Specific features of light energy conversion in biological photo-elements
Автор: Bukhanov E.R., Loginov Y.Y., Poznahareva O.A., Volochaev M.N., Shabanov V.P.
Журнал: Siberian Aerospace Journal @vestnik-sibsau-en
Рубрика: Technological processes and material science
Статья в выпуске: 1 vol.27, 2026 года.
Бесплатный доступ
The study aims to analyze the influence of structural changes in thylakoid membranes, induced by foliar fertilization, on energy transfer in the photosynthetic system using fluorescence spectroscopy methods. Control and experimental plant groups were established for the research. Structural analysis was performed using transmission electron microscopy (TEM), which provided images of ultra-thin leaf sections followed by quantitative assessment of the morphometric parameters of chloroplast grana. Micrograph processing and statistical analysis were carried out using artificial intelligence-based software (GRANA). The functional state of the photosystems was evaluated based on chlorophyll fluorescence spectra were obtained using a spectrofluorometer in a 90-degree geometry mode upon excitation with light at a wavelength of 425 nm. Results. It was found that the application of foliar fertilization leads to a statistically significant structural reorganization of the thylakoid membranes. The experimental samples showed a reduction in the median grana area by ~ 38 % and their perimeter by ~ 16 %, indicating denser and more ordered packing. Simultaneously, fluorescence spectroscopy revealed a narrowing of the peak half-width corresponding to photosystem II by 1.8 nm (8%) in the treated plants. This change indicates a reduction in the energetic heterogeneity of the antenna complexes and an increase in the efficiency of excitation energy transfer. Conclusion. Based on the comprehensive studies, it was concluded that the primary mechanism of action of foliar fertilizers is the optimization of the thylakoid membrane nanostructure, which, within the concept of photonic crystals, creates more favorable conditions for light energy conversion. An increase in the regularity of the ultrastructural organization was demonstrated. The obtained results confirm the potential of using fluorescence spectroscopy as a rapid, non-invasive method for diagnosing the physiological state of plants in closed space systems.
Solar energy conversion, biological photonic crystals, thylakoid membranes, fluorescence spectroscopy, transmission electron microscopy, photosynthesis, energy transfer
Короткий адрес: https://sciup.org/148333277
IDR: 148333277 | УДК: 535.37 | DOI: 10.31772/2712-8970-2026-27-1-158-171
Текст научной статьи Specific features of light energy conversion in biological photo-elements
Efficient conversion of solar energy is a fundamental challenge for supporting closed space systems and satellite communications. From a biospheric energy perspective, photosynthesis is a key cosmic process, as it alone accumulates energy from an external source (sunlight), thereby increasing free energy and decreasing entropy in the system. Thus, it serves as the foundation for life, generating oxygen and creating all the necessary organic substances.
In recent years, research has been actively conducted on the conversion of light energy into bioelectricity using photosynthetic bioelectrochemical systems [1–3].
While in photovoltaics, engineers optimize the efficiency by improving the morphology and materials of photoactive layers, in agronomy, a similar problem is solved by increasing the efficiency of the plant photosynthetic apparatus. Photosynthesis, at its core, is the process of converting light energy into chemical energy, with the chloroplast acting as a highly organized biological “solar battery” [4].
In a review by A. G. Yakovlev and V. A. Shuvalov [5], it is reported that the physical stage of photosynthesis demonstrates a phenomenally high efficiency of light energy conversion. The quantum efficiency of primary charge separation in reaction centers approaches 100%, meaning that virtually every absorbed photon results in the transfer of one electron. This is achieved through exceptionally rapid and directed multi-step electron transfer along the pigment chain (P* → Bₐ → Hₐ → Qₐ), where each subsequent step is accompanied by a loss of energy but an increase in the stability of the separated charges. The high rate of forward reactions, significantly exceeding the rates of reverse processes and energy losses, is ensured by the optimal spatial arrangement of pigments and the influence of the protein environment. Such perfect organization makes the primary act of photosynthesis a unique example of highly efficient energy conversion in living nature.
In the context of commercialization, where every resource, including water and nutrient substrates, is strictly limited, highly efficient and resource-saving agricultural technologies are particularly relevant. One such technology is foliar feeding, which enables the targeted delivery of macro- and micronutrients directly to metabolically active plant tissues, bypassing the soil [6; 7]. This approach not only ensures rapid nutrient absorption but also minimizes inevitable losses, making it economically and environmentally preferable for use in agro-industrial complexes.
In this study, we consider foliar feeding not simply as a cultural practice, but as a tool for targeted “engineering” of chloroplast ultrastructure. We hypothesize that certain fertilizer formulations may act similarly to dopants in semiconductors [8] or optical coatings in solar cells [9], minimizing energy loss through structural defects and increasing energy efficiency.
The key mechanism determining the effectiveness of this technology is its impact on the ultrastruc-tural organization of the photosynthetic apparatus, particularly the thylakoid membranes of chloroplasts. Optimization of the spatial arrangement of thylakoids, which act as natural optical transducers, directly determines the efficiency of energy transfer [10]. For non-invasive diagnostics of the functional state of photosystems, this study utilizes fluorescence spectroscopy, which enables highly accurate monitoring of changes in the primary processes of photosynthesis induced by foliar feeding, with structural rearrangements observable by electron microscopy.
The aim of this study was to investigate the impact of structural changes in thylakoid membranes induced by foliar feeding on energy transfer in the photosynthetic apparatus. For this purpose, changes in the organization of thylakoid membranes were studied by transmission electron microscopy and the functional state of photosystems was assessed using fluorescence spectroscopy. The established correlations between structure, spectral characteristics, and productivity allow us to propose a new approach to optimizing “biological solar cells”.
Materials and methods of research
Cultivation of plants. At present, rapeseed is one of the leading agricultural crops in the world, the area of cultivation of winter and spring rapeseed is extremely wide and covers almost all natural and climatic zones of agriculture. The scope of its application is also extremely diverse. Spring rapeseed seeds contain 40–48 % edible oil and 18–22 % high-quality feed protein, used to produce oil for food and technical purposes.
Field research was organized on the territory of the experimental farm “Mikhailovskoye”. Studies on the effect of foliar top dressing were carried out on spring rapeseed, the “Siberian variety”. Two experimental sites were identified for comparative analysis. At the control site, only basic chemical treatment was used, aimed at protecting against weeds and pathogens: no huminatrin treatment, the seeds were treated with Tabu Neo, 6 l/t; treatment with herbicides in the stalking phase (before budding); treatment with insecticide in the phase of the beginning of flowering. On the other site, in addition to the indicated protective measures, leaf nutrition of plants was carried out with a specialized composition: pickling of Tabu Neo seeds, 6 l/t + Huminatrin, 0.5 l/t; treatment with herbicides in the stalking phase (before budding); treatment with insecticide in the early flowering phase. As a result, the yield was 20.8 c/ha for the control samples and 21.9 c/ha for the experimental ones. The raw weight of plants increased from 7.98 to 11.72 g, and the weight of leaves from 4.6 to 6.30 g. At the same time, the concentration of photosynthetic pigments in the leaves decreased from 4.2 to 4.156 mg/g of crude weight.
Electron microscopic analysis. To study the ultrastructural features, the flag leaves of plants at the earing stage were selected. The primary fixation of the samples was carried out using a 2.5 % solution of glutaraldehyde on a cacodylate buffer (pH 7.4). Subsequent fixation was performed in a 1 % OsO4 solution. Dehydration was carried out in alcohols of increasing concentration and acetone, followed by polymerization in resin (EMbed 812 Kit, Merck). Ultrathin sections, 20–30 nm thick, were obtained using a Leica EM UC7 ultramicrotome. Additional contrast of the slices was performed using uranyl acetate and lead citrate.
Images of the chloroplast structure on ultrathin sections were obtained using a Hitachi HT7700 transmission electron microscope. The analysis was performed at an accelerating voltage of 90 kV in the light field mode. To obtain representative data on the spatial organization of the thylakoid membranes, serial imaging was performed at various sample orientations.
Fluorescence spectroscopy. The fluorescence spectra were recorded using a CM-2203 spectro-fluorimeter (manufactured by CJSC Spectroscopy, Optics and Lasers – Modern Developments (SOLAR), Republic of Belarus). An FX-4401 pulsed xenon lamp was used as an excitation source. The detection was carried out by a radiation receiver – FEU R928. The samples for the study were prepared by cutting out segments of a sheet plate 20–25 mm long, followed by mounting on a special holder. The measurements were carried out in a 90-degree geometry with a sample tilt angle of 60° to the detection direction.
Fluorescence was excited at a wavelength of 425 nm (slit width of 20 nm), spectra were recorded in the range of 600–800 nm (slit width of 5 nm). The scanning was performed in 1 nm increments with the maximum sensitivity of the detection system.
For the control and experimental samples, 100 images were selected for each, where at least one facet was clearly observed. Additional image contrast was also performed using software.
Research results
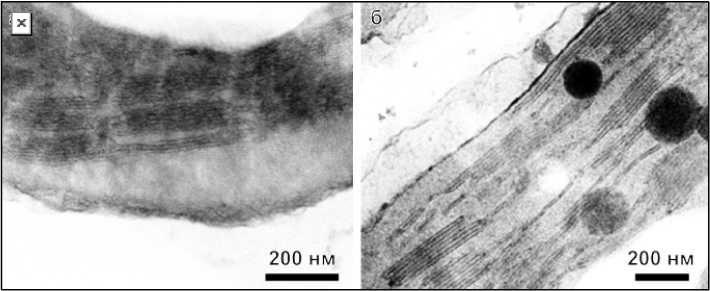
Figure 1 shows the average micrographs of the structure of chloroplasts of rapeseed leaves obtained using transmission electron microscopy for the control (Fig. 1, а ) and experimental (Fig. 1, б ) samples. From a visual comparison of the figures, it can be seen that in the first case, the grana have a more deformed appearance. This concerns both the tightness of the thylakoid membranes to each other and the geometric distortions.
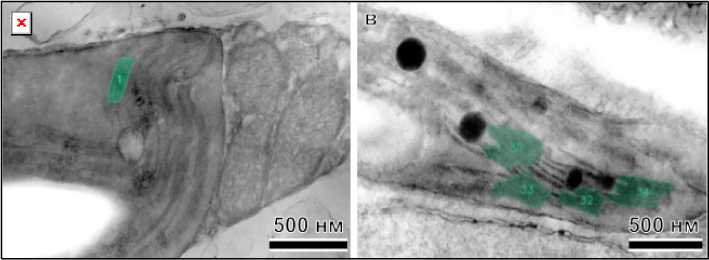
Measuring the parameters of thylakoid membranes by the manual method is a rather complicated and time-consuming process, for example, as it was implemented in paper [10]. The main advantage of this approach is that having a complete data set allows you to calculate any dependencies and correlations. In 2025, a tool called GRANA was presented and described in detail in [11] to accelerate the analysis of nanomorphology of chloroplast grana based on artificial intelligence. This method allows you to quickly evaluate hundreds of images of the structure, but its main disadvantages are the rather poor recognition of grana with a geometry other than a straight line and the limited choice of output parameters. The results of processing the average GRANA images for both types of samples are shown in Fig. 2.
б
Рис. 1. Снимки структуры хлоропластов листьев рапса, не получавших ( а ) и получавших ( б ) добавку
Fig. 1. Photographs of the chloroplast structure of rapeseed leaves that did not receive ( a ) and received ( б ) the additive
а б
а б
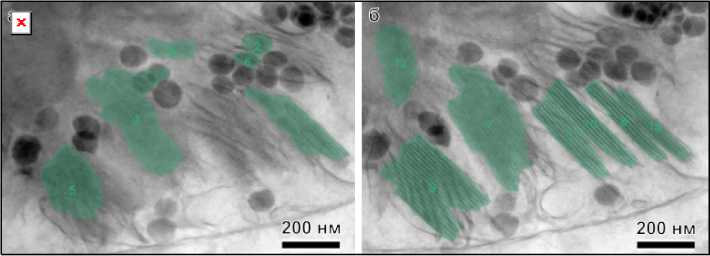
Рис. 2. Снимки структуры хлоропластов листьев рапса после обработки с помощью GRANA, не получавшие ( а , б ) и получавшие добавку ( в , г )
Fig. 2. Images of the chloroplast structure of rapeseed leaves after treatment with GRANA, not treated ( a , б ) and treated with the additive ( в , г )
A comparison of images between control and experimental samples clearly demonstrates that the program is better at recognizing grana in the latter case. By visually counting all clearly visible grana in the analyzed images and all those identified using artificial intelligence, the first numerical characteristic can be determined for comparing samples. The proposed automatic grana recognition program was able to identify 39 % of visible grana for control samples versus 61% for experimental samples. This can be explained by the fact that in the latter case, grana have a more typical shape and more clearly defined thylakoid membranes. Furthermore, the lower contrast in the control samples may indicate an insufficient supply of components necessary for healthy plant life. Based on scanning images of chloroplast structure, the data presented in Table 1 were obtained.
Table 1
Results of chloroplast structure image analysis for control and experimental samples
Результаты анализа изображений структуры хлоропластов для контрольных и опытных образцов
|
Control |
Experiment |
|
|
Area (nm2) |
40622 ± 37344 |
25054 ± 13495 |
|
Perimeter (nm) |
980 ± 423 |
824 ± 270 |
|
Diameter (nm) |
314 ± 77 |
283.89 ± 82.35 |
|
Height (nm) |
101.63 ± 28.40 |
92.16 ± 30.29 |
|
Number of thylakoids |
– |
7.50 ± 2.12 |
|
Grana repetition parameter (nm) |
– |
15.01 ± 1.15 |
The data presented in Table 1 show the median value for each parameter, as well as the standard deviation around the median. Dashes in the last two lines for the control samples indicate that the program was unable to determine these parameters. A comparison of the results reveals that the chloroplast structure characteristics of the experimental samples were significantly reduced (by approximately 16% for key parameters). This may be directly related to the denser and more linear ordering of the thylakoid membranes in the grana. It has been noted in [10; 12; 13] that dense grana packing can promote more efficient electron transfer between photosystems and, consequently, more efficient photosynthesis. This effect can be explained by the fact that foliar fertilizers favorably influence the achievement of the most efficient photosynthesis process and can directly influence the internal architecture of the leaves.
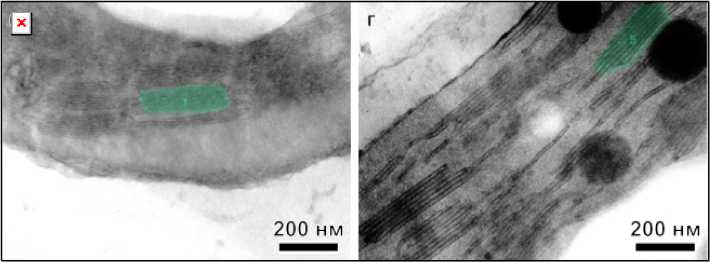
In [14; 15], it is noted that the observation angle relative to the sample directly affects the structural results. For example, if the sample is tilted so that a grana is not perpendicular to the electron beam, the final dimensions and appearance of the thylakoid membranes may be distorted. Figure 3 shows images of the chloroplast structure obtained using transmission electron microscopy after processing with GRANA software. In Figure 3 a , the sample was at an angle of 60° relative to the electron beam, while in Figure 3 б , it was at an angle of 90°. A comparison of the figures shows that tilting the sample by 30° from the plane perpendicular to the grana results in a blurring of the grana. The artificial intelligence-based program partially identified three grana, while with the beam orthogonal to the grana, all grana are clearly visible in the image.
б
Рис. 3. Снимки структуры хлоропластов листьев высших растений после обработки с помощью GRANA при угле наблюдения к гранам 30о ( а ) и под прямым углом ( б )
Fig. 3. Images of the chloroplast structure of higher plant leaves after processing using GRANA at an observation angle of 30° to the grana ( a ) and at a right angle ( б )
Based on the scanning results of the chloroplast structure images from Fig. 3, the data presented in Table 2 were obtained. The scanning results show that, when the edges were distorted, the artificial intelligence was unable to determine even half of the parameters, while when evaluating the image at a right angle to the edge, the program was able to determine all available characteristics.
Table 2
Results of image analysis of chloroplast structure for 30° and a right angle Результаты анализа изображений структуры хлоропластов для 30о и прямого угла
|
Parameter |
Right angle |
|
Area (nm2) |
130221 ± 101300 |
|
Perimeter (nm) |
1928 ± 878 |
|
Diameter (nm) |
650.99 ± 65.71 |
|
Height (nm) |
287.23 ± 134.61 |
|
Number of thylakoids |
11.67 ± 5.07 |
|
Grana repeat parameter (nm) |
24.01 ± 1.02 |
To assess the efficiency of energy transfer through structural modification, it was of interest to measure fluorescence spectra. The fluorimetric results are presented in Figure 4. A comparison of the spectral widths of the fluorescence peaks at half-maximum corresponding to photosystem II (685 nm) reveals a significant difference. The peak width for the control samples was 24.2 nm, while the peak width for the experimental samples was 22.4 nm. Thus, the difference between the spectral widths of the peaks at half-maximum was 1.8 nm in absolute terms or 8 % in relative terms.
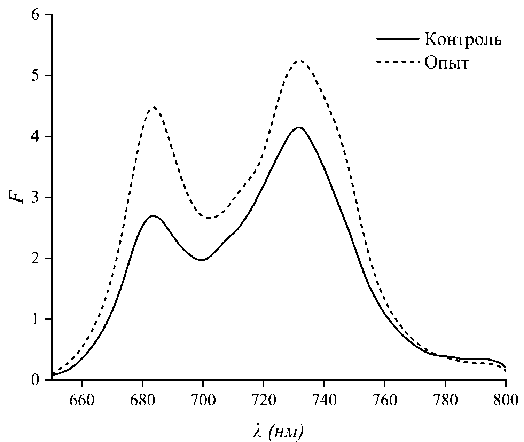
Рис. 4. Спектры флуоресценции листьев рапса, не получавших (контроль) и получавших добавки (опыт)
Fig. 4. Fluorescence spectra of rapeseed leaves that did not receive (control) and received additives (experiment)
Discussion
A key structural consequence of the treatment was an increase in the ordering of thylakoid membranes in the grana of the experimental chloroplasts. The obtained electron microscopic data indicate a decrease in the average grana size and their denser packing. A decrease in the geometric parameters of the grana and an increase in the homogeneity of their size distribution leads to a decrease in the degree of disorder in the system. These structural changes should affect the parameters of PS II. According to current concepts, plant leaves contain two photosystems [16], which are protein complexes containing multiple (antenna) light-harvesting pigments, conductive components for electron transfer, and a reaction center.
The photochemical reaction center includes chlorophyll molecules in plants, as well as chlorophylllike pigments in algae and photosynthetic bacteria, which are oxidized by light and act as primary electron donors.
These special chlorophyll-like pigments with an absorption maximum at 700 nm, along with their light-harvesting antennae, are called the PS I system, while those with an absorption maximum at 680 nm are called the PS II system.
The studied fluorescence spectrum of plant leaves represents a combination of contributions from two photosystems. It is generally accepted that at room temperature, the majority of this fluorescence is emitted by the PS II system. The contribution from PS I at a wavelength of 683 nm is approximately 5%, and at a wavelength of 700–740 nm, 30–35%. Moreover, it has been found [17] that PS I fluorescence depends weakly on the state of the reaction center.
As for the fluorescence spectrum of PS II, everything is simpler here, since a simple technique for chloroplast fractionation has been developed [18]. The extracted PS II (usually called the VVU grana) is considered native, since it contains both the reaction center and the light-harvesting complex (LHC-II).
For the first time [19], the intrinsic fluorescence emission spectrum of PS II was obtained for several concentrations of isolated PS II suspensions. Its dependence on various external conditions was subsequently studied, significantly elucidating many microprocesses occurring in green plant organelles. Unfortunately, it was not possible to achieve quantitative CO2 uptake comparable to that of a green leaf. Therefore, the question of the optimal structural parameters of leaf grana, pigment concen- trations, their proportions, and the regularity of the ultrastructural organization of chloroplasts during photosynthesis remains open.
To address this issue, we proposed a method for studying changes in the above-mentioned parameters for systems that differ in their effectiveness. It is known that foliar feeding can, in some cases, increase crop yields.
In paper [20], numerical simulations of one-dimensional photonic crystals with gradually increasing layer thickness disorder were performed. Figure 5 shows a fragment of these results, showing transmission spectra from 0 to 30 % disorder. In [20], it was noted that the band gap is maintained with random layer thickness variations of up to 40 %. However, it was not noted that the reflection band can shift toward longer wavelengths as the degree of layer disorder decreases. Figure 5 shows a dotted line corresponding to the spectral position of the maximum of the short-wavelength reflection band in the absence of layer distortion. As the degree of order increases, it is evident that the maximum of this band shifts toward lower frequencies or toward longer wavelengths. This effect is consistent with the results presented in Fig. 4. In the fluorescence spectra, in the case of a more ordered structure, an additional shoulder is clearly observed in the long-wavelength region.
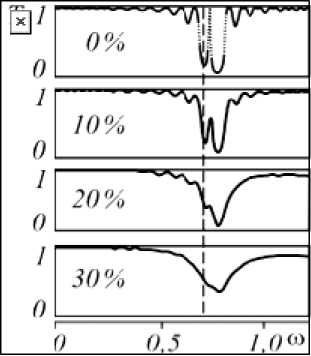
Рис. 5. Усредненные спектры пропускания ФК-решетки с варьируемой толщиной слоёв в зависимости от максимальной амплитуды случайных отклонений линейных размеров от первоначально заданных [20]
Fig. 5. Averaged transmission spectra of a PC grating with variable layer thickness depending on the maximum amplitude of random deviations of linear dimensions from the initially specified ones [20]
A comparative analysis of the control and experimental samples revealed statistically significant changes at both the ultrastructural and optical levels, suggesting a direct impact of the applied agrochemicals on photosynthetic metabolism. Consequently, an increase in the density of photon states at the edges of the stop band is expected in such a structure, theoretically predicting more efficient retention and conversion of light energy [21].
Combining two key observations – an increase in thylakoid structural order and a decrease in energy heterogeneity detected by fluorescence – allows us to construct a consistent cause-and-effect relationship. The application of foliar fertilizers containing macro- and microelements, and likely surfactants, initiates a restructuring of chloroplast ultrastructure. This restructuring is expressed in an optimization of the spatial organization of thylakoid membranes, which, in turn, creates more favorable conditions for excitation energy migration. A reduction in structural disorder directly reduces energy loss due to scattering, which manifests itself as a narrowing of the PS II fluorescence peak.
Thus, the primary mechanism underlying the observed effect is increased efficiency of the primary stages of photosynthesis due to optimization of the light stage. More efficient energy transfer in the PS II antenna complex increases the probability of a light quantum reaching the reaction center. This creates the preconditions for increased efficiency in all subsequent stages of photosynthetic electron transport, including ATP and NADPN synthesis, which ultimately should lead to increased plant biochemical productivity, expressed by increased yield, as demonstrated in similar studies [7].
Conclusion
-
1. Transmission electron microscopy revealed that foliar feeding resulted in a decrease in grana size in the experimental samples compared to the control. This is due to the ordering of the thylakoid membranes in the experimental samples.
-
2. A narrowing of the half-width of the photosystem II fluorescence peak was detected in the experimental sample compared to the control, indicating increased excitation energy migration efficiency.
-
3. It was shown that denser grana packing in chloroplasts contributes to increased excitation energy transfer efficiency between photosystems.
These results not only contribute to a fundamental understanding of the mechanisms by which agrochemicals affect the photosynthetic apparatus but also have practical significance. Fluorescence spectroscopy confirms its effectiveness as a rapid and non-invasive tool for diagnosing plant functional status and predicting the effectiveness of applied technologies, including in closed space systems.
Acknowledgment. The work was carried out within the framework of the scientific topic of the state assignment of the FRC SB RAS.


