Toxicity concerns of semiconducting nanostructures on aquatic plant Hydrilla verticillata
Author: Mishra Priya, Shukla Vineet K., Yadav Raghvendra S., Pandey Avinash C.
Journal: Журнал стресс-физиологии и биохимии @jspb
Article in issue: 2 т.9, 2013.
Free access
In this article, we have examined toxicity of nanostructures such as flower-like ZnO capped with starch, spherical uncapped ZnO and spherical CdS on aquatic plant Hydrilla verticillata, which has not done before. Hydrilla plant was exposed by these nanoparticles at a concentration of 400 mg/L for 7 days and changes in the biochemical parameters such as catalase activity, chlorophyll content and protein content were observed. It was perceived that spherical CdS nanoparticles were more toxic than the corresponding ZnO nanoparticles since there was a decrease in chlorophyll content and increase in catalase activity. This effort upsurge an interest in understanding the hazards of nanomaterials and their risk, which poses an impact on our environment and how they can be monitored via simple biochemical assays on plant systems.
Nanomaterials, hydrilla, catalase, chlorophyll, protein
Short address: https://sciup.org/14323747
IDR: 14323747
Text of the scientific article Toxicity concerns of semiconducting nanostructures on aquatic plant Hydrilla verticillata
Nanoparticle magnetizes a significant awareness because of its wide range of applications in emerging areas of nanoscience and technology (Moore et al., 2006). Size, shape, and surface morphology play pivotal roles in controlling the physical, chemical, optical, and electronic properties of these nanoscopic materials (Handy et al., 2008; Farre et al., 2009). Increasing production and use (cosmetics to medicine) of nano-sized materials have raised concerns about their possible impacts on environmental and human health. Due to their small size, nanoparticles (NPs) display unique properties that are used in nanotechnology (Moore et al., 2006). Nanotechnology is growing very fast, and it has been estimated that will contribute $1 trillion to the U.S. economy by 2015 (Handy et al., 2008) As with any technological advance, the applications of nanotechnology could bring significant benefits but also great risk in terms of the environmental and human health. Nanoparticles are highly reactive due to their extremely small size and high surface to the volume ratio. It has been established that the toxic effect of nanoparticles is due to their ability to catalyze the production of reactive oxygen species (Nel et al., 2006; Xia et al., 2006).
The production, use and disposal of nanoparticles can be an inevitable impact on air, water and soil. Their release into aquatic ecosystem such as lakes, ponds and river increases the risk to aquatic organisms (Moore et al ., 2006; Handy et al ., 2008; Farre et al ., 2009). Risk assessment and nanotoxicological studies have been made on algae (Wang et al ., 2008), bacteria (Jiang et al ., 2009; Johansen et al ., 2008), nematodes and crustaceans (Wang et al ., 2009; Heinlaan et al ., 2008) fish, rats. (Griffitt et al ., 2008; Elgrabli et al ., 2008) and on crop plants (Heinlaan et al ., 2008 Griffitt et al ., 2008; Elgrabli et al ., 2008) however, to our knowledge, nothing is known about the potential effects of NP on aquatic Hydrilla plants. Since they have acquired special characteristics to survive in harsh environments (Lu et al ., 2002), it is hypothesized that plants from aquatic ecosystems will respond differently to NPs than plants in other ecosystems. Importantly, aquatic plants such as Hydrilla verticillata improve water, since they absorb heavy-metal ions such as lead, mercury, uranium, etc. and play an important role in the stability and conservation of other species (Hong et al. , 2005).
However, very few studies have been reported on plants. At low concentrations, nano-sized silicon dioxide and titanium dioxide, increases nitrate reductase activity, stimulates antioxidant system and enhances the ability to absorb water and fertilizer. Lu et al. (2002) reported that the growth of spinach was promoted by addition of nano-sized titanium dioxide by protecting the chloroplasts from ageing during long term illumination (Lu et al., 2002; Zheng et al., 2005). When ryegrass was exposed to 1000 mg/L of nano zinc oxide, it has individual nano zinc oxide particles in endodermal and vascular cells (Lin et al., 2008). Another study found that, when Arabidopsis thaliana was exposed to nZnO, nFe3O4, nSiO2 and nAl2O3 at three different concentrations of 400, 2000 and 4000 mg/L, it was found that nZnO was most phytotoxic and inhibited seed germination at 400mg/L (Lee et al., 2010).
Heavy metals are widely acknowledged to inhibit seed germination, growth and progress of plants and disrupt their biochemical and physiological processes (Prasad et al., 2004). In the present study, Hydrilla verticillata which is an aquatic plant was selected as test plant species. Its exposure to 400 mg/L nZnO, releases 14.6 mg/L of soluble Zn, preventing 94% of the seeds from germinating and completely halted root elongation. In contrast, exposure to equivalent concentrations of soluble Zn (added as ZnCl2 salt, without nanoparticles) resulted in significantly lower toxicity. On the other hand, highest concentration (500 mg/L) of soluble Zn, which is one order of magnitude higher than the amount released by toxic levels of nZnO, inhibited germination (100% of seeds failed to germinate). These data suggest that the phytotoxicity of nanosized metal oxides cannot be explained solely by the dissolved metal species, but the particles themselves also contribute to phytotoxicity (Lee et al., 2010). Therefore, concentration of 400mg/L each of ZnO, CdS and starch capped ZnO nanoparticles was selected to observe its effect on three biochemical parameters viz. protein content, chlorophyll content in leaves and activity of antioxidant enzyme catalase (Figure 1). ZnO nanoparticles are made of Zn, which is essential microelement that is indispensable for normal plant growth at low concentration and is toxic only at high concentration, while CdS nanoparticles are made of Cd, which has no vital function in plant. All the experiments were done in triplicates, and results were presented as mean ±SE (n=3).Statistical significance was accepted when probability of result assuming the null hypothesis (p) is less than 0.05 (level of probability).
MATERIALS AND METHODS
Synthesis of different nanostructures
CdS nanoparticles were synthesized by simple precipitation method. 1M cadmium acetate was dissolved in 50ml double distilled water and then 1M sodium sulphide was added under continuous stirring resulting in yellowish solution. This solution was kept for stirring for 5 h. The solution was centrifuged, washed with distilled water followed by alcohol. The obtained yellow solid product was dried at 60 °C.
Starch capped ZnO nanoparticles were prepared using the sonochemical method. 0.5 g starch was dissolved in 10 ml of double distilled water. 10 ml, 0.1 M aqueous solution of zinc acetate and 10 ml 1M aqueous solution of sodium hydroxide were added to 25 ml alcohol followed by starch solution. The solution containing beaker was then kept in sonication bath (33 kHz, 350 W) at room temperature (Mishra et al ., 2010).
In a typical hydrothermal synthesis, 1 g of zinc acetate dihydrate was put into 110 mL of double distilled water under vigorous stirring. After 10 min stirring, 10 mL of 2M NaOH to maintain pH value at 12. The whole solution was transferred into a stainless steel autoclave, sealed and maintained at the temperature of 110 °C for 4 h. The resulting white solid products were centrifuged, washed and finally dried at 60 °C in air (Yadav et al., 2007).
Characterizations
The crystal structure, shape and size of synthesized starch capped ZnO nanoparticle, ZnO nanoparticle and CdS nanoparticles were characterized by XRD on Rigaku D/max-2200 PC diffractometer operated at 40 kV/20 mA, using Cu– Kα radiation with a wavelength of 1.54 Ǻ in a wide-angle region from 25˚ to 70˚ on a 2θ scale. UV–vis absorption spectroscopy was taken on Perkin Elmer model L35 spectrometer. Transmission Electron Microscopy (TEM) images were taken on Tecnai 30 G2 S-Twin (FEI Company) operated at 300kV accelerating voltage. Scanning electron microscope (SEM) images were taken with a FEI, model Quanta 200 environmental Field Emission SEM microscope.
Plant materials and treatment conditions
Hydrilla verticillata plants were collected from university campus. Plants were washed thoroughly, and a known weight of plants was kept in flasks containing nanoparticles and was allowed to grow for 7 days. After 7 days, four sets of grown Hydrilla plants were made. For performing the experiment one set was kept as control in which no nanoparticles were added and in rest three ZnO, starch capped ZnO and CdS nanoparticles at 400 mg/L concentration was added. Each set of experiment was done in triplicates. Plants were exposed to nanoparticles for 7 days.
Extraction and estimation of total leaf protein
Total leaf protein was extracted by polyvinyl pyridine (PVP) precipitation method. Fresh leaf tissues 0.1 g w homogenized in 10 ml of 50 mM sodium phosphate buffer with 1% PVP and 1mM EDTA in homogenizer. The homogenates were centrifuged at 45000 rpm for 30 min at 4 ˚C. The supernatant was collected and kept at 4 ˚C for protein estimation and enzyme assay. The protein estimation was done by Lowry et al. method (Lowry et al., 1951). Protein in the unknown sample was estimated at 660 nm via bovine serum albumin as standard and expressed per ml.
Chlorophyll measurement
Plants were pulverized in 80% acetone for chlorophyll determination according to Arnon's method using Perkin-Elmer UV-vis spectrophotometer LS 35. The absorbance of the supernatant was observed at 645 and 663 nm, after centrifugation at 45000 rpm for 15 min. The chlorophyll concentrations were calculated using the formula of ( Arnon et al .,1949).
Enzyme assay measurement
Catalase (CAT, EC. 1.11.1.6) activity was measured according to method of (Aebi et al ., 1983). The reaction mixture contains 2.6 ml phosphate buffer, 10 mM H 2 O 2 and 0.5 ml of enzyme extract in a final volume of 3 ml. The absorbance of the reaction concoction was measured at 240 nm compared to the reaction mixture without an enzyme extract. The reaction was initiated by adding 0.5 ml of enzyme extract. One unit of CAT activity (U/mg protein) was defined as the amount of CAT, which decomposed 1μmol/L hydrogen peroxide in 1 min ( Ɛ =39.4 mM-1 cm-1).
RESULTS
Figure 2 shows XRD pattern of ZnO nanoparticle synthesized by hydrothermal, starch capped ZnO nanoparticle synthesized by sonochemical method and CdS nanoparticle synthesized by simple coprecipitation method. Diffraction peaks for ZnO with hexagonal phase can be indexed as (100), (002), (101), (102), (110), (103), (200), (112) and
(201) (JCPDF Card File No. 36145). The XRD pattern of CdS nanoparticles shows three broader peaks corresponding to (111), (220) and (311) planes of cubic CdS phase (JCPDS card No. 42- 1411) (Li et al ., 2003). All the diffraction peaks of CdS were indexed as cubic structure of CdS. Observed XRD broad peaks indicated towards the nanocrystalline nature of CdS particles.
Figure 3 shows TEM and SEM images of synthesized nanoparticles. Figure 3A shows TEM image of starch capped ZnO nanoparticle reveals rods of average length 120 nm and diameter of about 20 nm due to its flower like structure which is clearly observed in its respective SEM image (Figure 3B). These nanorods are made of several spherical nanoparticles of average diameter about 20 nm. Figure 3C shows the average particle size of uncapped ZnO nanoparticle synthesized using the hydrothermal method is about 66 nm. TEM image of CdS nanoparticle reveals spherical morphology with some aggregation, having the particle sizes are between 3 nm to 8 nm (Figure 3D).
Figure 4A shows the effect of different nanoparticles on the content of plant chlorophyll. When Hydrilla plants were exposed to 400 mg/L CdS, ZnO and starch capped ZnO nanoparticles for 7 days, there were 57.7%, 26.82% and 18.18% decrease in chlorophyll content. In all the three cases there has been a significant decrease (p<0.05) in comparison to control. The decrease in the chlorophyll content is either due to the reduced synthesis or accelerated degradation of the chlorophyll pigment. Cd is an effective competitor for essential metal co-factors participating in the chlorophyll biosynthesis (Toppi et al., 1999). The decrease in chlorophyll content is due to interference of Cd+2 at the sulfhydryl site of all enzymes involved in the chlorophyll biosynthesis. It is understood that during photosynthesis, protochlorophyllide reductase enzyme binds NADH, which contains sulfhydryl groups (Mohan et al., 2006). The Cd+2 interfere with sulphydryl groups of enzyme involved in biosysnthesis of chlorophyll. This causes decrease in the chlorophyll content of Hydrilla plant via CdS nanoparticles. Since, Zn is an essential co-factor of chlorophyll synthesizing d-aminolevulinic acid dehydratase, so both ZnO nanoparticles and starch capped ZnO nanoparticles have not caused much decrease in the chlorophyll amount in comparison to CdS, as some Zn ions are released in the solution form of nanoparticles. The current trend of bioassay is to conduct acute lethal toxicity up to 4 days and sub-lethal chronic toxicity for 7 days or still longer (APHA.: Standard methods for the examination of water and wastewater 15th,ed. Washington, DC, APHA, NY. 1995; Michael et al., 2005). Krupa et al. (Krupa et al., 1996) suggested that the determination of total chlorophyll is a reliable marker of cadmium toxicity in higher plants. In our approach, we are getting 57.7%, decrease in chlorophyll content, when treated with 400 mg/L of CdS for 7 days. This shows that CdS is more toxic than ZnO, and starch capped ZnO nanoparticles.
The effect of different nanostructures on the activity of catalase of Hydrilla plant after 7 days treatment is observed and shown in Figure 4B. On repeating the experiment in a triplet, we reported that the catalase activity increases significantly in plants treated with ZnO nanoparticles, whereas in plants treated with starch capped ZnO and CdS nanoparticles the increase in activity is not significant (p<0.05) in comparison to the control plants. Catalase is a major primary antioxidant defense component that primarily catalyses the decomposition of potentially harmful oxidizing agent such as H2O2 into H2O and O2. When plants are exposed to stress off any kind reactive oxygen species (ROS) such as superoxide radical or hydroxyl radical, hydrogen peroxide is produced. The increased activity indicates towards increased production of secondary metabolite hydrogen peroxide, which potency enhances the production of antioxidant enzyme catalase. As a result, it breaks down hydrogen peroxide into water and oxygen rendering it non-toxic. Catalase is also found in all aerobic cells containing cytochrome (Percy et al., 1984), and it is one of the most potent catalysts known. It is the first enzymes which can be purified and crystallized. In plants, catalase scavenges H2O2 generated during mitochondrial electron transport, beta oxidation of fatty acids and most importantly photo respiratory oxidation (Scandalios et al., 1997). This acuumulating evidence indicates that catalase plays an important role in plant defence, aging and senescence (Mura et al., 2007; Conrath et al., 1955). Catalase activity was proportionally increased in starch capped ZnO and CdS nanoparticles but this increase is not significant (p<0.05).Increased activity of catalase indicates that stress has been caused by nanoparticles in the form of enhanced production of reactive oxygen species which is responsible for increase in the enzyme activity. More studies are required in this area to find out the mode of induction, which might be due to increased enzyme synthesis or activity.
Increased activity of catalase indicates that stress has been caused by all the three nanoparticles in the form of enhanced production of reactive oxygen species, which is responsible for the increase in the enzyme activity. Previous study reported (Lee et al., 2010) that 400 mg/L of ZnO nanoparticles released about 14.6 mg/L of soluble zinc. Therefore, we have taken this amount, which was sufficient to increase the activity of catalase enzyme in the plants. More studies are required in this area to find out the mode of induction, which might be due to increased enzyme synthesis or activity.
Figure 4C shows the effect of nanoparticles treatment on changes in protein content of Hydrilla plant. There was significant (p<0.05) increase in protein content of plants treated with CdS nanoparticles. The bivalent cadmium absorbed by plants is released from nanoparticle forms stable complexes with COOH group of protein molecules, present in plant cell which prevents degradation by protease enzyme (Sen et al., 1994). Increase in protein content of plants treated with starch capped ZnO nanoparticles is not significant (p<0.05). In case of plants treated with ZnO nanoparticles there has been significant (p<0.05)
decrease in protein content. Figure 4D shows the tabulated observed results with standard error for controlled and different nanostructures treated plants respectively.
Figure 5 shows the decrease in absorbance with time at 240 nm for 5 min under different nanoparticle treatment on Hydrilla plant from which catalase activity was calculated.
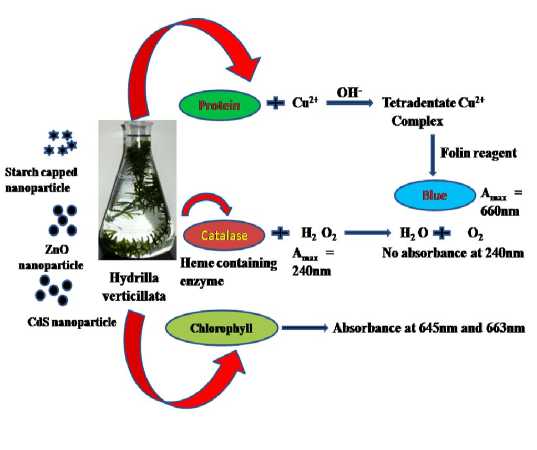
Figure 1 : Schematic graphical representation for Toxicity measurement.
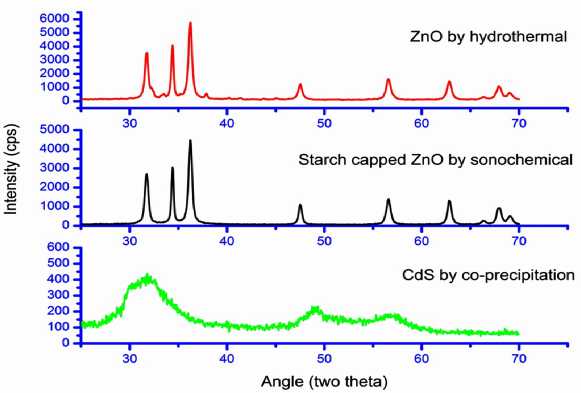
Figure 2 : XRD-diffraction pattern starch capped ZnO nanoparticle synthesized by sonochemical method, ZnO nanoparticle synthesized by hydrothermal and CdS nanoparticle synthesized by simple co-precipitation method.
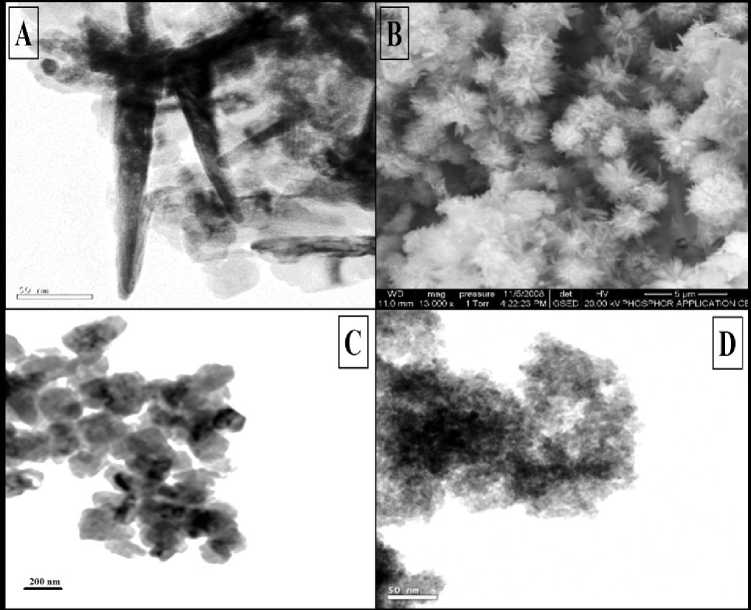
Figure 3 : [A] TEM micrograph of starch capped ZnO. [B] SEM micrograph of flower like starch capped ZnO (Mishra et al., 2010). [C] TEM micrograph of uncapped ZnO. [D] TEM micrograph of uncapped CdS.
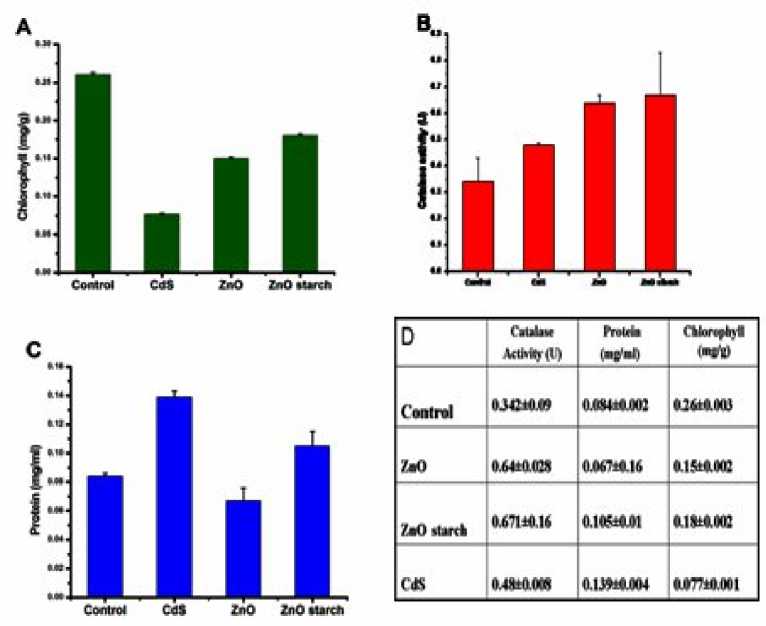
Figure 4 : [A] Effect of different nanoparticles on the content of plant chlorophyll. [B] Effect of different nanostructures on the activity of enzyme catalase. [C] Effect of nanoparticles treatment on changes in protein content. [D] Observed results with standard error for controlled and different nanostructures treated plants.
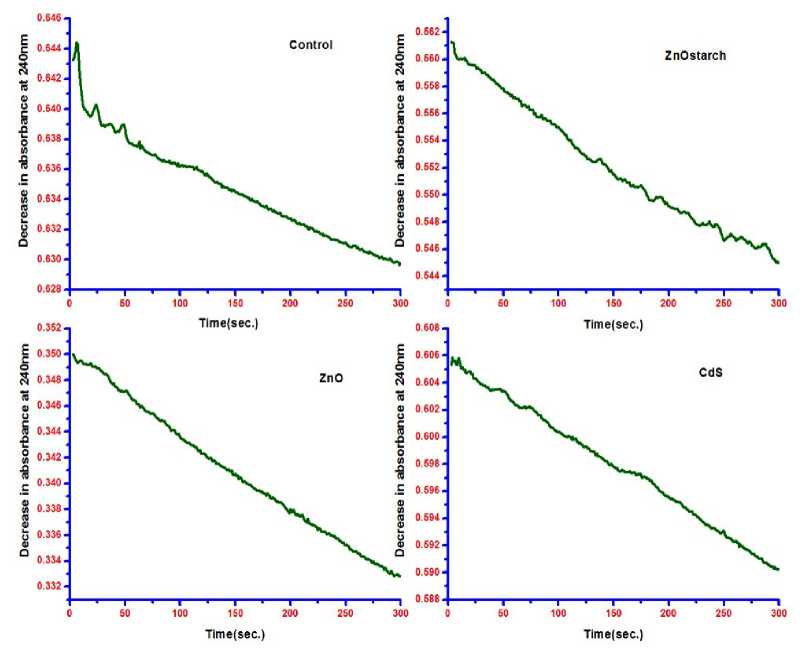
Figure 5 : Decrease in absorbance with time at 240 nm plant.
under different nanoparticle treatment on Hydrilla
DISCUSSION
In conclusion, the applied nanoparticles clearly affected the hydrilla plant as evidenced by changes in enzyme activity and chlorophyll content. The toxicity levels of spherical CdS nanoparticles are more than that of starch capped ZnO nanoparticles having flower-like morphology as well as uncapped spherical ZnO nanoparticles prepared via hydrothermal method. Chlorophyll is one of the most sensitive parameters for toxicity, and our study shows that there has been a decrease in chlorophyll content by about 57.7% in CdS treated plant with respect to 26.82% in ZnO treated plants and 18.18% in starch capped ZnO treated plants. Furthermore, the increase in protein content and activity of catalase enzyme supports more toxicity of CdS nanoparticles. The nanoparticles applied are toxic to the plant and the toxicity is either due to mere presence of nanoparticles or due to dissolution of ions in the solution. When CdS np are applied cadmium is released which is known to be a potentially harmful toxicant so its toxicity increases more both due to extremely small size as well as due to release of ions however when ZnO np are applied zinc is released which is one of the most essential micronutrients for plant system and is toxic only at very high concentration so the toxicity results only from small size of particles hence less as compared to CdS np. Our effort upsurge an interest in understanding the hazards of nanomaterials and their risk, which poses impact on our environment and how they can be monitored via simple biochemical assays on plant systems.
ACKNOWLEDGEMENTS
Priya Mishra and Vineet Kumar Shukla acknowledge the Council of Scientific and Industrial
Research, whereas Raghvendra S. Yadav and Avinash C. Pandey acknowledge the Department of Science and Technology (DST), Govt. of India and FA5209-11-P-0160, ITC-PAC for funding support. We are also thankful to all the scientific members of the Nanotechnology Application Centre (NAC), University of Allahabad for their scientific and technical support.
References Toxicity concerns of semiconducting nanostructures on aquatic plant Hydrilla verticillata
- Aebi, H. (1983) Catalase, In H Bergmeyer, ed, Methods of Enzymatic Analysis 3. Verlag Chemie, Weinheim, Germany. 273-277.
- APHA.: Standard methods for the examination of water and wastewater 15th,ed. Washington, DC, APHA, NY. 1995.
- Arnon, D.I. (1949) Copper Enzymes In Isolated Chloroplasts Polyphenoloxidase In Beta Vulgaris. Plant Physiology. 24: 1-15.
- Conrath, U., Chen, Z., Ricigliano, J.R., Klessig, D.F. (1955) Two inducers of plant defense responses, 2,6-dichloroisonicotinec acid and salicylic acid, inhibit catalase activity in tobacco. Proceedings of the national academy of sciences USA. 92(16): 7143-7147.
- Elgrabli, D., Floriani, M., Abella-Gallart, S., Meunier, L., Gamez, C., Delalain, P., Rogerieux, F., Boczkowski, J., Lacroix, G. (2008). Biodistribution and clearance of instilled carbon nanotubes in rat lung. Particle and Fibre Toxicology. 5: 20.
- Farre, M., Gajda-Schrantz, K., Kantiani, L., Barcelo, D. (2009). Ecotoxicity and analysis of nanomaterials in the aquatic environment. Analytical and Bioanalytical Chemistry. 393: 81-95.
- Griffitt, J.R., Luo, J., Gao, J., Bonzongo, J.C., Barder, S.D. (2008). Effects of particle composition and species on toxicity of metallic nanomaterials in aquatic organisms. Environmental Toxicology Chemistry.27:1972-1978.
- Gulden, M., Morchel, S., Seibert, H. (2005) Comparison of mammalian and fish cell line cytotoxicity: impact of endpoint and exposure duration. Aquatic Toxicology. 71(3): 229-236.
- Handy, R.D., Owen, R., Valsami-Jones, E. (2008). The ecotoxicology of nanoparticles and nanomaterials: Current status, knowledge gaps, challenges, and future needs. Ecotoxicology. 17: 315-325.
- Heinlaan, M., Ivask, A., Blinova, I., Dubourguier, H.C., Kahru, A. (2008). Toxicity of nanosized and bulk ZnO, CuO and TiO2 to bacteria Vibrio fischeri and crustaceans Daphnia magna and Thamnocephalus platyurus. Chemosphere. 71: 1308-1316.
- Hong, F.S., Yang, F., Liu, C., Gao, Q., Wan,Z., Gu,F., Wu,C., Ma,Z., Zhou,J., Yang, P. (2005). Influences of nano-TiO2 on the chloroplast aging of spinach under light. Element Research Biological Trace. 104: 249-260.
- Jiang, W., Mashayekhi, H., Xing, B. (2009). Bacterial toxicity comparison between nano-and micro-scaled oxide particles. Environmental Pollution. 157: 1619-1625.
- Johansen, A., Pedersen, L.A., Jensen, A.K., Karlson, U., Hansen, M.B., Scott-Fordsmand, J.J., Winding, A. (2008). Effects of C60 fullerene nanoparticles on soil bacteria and protozoans. Environment Toxicology Chemistry. 27: 1895-1903.
- Krupa, Z., Baranoneska, M., Orzol, D. (1996) Effect of cadmium on chlorophyll molecule. Acta Physiologiae Plantarum. 18: 147-151.
- Lee, C., Mahendra, S., Zodrow, K., Li,D., Tsai, Y.C., Braam, J., Alvarez. P.J.J (2010). Developmental phytotoxicity of metal oxide nanoparticles to Arabidopsis thaliana. Environment Toxicology Chemistry. 29: 669-675.
- Li, Z., Du, Y. (2003) Biomimic Synthesis of CdS Nanoparticles with Enhances Luminescence. Material Letter. 57: 2480-2484.
- Lin, D., Xing, B. (2008) Root uptake and phytotoxicity of ZnO nanoparticles Environment Science Technology. 42: 5580-5585.
- Lowry, O.H., Rosebrough, N.J., Farr, A.L., Randall, R.J., (1951) Protein measurement with the Folin-Phenol reagents. Journal of Biological Chemistry. 193: 265-275.
- Lu, C.M., Zhang, C.Y., Wen, J.Q., Wu, G.R., Tao, M.X. (2002). Research of the effect of nanometer materials on germination and growth enhancement of Glycine max and its mechanism. Soybean Science. 21: 168-172.
- Mishra, P., Yadav, R.S., Pandey, A.C. (2010). Growth Mechanism and Photoluminescence property of flower-like ZnO nanostructures synthesized by starch-assisted sonochemical synthesis. Ultrasonic Sonochemistry. 17: 560-565.
- Mohan, B.S., Hosetti, B.B. (2006) Phytotoxicity of cadmium on the physiological dynamics of Salvinia natans L. grown in macrophyte ponds. Journal of Environmental Biology. 27: 701-704.
- Moore, M.N. (2006). Do nanoparticles present ecotoxicological risks for the health of the aquatic environment?. Environment International. 32: 967-976.
- Mura, A., Pintus, F., Medda, R., Floris, G., Rinaldi, A.C., Padiglia, A. (2007) Catalase and antiquitin from euphorbia characias:two proteins involved in plant defence? Biochemistry (Mosc), 72 (5): 501-508.
- Nel, A., Xia, T., Madler, L., Li, N. (2006). Toxic potential of materials at the nanolevel. Science. 311: 622-627
- Percy, M.E. (1984) Catalase: an old enzyme with a new role?. Journal of Biochemistry and Cell Biology. 62: 1006-1014.
- Prasad, M.N.V. (2004) Heavy Metal Stress in Plants: From Biomolecules to Ecosystems, 2nd, ed. Springer, New York, USA.
- Sanita di Toppi, L., and Gabbrielli, R. (1999). Responses to cadmium in higher plants. Environmental Experiment Botany. 41: 105-130.
- Scandalios, J.G., Guan, L.M., Polidoros, A.N. (1997) Oxidative stress and the molecular biology of antioxidant defences. Scandalios, J.G. (Ed.). Cold Spring Harbor Lab. Press, Plainview, NY 343-406.
- Sen, A.K., Mondal, N.G., Mondal, S. (1994) Toxic effects of chromium (VI) on the plants Salvinia natans. Environmental Ecology. 12: 279-283.
- Wang, H., Wick, L.R., Xing, B. (2009).Toxicity of nanoparticulate and bulk ZnO, Al2O3 and TiO2 to the nematode Caenorhabditis elegans. Environmental Pollution. 157: 1171-1177.
- Wang, J., Zhang, X., Chen, Y., Sommerfeld, M., Hu, Q. (2008). Toxicity assessment of manufactured nanomaterials using the unicellular green alga Chlamydomonas reinhardtii. Chemosphere. 73: 1121-1128.
- Xia, T., Kovochich, M., Brant, J., Hotze, M., Sempf, J., Oberley, T. (2006). Comparison of the abilities of ambient and manufactured nanoparticles to induce.cellular toxicity according to an oxidative stress paradigm. Nano Letter. 6(8): 1794-1807.
- Yadav, R.S., Pandey, A.C. (2007) Small angle X-ray scattering and photoluminescence study of ZnO nanoparticles synthesized by hydrothermal process. Journal of Experimental Nanoscience. 2:177-180.
- Zheng, L., Hong, F., Lu, S., Liu, C. (2005) Effect of nano-TiO(2) on strength of naturally aged seeds and growth of spinach. Biological Trace Element Research.104: 83-91.

