardial infarction (1), the pancreonecrosis (2), the pancreatic fistula type С (1), intestinal anastomosis leakage with the consequent peritonitis (1).
Postsurgical complications were registered in 21 (42.0 %) patients after standard PDR and in 9 of 18 (50.0 %) cases after angioplasty PDR/PDE. This difference was not significant (two-tailed Fisher's exact test, p=0.590). The post-surgical mortality rate was 4.0 % (2 of 50) in the standard group and 16.7 % (3 of 18) in the angioplasty group. This difference was not significant either (two-tailed Fisher's exact test, p=0.111). It should be noted we have not observed specific vascular complications which ended up with death.
There were only 12 of 18 cases of true vein invasion according to pathologists' reports. 40 (58.8 %) patients received chemotherapy. NACT was assigned in 3 (4.4 %) patients and ACT in 37 (54.4 %) ones. The following regimes were applied as preoperative or postoperative modalities: FOLFIRINOX, GEMOX, GEM, GEMCAP, XELOX, FOLFOX, FOLFIRI, capecitabine, and 5-fluorouracil as monotherapy.
Twenty-three patients did not receive ACT for the following reasons: early tumor progression (4), post-surgical complications (5), PS ECOG≥3 (4), patient's refusal (10). All patients completed NACT. Only 10 (16.7 %) of the remaining 60 patients received appro-
Note: * – in accordance with the current International Study Group for Pancreatic Surgery (ISGPS) classification. DGE, delayed gastric emptying;
** – the patient has undergone PDE with right-sided hemicolectomy.
Примечание: * – в соответствии с текущей классификацией International Study Group for Pancreatic Surgery (ISGPS); ** – пациенту выполнена ПДЭ с правосторонней гемиколэктомией.
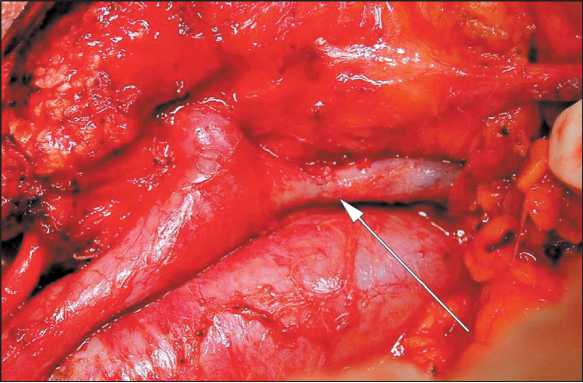
Fig. 1. Sleeve SMV/PV resection with direct suturing of vein cuffs. The arrow points at the vascular anastomosis
Рис. 1. Циркулярная резекция ВБВ/ВВ с формированием прямого анастомоза. Анастомоз указан стрелкой
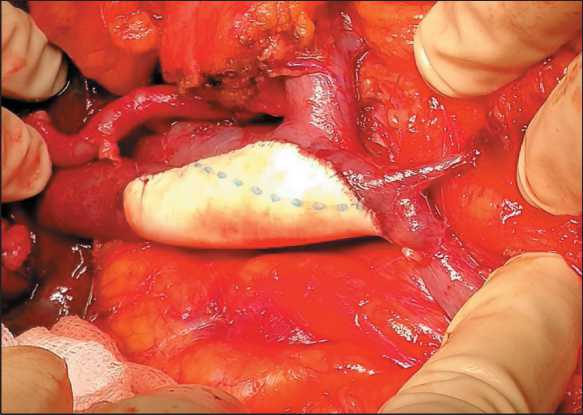
Figure 3. SMV/PV defect has been closed with a patch Рис. 3. Замещение дефекта ВБВ/ВВ заплатой priate adjuvant treatment. ACT was terminated in 27 patients due to developed intolerable toxicity (adverse effects) or worsening of PS ECOG ≥ 3.
Long-term results were assessed as standard modalities of disease-free survival (DFS) and overall survival (OS). Among three patients who underwent surgery after NACT, two died in 5.2 and 17.9 months, respectively, due to tumor progression. The third one has been alive for 16.8 months without recurrence of the tumor.
We have assessed independent predictors of DFS and OS based on the Cox regression analysis. The following variables were regarded as potential predictors: 1) age, 2) gender, 3) TNM-T, 4) pN+ (yes, no), 5) true invasion of the vein (yes, no), 6) complete NACT or ≥ 6 courses of ACT (yes, no), 7) postsurgi-cal complications (yes, no), 8) tumor stage, 9) vein resection (yes, no).
The tumor stage IB (HR=80.5; 95 % CI, 2.9– 2207.4; р=0.009), stage IIA (HR=10.5; 95 % CI, 1.3–87.3; p=0.030), stage IIB (HR=18.8; 95 % CI, 2.1–164.1; p=0.008), stage IV (HR=125.7; 95 %
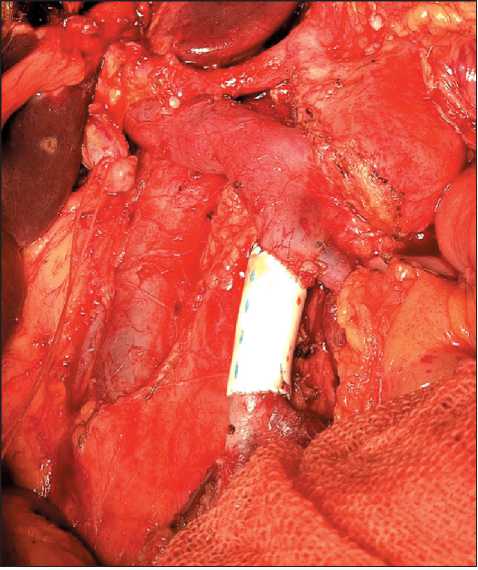
Fig. 2. SMV resection with prosthesis replacement Рис. 2. Резекция ВБВ с протезированием
CI, 10.1–1571.7; p<0.001) and the presence of post-surgical complications (HR=0.3; 95 % CI, 0.1–0.8; р=0.010) were significant predictors of DF (the model χ2=29.4; р<0.001). рN+ (HR=2.4; 95 % CI, 1.2–4.9; р=0.012) and vein resection performed (HR=6.9; 95 % CI, 2.7–17.4; p<0.001) were the only predictors of OS (the model χ2=24.5; р<0.001).
In the group of vein resection, the median of OS was 9.4 mo (95 % CI, 5.3–13.5). Even 2-year survival has not been obtained and the maximum follow-up period was 17.9 mo. In the group of standard operations, the median of OS was 26.9 mo (95 % CI, 17.2–36.6), the 5-year survival was 29.4 ± 10.1 %. This difference was significant (Log Rank test, χ2=18.7, р<0,001). Plots of OS are given in Fig. 4.
Although NACT/ACT has not been revealed as an independent predictor of survival we evaluated OS of patients who underwent vein resection combined or not combined with complete NACT or 6 or more courses of ACT (NACT/6АСT). We limited ACT with 6 or more courses because our patients were assigned different regimes of chemotherapy due to diverse objectives, thus stratification of patients in accordance with particular anticancer agents was thought to be inappropriate statistically. Among patients without vein resection, the median of OS was 26.9 vs 29.9 mo in the group of NACT/6ACT and no chemotherapy, respectively (Log Rank test, χ2=0,36, р=0.546). Among patients with vein resection, analogous figures were 17.7 vs. 8.9 mo, respectively (Log Rank test, χ2=0.60, р=0.439) (Fig. 5).
Discussion
In the present investigation, we attempted to answer the question of whether vein invasion and, consequent
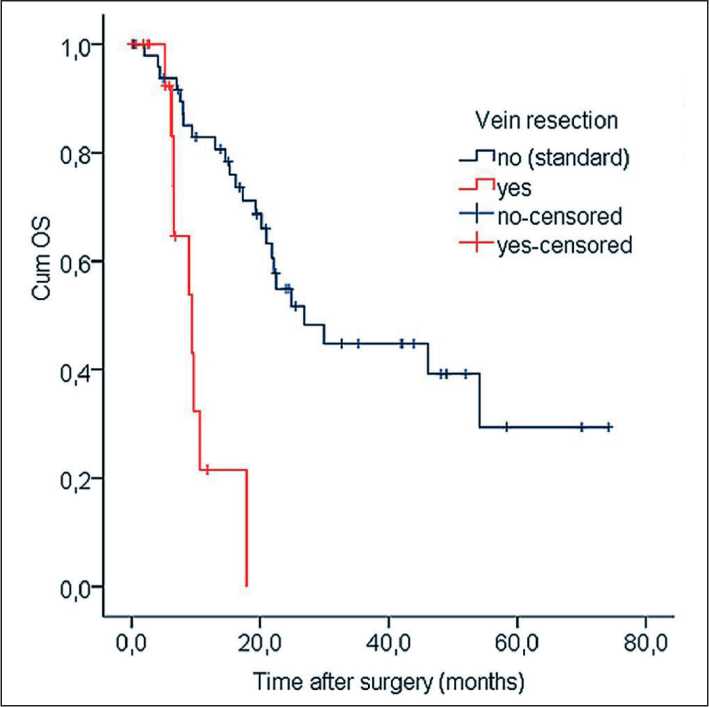
Fig. 4. Kaplan–Meier curves of OS depended on vein resection Рис. 4. Показатели общей выживаемости по Kaplan–Meier в зависимости от наличия резекции вены
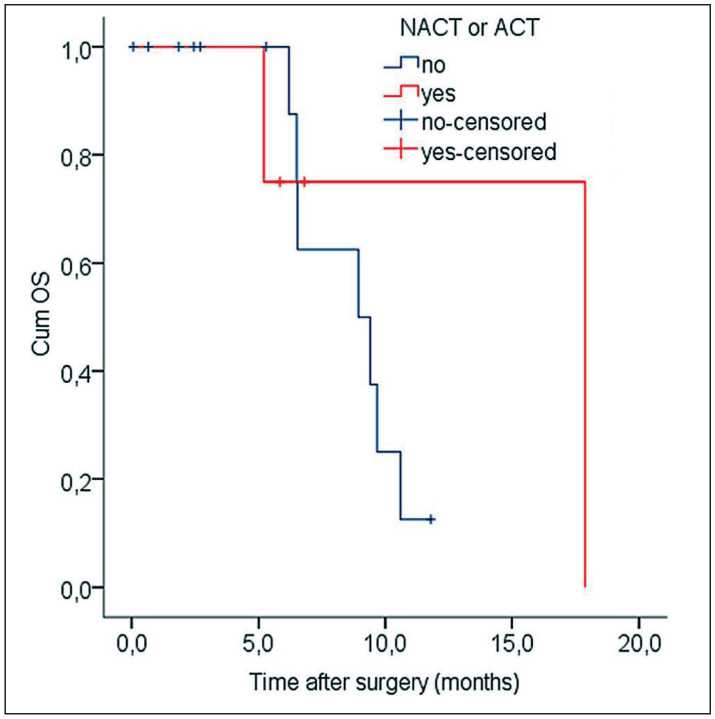
Fig. 5. Kaplan–Meier curves of OS depending on NACT/6ACT in the angioplasty group
Рис. 5. Показатели общей выживаемости по Kaplan–Meier в зависимости от НАХТ/6АХТ в группе ангиопластик
or mere, vein resection can be regarded as a reliable and essential criterion of borderline resectability. On the one hand, some authors [2, 3, 11, 15] believe that vein resection has become a safe standard procedure and survival in this group is similar to that in the group of standard operations. On the other hand, after successful vein resection, the tumor is considered to be resectable as well as it becomes unresectable locally advanced in the case of failed angioplasty. The problem of borderline resectability is of interest nowadays [17]. Nevertheless, vein invasion can affect survival, this fact has not been reflected in the 8th edition of the TNM classification yet. Thereafter we analyzed only complete vein resections taking into account two aspects: 1) immediate results (safety of the procedure) and 2) survival in the group of accomplished angioplasty PDR/PDE compared to standard ones. In addition, we intended to make our investigation as close to “intention-to-treat” principles as possible but in the group of patients who can undergo complete surgery combined with NACT and/or ACT.
In a technical sense our conception of resectability, variants of angioplasty are determined perfectly well and in general, coincide with current classifications proposed [18].
While the relatively high rate of post-surgical complications is evident in our series, vein resection did not increase this rate significantly. Specific complications consequent to angioplasty were not registered at all. Besides, 2/3 of all complications were controlled and not life-threatening according to the Clavien–Dindo classification. M. Park et al. report analogous results with the rate of morbidity from 28.6 % to 38.6 %
[19]. Postsurgical mortality rate was 7.4 % but non-significantly higher (p=0.111) in the group of vein resections. All three cases of death in this group have not been connected with vein resection, in our opinion. Therefore, we believe that vein resection may be regarded as a safe enough procedure which does not worsen a surgical outcome.
Some authors [20] consider true vein invasion to be a negative predictor of long-term outcomes. We have found out true vein invasion only in 12 of 18 patients with vein resection. In our series, true vein invasion did not affect DFS whereas the procedure of vein resection by itself appeared as a significant but unfavorable predictor of OS. Thereafter vein resection may be approved as a technical demand only. According to current meta-analyses, NACT increases OS significantly but marginally in patients with PDC [4]. This was a reason for recommending it as a preferable treatment in many clinical guidelines. Unfortunately, in our series only 4.4 % of patients received NACT. Such a small amount of cases can be explained by the insufficiency of preoperative biopsies and our reserved attitude due to some controversial reports [7, 10]. M. Assifi et al. [6], S. Gillen et al. [7] informed on lack of significant efficacy of NACT, intolerable adverse effects, and failed tumor response, hence they lost over 20 % of patients.
In our investigation ACT was assigned to only 54.4 % of patients, which cannot be a satisfactory indicator. Approximately 65 % or more of patients end up ACT according to current reports. Besides, the majority of patients in our series could not undergo a complete course of ACT for reasons beyond control. Only 10 (16.7 %) patients have received six or more courses of ACT. We consider the treatment protocol to be complete in 13 of 68 (19.1 %) patients. Such outcome cannot be satisfactory and reveals no advantages of ACT.
A tumor stage, postsurgical complications, pN+, vein resection were independent significant predictors of DFS and OS, respectively, in the frame of the Cox regression analysis. Conclusions concerning a stage, postsurgical complications, and regional metastases are trivial and do not need to be discussed, in our opinion.
The negative influence of vein resection and angioplasty on OS may be explained by a higher rate of occult metastases consequent to true vein invasion and bulky tumor extent [11]. This suspicion is thought to be a reason for providing further prospective investigations to change TNM-T similar to arterial invasion. We heard controversial reports on how vein involvement affects OS [15]. Although vein resection seems to be as safe as the standard PDR procedure, it should not be excluded from the criteria of borderline resectability because it discriminates PDC into different groups with diverse survival. Nevertheless, OS in the group of vein resections is expected to be much higher if NACN/ ACT is complete. Although there was no significant difference of OS curvatures in our series (р=0.439), in the vein resection group with/without chemotherapy the effect size in patients with complete NACT/ACT was approximately twice as high (the median of OS
Список литературы Single vein resection in surgery for pancreatic ductal carcinoma as a criterion of resectability
- Versteijne E., Suker M., Groothuis K., Akkermans-Vogelaar J.M., Besselink M.G., Bonsing B.A., Buijsen J., Busch O.R., Creemers G.M., van Dam R.M., Eskens F.A.L.M., Festen S., de Groot J.W.B., Groot Koer-kamp B., de Hingh I.H., Homs M.Y.V., van Hooft J.E., Kerver E.D., Luelmo S.A. C., Neelis K.J., Nuyttens J., Paardekooper GM.R.M, Patijn G.A., van der Sangen M.J.C., de Vos-Geelen J., Wilmink J.W., ZwindermanA.H., Punt C.J., van Eijck C.H., van Tienhoven G.; Dutch Pancreatic Cancer Group. Preoperative Chemoradiotherapy Versus Immediate Surgery for Resectable and Borderline Resectable Pancreatic Cancer: Results of the Dutch Randomized Phase III PREOPANC Trial. J Clin Oncol. 2020; 38(16): 1763-73. doi: 10.1200/JCO.19.02274.
- Klaiber U., Hackert T. Conversion Surgery for Pancreatic Cancer-The Impact of Neoadjuvant Treatment. Front Oncol. 2020; 9: 1501. doi: 10.3389/fonc.2019.01501.
- Tang K., Lu W., Qin W., Wu Y. Neoadjuvant therapy for patients with borderline resectable pancreatic cancer: A systematic review and meta-analysis of response and resection percentages. Pancreatology. 2016; 16(1): 28-37. doi: 10.1016/j.pan.2015.11.007.
- Oba A., Ho F., Bao Q.R., Al-Musawi M.H., Schulick R.D., Del Chia-ro M. Neoadjuvant Treatment in Pancreatic Cancer. Front Oncol. 2020; 10: 245. doi: 10.3389/fonc.2020.00245.
- Schorn S., Demir I.E., Reyes C.M., Saricaoglu C., Samm N., Schirren R., TieftrunkE., HartmannD., Friess H., Ceyhan G.O. The impact of neoadjuvant therapy on the histopathological features of pancreatic ductal adenocarcinoma - A systematic review and meta-analysis. Cancer Treat Rev. 2017; 55: 96-106. doi: 10.1016/j.ctrv.2017.03.003.
- Assifi M.M., Lu X., Eibl G., Reber H.A., Li G., Hines O.J. Neoadjuvant therapy in pancreatic adenocarcinoma: a meta-analysis of phase II trials. Surgery. 2011; 150(3): 466-73. doi: 10.1016/j.surg.2011.07.006.
- Gillen S., Schuster T., Meyer Zum Büschenfelde C., Friess H., Kleeff J. Preoperative/neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of response and resection percentages. PLoS Med. 2010; 7(4). doi: 10.1371/journal.pmed.1000267.
- CasolinoR., Braconi C., Malleo G., Paiella S., Bassi C., MilellaM., Dreyer S.B., Froeling F.E.M., Chang D.K., Biankin A.V., Golan T. Reshaping preoperative treatment of pancreatic cancer in the era of precision medicine. Ann Oncol. 2021; 32(2): 183-96. doi: 10.1016/j. annonc.2020.11.013.
- Dumont R., PuleoF., Collignon J., MeurisseN., Chavez M., SeidelL., Gast P., Polus M., Loly C., Delvenne P., Meunier P., Hustinx R., Deroover A., Detry O., Louis E., Martinive P., Van Daele D. A single center experience in resectable pancreatic ductal adenocarcinoma: the limitations of the surgery-first approach. Critical review of the literature and proposals for practice update. Acta Gastroenterol Belg. 2017; 80(4): 451-61. Erratum in: Acta Gastroenterol Belg. 2018; 81(2): 358.
- Rahman S.H., UrquhartR., MolinariM. Neoadjuvant therapy for resectable pancreatic cancer. World J Gastrointest Oncol. 2017; 9(12): 457-65. doi: 10.4251/wjgo.v9.i12.457.
- Delpero J.R., Sauvanet A. Vascular Resection for Pancreatic Cancer: 2019 French Recommendations Based on a Literature Review From 2008 to 6-2019. Front Oncol. 2020; 10: 40. doi: 10.3389/fonc.2020.00040.
- Ferrone C.R., Marchegiani G., Hong T.S., RyanD.P., Deshpande V., McDonnell E.I., Sabbatino F., Santos D.D., Allen J.N., Blaszkowsky L.S., Clark J.W., Faris J.E., Goyal L., Kwak E.L., Murphy J.E., Ting D.T., Wo J.Y., Zhu A.X., Warshaw A.L., Lillemoe K.D., Fernández-del Castillo C. Radiological and surgical implications of neoadjuvant treatment with FOLFIRINOX for locally advanced and borderline resectable pancreatic cancer. Ann Surg. 2015; 261(1): 12-7. doi: 10.1097/ SLA.0000000000000867.
- Wittel U.A., Lubgan D., GhadimiM., Belyaev O., Uhl W., Bechs-tein W.O., Grützmann R., Hohenberger W.M., Schmid A., Jacobasch L., CronerR.S., Reinacher-Schick A., Hopt U.T., PirklA., OettleH., FietkauR., GolcherH. Consensus in determining the resectability of locally progressed pancreatic ductal adenocarcinoma - results of the Conko-007 multicenter trial. BMC Cancer. 2019; 19(1): 979. doi: 10.1186/s12885-019-6148-5.
- Katz M.H., Pisters P.W., Evans D.B., Sun C.C., Lee J.E., Fleming J.B., Vauthey J.N., Abdalla E.K., Crane C.H., Wolff R.A., Varad-hachary G.R., Hwang R.F. Borderline resectable pancreatic cancer: the importance ofthis emerging stage of disease. J Am Coll Surg. 2008; 206(5): 833-46. doi: 10.1016/j.jamcollsurg.2007.12.020.
- WangX., Demir I.E., Schorn S., Jäger C., Scheufele F., Friess H., Ceyhan G.O. Venous resection during pancreatectomy for pancreatic cancer: a systematic review. Transl Gastroenterol Hepatol. 2019; 4: 46. doi: 10.21037/tgh.2019.06.01.
- Bockhorn M., Uzunoglu F.G., Adham M., Imrie C., Milicevic M., SandbergA.A., AsbunH.J., Bassi C., BüchlerM., CharnleyRM., ConlonK., CruzL.F., Dervenis C., Fingerhutt A., FriessH., GoumaD.J., Hartwig W., LillemoeK.D., MontorsiM., Neoptolemos J.P., Shrikhande S. V., TakaoriK., Traverso W., Vashist Y.K., Vollmer C., Yeo C.J., Izbicki J.R.; International Study Group of Pancreatic Surgery. Borderline resectable pancreatic cancer: a consensus statement by the International Study Group of Pancreatic Surgery (ISGPS). Surgery. 2014; 155(6): 977-88. doi: 10.1016/j. surg.2014.02.001.
- Lopez N.E., Prendergast C., Lowy A.M. Borderline resectable pancreatic cancer: definitions and management. World J Gastroenterol. 2014; 20(31): 10740-51. doi: 10.3748/wjg.v20.i31.10740.
- Nakao A. Selection and outcome of portal vein resection in pancreatic cancer. Cancers (Basel). 2010; 2(4): 1990-2000. doi: 10.3390/ cancers2041990.
- Park M.Y., Lee W, Kwon J., Song K.B., Hwang D.W., Lee J.H., Kim S.C. Comparison of perioperative outcomes in pancreatic head cancer patients following either a laparoscopic or open pancreaticoduodenectomy with a superior mesenteric artery first approach. Ann Hepatobiliary Pancreat Surg. 2021; 25(3): 358-65. doi: 10.14701/ahbps.2021.25.3.358.
- Kishi Y., Nara S., Esaki M., Hiraoka N., Shimada K. Feasibility of resecting the portal vein only when necessary during pancreatoduodenectomy for pancreatic cancer. BJS Open. 2019; 3(3): 327-35. doi: 10.1002/bjs5.50130.


