Beta-Blockers no Longer Needed Post Myocardial Infarction in Patients without Heart Failure: A Meta-Analysis with GRADE Assessment
Author: Atta K.A., Davydkin V.I., AbdelAziz M.S., Sobhy A.M., Abdelaziz A.S.
Journal: Медицина и биотехнологии @medbiosci
Section: Внутренние болезни
Article in issue: 1 т.2, 2026.
Free access
Introduction. Beta-blockers are commonly prescribed after myocardial infarction; however, their benefit in patients without heart failure and with preserved left ventricular ejection fraction in the modern era of reperfusion therapy remains unclear. The aim of the study is to evaluate the effect of beta-blockers on clinical outcomes in patients without heart failure. Materials and methods. The authors conducted a systematic review and meta-analysis of randomized controlled trials evaluating beta-blocker therapy after discharge in patients with myocardial infarction without clinical manifestations of heart failure. Searches in PubMed, Scopus, Web of Science, and Cochrane CENTRAL were performed in February 2026. The primary outcome was major adverse cardiovascular and cerebrovascular events. Secondary outcomes included mortality, recurrent myocardial infarction, heart failure, revascularization, and stroke. Hazard ratios with 95% confidence intervals were pooled using a random-effects model. Results. Four randomized controlled trials including 19,826 patients were analyzed. Beta-blocker therapy was not associated with a significant reduction in the incidence of major adverse cardiovascular and cerebrovascular events (hazard ratio 0.98, 95% confidence interval: 0.91–1.06, p = 0.64) or any of the secondary outcomes: all-cause mortality (hazard ratio 0.98, 95% confidence interval: 0.85–1.13, p = 0.78), recurrent myocardial infarction (hazard ratio 0.88, 95% confidence interval: 0.72–1.07, p = 0.20), heart failure (hazard ratio 0.82, 95% confidence interval: 0.63–1.07, p = 0.14), revascularization (hazard ratio 1.01, 95% confidence interval: 0.87–1.17, p = 0.86), or stroke (hazard ratio 1.24, 95% confidence interval: 0.97–1.59, p = 0.09). Discussion and conclusion. In patients with acute myocardial infarction without heart failure and with preserved left ventricular function, routine beta-blocker therapy after discharge was not associated with a reduction in the incidence of major adverse cardiovascular and cerebrovascular events or their individual components in the context of contemporary reperfusion therapy. Further large-scale studies are required to develop individualized therapeutic approaches.
Beta-blockers, myocardial infarction, preserved ejection fraction, heart failure, meta-analysis
Short address: https://sciup.org/147253463
IDR: 147253463 | UDC: 616-005.8:616.12-008.46 | DOI: 10.15507/3034-6231.002.202601.022-036
Бета-блокаторы больше не требуются после инфаркта миокарда у пациентов без сердечной недостаточности: метаанализ с оценкой качества доказательств по системе GRADE
Введение. Бета-блокаторы обычно назначаются после инфаркта миокарда, однако их польза у пациентов без сердечной недостаточности и с сохраненной фракцией выброса левого желудочка в современную эпоху реперфузионной терапии остается неясной. Цель исследования – оценить влияние бета-блокаторов на клинические исходы у пациентов без сердечной недостаточности. Материалы и методы. Авторы провели систематический обзор и метаанализ рандомизированных контролируемых исследований, в которых оценивалась терапия бета-блокаторами после выписки у пациентов с инфарктом миокарда без клинических проявлений сердечной недостаточности. Поиск по данным PubMed, Scopus, Web of Science и Cochrane CENTRAL проводился в феврале 2026 г. Первичным исходом были серьезные неблагоприятные сердечные и цереброваскулярные события. Вторичные исходы включали смертность, повторный инфаркт миокарда, сердечную недостаточность, реваскуляризацию и инсульт. Отношения рисков с 95% доверительными интервалами были объединены с использованием модели случайных эффектов. Результаты. Были проанализированы четыре рандомизированных контролируемых исследования, включавшие 19 826 пациентов. Терапия бета-блокаторами не ассоциировалась со значимым снижением частоты серьезных неблагоприятных сердечных и цереброваскулярных событий (отношение риска 0,98, доверительный интервал 95 %: 0,91–1,06, p = 0,64) и ни одного из вторичных исходов: общей смертности (отношение риска 0,98, доверительный интервал 95 %: 0,85–1,13, p = 0,78), повторного инфаркта миокарда (отношение риска 0,88, доверительный интервал 95 %: 0,72–1,07, p = 0,20), сердечной недостаточности (отношение риска 0,82, доверительный интервал 95 %: 0,63–1,07, p = 0,14), реваскуляризации (отношение риска 1,01, доверительный интервал 95 %: 0,87–1,17, p = 0,86) или инсульта (отношение риска 1,24, доверительный интервал 95 %: 0,97–1,59, p = 0,09). Обсуждение и заключение. У пациентов с острым инфарктом миокарда без сердечной недостаточности и с сохраненной функцией левого желудочка рутинная терапия бета-блокаторами после выписки не ассоциировалась со снижением частоты серьезных неблагоприятных сердечных и цереброваскулярных событий или его отдельных компонентов в современных условиях реперфузионной терапии. Необходимы дальнейшие крупномасштабные исследования для выработки индивидуализированных подходов к терапии.
Text of the scientific article Beta-Blockers no Longer Needed Post Myocardial Infarction in Patients without Heart Failure: A Meta-Analysis with GRADE Assessment
EDN:
4Университетская больница Кафр-эль-Шейха, Кафр-эль-Шейх, Египет,
5Система здравоохранения Монтефиоре, Медицинский центр Монтефиоре, Нью-Йорк, США,
Before the integration of invasive reperfusion techniques, beta-blockers (BBs) were both the standard of care for patients after myocardial infarction (MI) and the quality indicator of secondary prevention [1]. Their use was initially supported by early randomized trials, which showed around 20% lower risk rates of death among patients who received BBs after discharge from the hospital compared to those who didn’t receive BBs [2; 3]. However, these trials were conducted in an era that was before the modern reperfusion invasive management. In the era of modern rapid reperfusion, percutaneous coronary intervention (PCI) has been recommended as the standard treatment for patients with acute myocardial infarction (AMI) rather than fibrinolytic therapy (Class I, Level A) according to the American College of Cardiology (ACC) / American Heart Association (AHA) Guidelines, published in 2004 [4].
Clinical outcomes following MI have improved, which has resulted in a significant decline in death rates [5]. As a result, the effectiveness of BBs would be diminished or diluted by the modern treatment modality and limited to patients with heart failure with reduced ejection fraction (HFrEF) or multivessel coronary artery disease (MVD) [6]. Recently, randomized controlled trials (RCTs) published in 2024 found that long-term treatment with BBs did not significantly reduce mortality, MI, rehospitalization and stroke rates as opposed to not using BBs [7; 8].
Therefore, we conducted this systematic review and meta-analysis to validate the efficacy of BBs in post-MI patients after discharge from hospital without HF.
MATERIALS AND METHODS
The authors conducted this systematic review and meta-analysis according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) [9], and we followed the methodologies proposed by the Cochrane Handbook for Systematic Reviews of Interventions [10].
Literature Search. The authors conducted a comprehensive search on PubMed, Scopus, Web of Science (WOS), and Cochrane CENTRAL from 2016 to Feb 2026, using the following search term: “acute myocardial infarction” OR “AMI” OR “ST segment elevation myocardial infarction” OR “STEMI” OR “non-ST segment elevation myocardial infarction” OR “NSTEMI” AND “β blocker” OR
“beta blocker” OR “adrenergic beta antagonists” OR “adrenergic beta acceptor blockader” OR “beta adrenergic blocking agents” OR “BB”. We further restricted the search filter for RCT and English language. Additionally, we performed manual forward and backward citation analyses for the relevant studies to ensure all potential studies are being considered for inclusion.
Eligibility Criteria and Endpoints. We screened the retrieved articles in a two-step screening process. We first screened titles and abstracts for all citations using Rayyan software1. Then, we performed full-text screening for the studies assessed for eligibility, identifying all articles that met our inclusion criteria. Any disagreement between the authors were resolved through extensive discussion. We included only peer-reviewed RCTs which met the following criteria:
-
(a) included adult patients with AMI, without HF and with at least left ventricular ejection fraction (LVEF) > 40%,
-
(b) included patients received BBs medications at discharge from the hospital after PCI as the intervention group,
-
(c) included patients did not receive BBs at discharge or at any time of follow-up during the study time frame as the control group, and
-
(d) reported our outcomes of interest at mid-or long-term follow-up period. We further excluded uncompleted trials identified in ClinicalTrials.gov, conference abstracts, and studies that included patients with HFrEF < 40%.
The primary outcome of interest was the major adverse cardiac and cerebrovascular events (MACCE; composite of all-cause mortality, reinfarction, HF, revascularization, and stroke). The secondary outcomes were the individual components of the primary outcome.
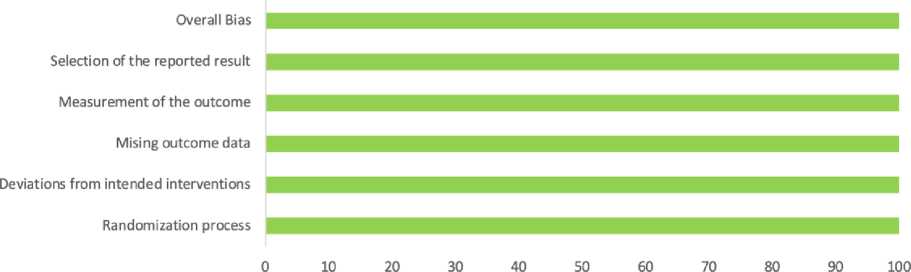
Quality Assessment. We assessed the risk of bias of the included studies using version 2 of the Cochrane risk-of-bias tool for randomized trials (ROB-2) [11]. The tool consists of five domains:
-
(1) bias arising from the randomization process,
-
(2) bias due to deviations form intended intervention,
-
(3) bias due to missing outcomes data,
-
(4) bias in measurement of the outcome, and
-
(5) bias in selection of the reported result.
The studies then evaluated as “low-risk”, “some concern” or “high-risk”.
Data Extraction and Analysis. We used a standardized Excel sheet to extract all the data from the included studies. The data were extracted as:
-
(a) summary and baseline characteristics of the included studies and patients: country, time frame, follow-up period, sample size, age, LVEF %, medical history, BB agent, and revascularization method,
-
(b) the ROB-2 domains, and
-
(c) measurements of the outcomes.
We extracted hazard ratios (HRs) with corresponding 95% confidence intervals (CIs) from each study for the time-to-event outcomes. We then transformed the HRs to logHRs and standard errors (SEs) were derived from reported CIs using established methods recommended by the Cochrane Collaboration Handbook for Systematic Reviews of Intervention [10] using the equation: SEl
log( HR )
ln ( Upper Cl ) - ln ( Lower Cl )
.
2 x 1.96
Pooled effect estimates were calculated using the inverse-variance method and random-effect DerSimonian – Laird model. The heterogeneity was assessed using the Cochrane Q test, and I2 measure was determined across all studies. A p -value < 0.05 and I2 value ≥ 50% were deemed as significant diversity among the included studies. Furthermore, we performed a leave-one-out sensitivity analysis by excluding one study at the time to evaluate the single-study effect on the overall pooled estimate. The results were reported as an overall HR with its 95% CI for each outcome and summarized using forest plots.
We used the Grading of Recommendations Assessment, Development and Evaluation (GRADE) scale [12] to evaluate the strength and level of evidence for recommendations stratified as follows: high quality, which indicates no further research is needed and unlikely to change the confidence of the effects estimations; moderate quality, which indicates that further studies may affect the confidence of the effects estimation; low quality, which indicates further research is likely to have a crucial impact on the confidence of the effects estimation and may change the estimation; and very low quality, which indicates that cannot be certain about this estimation. GRADE assessment was done using GRADEpro software2 and all other statistical analyses were performed by STATA software, version 19.53.
RESULTS
Literature Search. The initial search yielded 1,794 citations, and 458 references were excluded after duplicates removal, leaving 1,336 for first-step screening. After title-abstract screening, 15 articles were eligible for full-text screening. Finally, four studies were included in the analysis [7; 13–15]. The PRISMA flow chart of the selection process is shown in Figure 1.

F i g. 1. The PRISMA flow chart
Source: the figure is prepared by the authors by Microsoft Word.
Studies Characteristics and Quality Assessment. A total of four RCTs were included, from 2018 to 2025. The studies included 19,826 patients, 9,892 (49.9%) were allocated to receive BBs at discharge and 9,934 (51.1%) were allocated to the non-BBs group. The mean age of the total population was 63.3 ± 11.6 years, 79.4% (15,751 patients) were males, 8.9% (1,772 patients) had prior MI, and 2.3% (464 patients) had prior stroke. The mean follow-up was 3.65 years (44 months). All included patients had EF > 50%, except for 1,831 (9.2%) patients in REBOOT and BETAMI-DANBLOCK trials had EF 40–49%. Detailed baseline characteristics is summarized in Table 1.
All included RCTs were assessed for risk of bias using ROB-2 tool, and all of them had “low-risk” of bias in all domains, Figure 2.
Primary Outcome . The rates of MACCE were similar between the patients who received BBs at discharge and those who did not receive BBs, the event rate was 14.08% (1,393 events in 9,892 patients) vs. 14.7% (1,461 events in 9,934 patients), with no significant difference between the two groups (HR 0.98, 95% CI: 0.91–1.06, p = 0.64); and no between-studies heterogeneity was observed (I2= 0%, p = 0.60) (Fig. 3).
Secondary Outcomes . All-cause mortality occurred in 4% (396 of 9,892 patients) in the BBs group and 4.06% (404 of 9,934 patients) in the nonBBs group, with no significant difference (HR 0.98, 95% CI: 0.85 – 1.13, p = 0.78; I2= 0%, p = 0.84) (Fig. 4).
Reinfarction occurred in the BBs group 4.04% (400 of 9,892 patients) and in the non-BBs group 4.6% (456 of 9,934 patients), with no significant difference (HR 0.88, 95% CI: 0.72 – 1.07, p = 0.20; I2= 43.52%, p = 0.19) (Fig. 5).
Revascularization rates were only assessed by three studies, 4.8% (356 of 7,384 patients) in the BBs group and 4.8% (356 of 7,422 patients) required repeated coronary revascularization, the pooled estimate showed no significant difference (HR 1.01, 95% CI: 0.87–1.17, p = 0.86; I2= 0%, p = 0.91) (Fig. 6).
HF occurred in 1.07% (106 of 9,892 patients) in the BBs group and 1.28% (128 of 9,934 patients) in the nonBBs group, with no significant difference (HR 0.82, 95% CI: 0.63–1.07, p = 0.14; I2= 0%, p = 0.80) (Fig. 7).
Stroke occurred in 1.36% (135 of 9,892 patients) in the BBs group and 1.17% (117 of 9,934 patients), with no significant difference (HR 1.24, 95% CI: 0.97–1.59, p = 0.09; I2= 0%, p = 0.51) (Fig. 8).
The leave-one-out sensitivity analyses showed no significant single-study effect was observed in all outcomes, except for stroke outcome. When REDUCE-AMI was excluded, the pooled analysis showed higher risk of stroke in the BBs group (HR 1.41, 95% CI: 1.04–1.92, p = 0.027) (Fig. 9).
Grade Assessment . As shown in Table 2, the certainty of evidence was high for all-cause death, revascularization and HF. However, it was moderate for MACCE, reinfarction and stroke.
DISCUSSION AND CONCLUSION
Our meta-analysis included a large sample size of 19,826 MI patients without HF from four RCTs. The pooled analysis demonstrated that the routine use of BBs after MI was not statistically associated with significant reduction in MACCE or any of its components. Our analysis showed comparable rates of all-cause mortality, reinfarction, HF, repeated coronary revascularization, and stroke between the patients discharged on BBs therapy and those who were not. These findings are particularly relevant in the modern era of rapid reperfusion and innovation in PCI technology.
Previous landmark trials which were largely from the pre-PCI era, established BBs as a standard therapy following MI, reporting significant reductions in allcause mortality and cardiovascular mortality [2; 3; 16]. However, modern evidence driven by more rapid reperfusion strategies, optimizing secondary prevention, and modern trials designs, suggests that the benefit of the BBs in this post-MI patients without HF may be minimal or even absent [7; 14]. Our findings align with the most recent JAMA Cardiology meta-analysis that synthesized RCTs especially in post-MI patients with preserved EF, they found unnoticeable improvement in cardiovascular outcomes with BBs medication and did not significantly lower the composite of allcause death, recurrent MI, or HF hospitalization over a median follow-up of 3.5 years [17]. An individual patient data (IPD) meta-analysis published in The New England Journal of Medicine similarly showed neutral results regarding the primary composite endpoint (all-cause death, MI, and HF) between the BBs group and the control group [18]. These findings, which were consistent across mortality, reinfarction, and HF, indicate that the mechanistic benefits of BBs, such as decreased sympathetic drive and prevention of adverse remodeling, may not be translated into significant clinical outcomes differences when it comes to the portion of patients with normal pumping function and optimal therapy [19; 20].
T a b l e 1. Baseline characteristics of the included patients
CABG N/A N/A 92 (3.7) 103 (4.1) 6 (0.1) 10 (0.2) 46 (1.7) 56 (2.0)
Note: data present as n (%) or mean ± SD; AF – atrial fibrillation, BBs – beta-blockers, CABG – coronary artery bypass grafting, DM – diabetes mellitus, LVEF – left ventricular ejection fraction, MI – myocardial infarction, PCI – percutaneous coronary intervention, STEMI – ST-elevation myocardial infarction, N/A – not assessed.
Source: here and below all tables were compiled by the authors.
Risk of bias domains
Study
|
D1 |
D2 |
D3 |
D4 |
D5 |
Overall |
|
|
CAP1TAL-RCT |
+ |
+ |
+ |
|||
|
REDUCE-AMI |
+ |
+ |
+ |
|||
|
REBOOT |
+ |
+ |
+ |
|||
|
BETAMI-DANBLOCK |
+ |
+ |
+ |
|
Domains: |
Judgement |
|
D1: Bias due to the randomization process |
+ Low |
|
D2: Deviation from intended intervention |
I Some concern |
|
D3: Missing outcome data |
■ I High risk |
D4: Measurement of the outcome
D5: Selection of the reported result
As percentage (intention-to-treat)
-
■ Low risk Some concerns ■ High risk
F i g. 2. Risk of bias assessment using ROB-2
Source: the figure is prepared by the authors by Microsoft Excel.
|
CAPITAL-RCT |
127 / 394 |
130/400 |
.0099503 |
|
REDUCE-AMI |
265/2508 |
288/2512 |
-.0512933 |
|
REBOOT |
550 / 4207 |
536 / 4231 |
.0295588 |
|
BETAMI-DANBLOCK |
451 / 2783 |
507/2791 |
-.1053605 |
Overall
Heterogeneity: T2 = 0.00, I2 = 0.00%, H2 = 1.00
Test Of 0 = 9,: Q(3) = 1.85, p = 0.60
Test of 9 = 0: z = -0.47, p = 0.64
|
.1270629 .0858348 .0591331 .0872693 |
0------------ |
1.01 [0.79, 1.30] 10.07 0.95(0.80, 1.12] 22.07 1.03(0.92, 1.16] 46.50 0.90(0.76, 1.07] 21.35 0.98(0.91, 1 06] |
|
|
I |
|||
|
I |
|||
|
i i |
|||
|
0.76 |
1.30 |
||
|
Hazard ratio Weight |
|
|
Study |
BBs Non-BBs logHR SE(logHR) with 95% Cl (%) |
Random-effects REML model
F i g. 3. Random-effect model of MACCE rates
Source: from here and below the figures were prepared by the authors in the statistical software program (STATA, version 19.5, StataCorp LLC).
|
Study BBs Non-BBs logHR SE(logHR) |
Hazard ratio Weight with 95% Cl (%) |
||
|
CAPITAL-RCT 20/394 24/400 -.1508229 .3044075 REDUCE-AMI 97/2508 103/2512 -.0618754 .1422453 REBOOT 161 /4207 153/4231 .0582689 .1142086 BETAMI-DANBLOCK 118/2783 124/2791 -.0618754 .128911 Overall Heterogeneity: t2 = 0.00, I2 = 0.00%, H2 = 1.00 Test of 0, = 0,: Q(3) = 0.85, p = 0.84 Test of 0 = 0: z = -0.28, p = 0.78 |
1 |
0.86(0.47, 1.56] 5.48 0.94(0.71, 1.24] 25.08 1.06(0.85, 1.33] 38.91 0.94(0.73, 1.21] 30.54 0.98(0.85, 1.13] |
|
|
° 1 1 |
|||
|
0.5 |
|||
Random-effects REML model
F i g. 4. Random-effect model of all-cause mortality rates
Hazard ratio Weight
Study BBs Non-BBs logHR SE(logHR) with 95% Cl (%)
CAPITAL-RCT 7/394 10/400 -.3424903 .4991937 •-----°---1--0.71 [ 0.27, 1.89]3.72
REDUCE-AMI 112/2508 117/2512 -.040822 .1316879 ---rC--- 0.96 [ 0.74, 1.24]29.45
REBOOT 143/4207 143/4231 .0099503 .1178981 -^O--- 1.01 (0.80,1.27]32.82
ВЕТАМ l-DANBLOCK 138/2783 186/2791 -.3147107 .1133294 --ф-т 0.73(0.58, 0.91]34.01
Overall -^^- 0.88(0.72, 1.07]
Heterogeneity: t2 = 0.02,12 = 43.52%, H2 = 1.77
Test of 0i = 0):Q(3) = 4.71, p = 0.19
Test of 0 = 0: z = -1.29, p = 0.20।
0.5 12
Random-effects REML model
F i g. 5. Random-effect model of reinfarction rates
|
Study BBs Non-BBs logHR SE(logHR) |
Hazard ratio Weight with 95% Cl (%) |
||
|
CAPITAL-RCT 78/394 75/400 .076961 .1633937 REBOOT 170/4207 171 /4231 0 .1086307 BETAMI-DANBLOCK 108/2783 110/2791 -.0100503 .1349691 Overall Heterogeneity: t2 = 0.00,12 = 0.00%, H2 = 1.00 Test of 0 = 0; Q(2) = 0.20, p = 0.91 Test of 0 = 0: z = 0.18, p = 0.86 |
1.08(0.78, 1.49] 21.15 1.00(0.81, 1.24] 47.85 0.99(0.76, 1.29] 31.00 1.01 [0.87, 1.17] |
||
|
------< -------c |
к------- i------------------------------------- I |
||
|
1 1 1 |
|||
|
0.76 Random-effects REML model |
1.49 |
||
F i g. 6. Random-effect model of revascularization rates
Hazard ratio Weight
|
Study |
BBs |
Non-BBs |
|
CAPITAL-RCT |
5/394 |
10/400 |
|
REDUCE-AMI |
20 / 2508 |
22 /2512 |
|
REBOOT |
39 / 4207 |
44/4231 |
|
BETAMI-DANBLOCK |
42 / 2783 |
52 /2791 |
-.6733446
-.0943107
-.1165338
-.2484614
logHR SE(logHR) with 95% Cl (%)
Overall
Heterogeneity: t2 = 0.00,12 = 0.00%, H2 = 1.00
Test of 9, = 0,: Q(3) = 1.02, p = 0.80
TestofS = 0:z =-1.48, p = 0.14
|
0.51 [0.17, 1.54] |
5.50 |
|
0.91 [0.50, 1.66] |
18.67 |
|
0.89 [0.58, 1.37] |
35.79 |
|
0.78 [0.52, 1.17] 0.82 [0.63, 1.07] |
40.04 |
0.25 0.5 1 2
Random-effects REML model
F i g. 7. Random-effect model of HF rates
Hazard ratio Weight
|
Study BBs Non-BBs logHR SE(logHR) |
with 95% Cl (%) |
||
|
CAPITAL-RCT 17/394 11 /400 .463734 .3929707 REDUCE-AMI 36/2508 46/2512 -.040822 .2202291 REBOOT 37/4207 25/4231 .4054651 .2596029 BETAMI-DANBLOCK 45/2783 35/2791 .2623643 .2250993 Overall Heterogeneity: t2 = 0.00,12 = 0.00%, H2 = 1.00 Test of 9 = 0; Q(3) = 2.33, p = 0.51 Test of 0 = 0: z = 1.70, p = 0.09 |
1.59 [ 0.74, 3.43] 10.50 0.96 [0.62, 1.48] 33.43 1.50 [0.90, 2.49] 24.06 1.30 [0.84, 2.02] 32.00 1.24 [0.97, 1.59] |
||
|
----0 |
------------------------1-------------------- — О— —Iе--- |
||
|
0.625 |
|||
Random-effects REML mode!
F i g. 8. Random-effect model stroke rates
|
Omitted study |
BBs |
Non-BBs |
Hazard ratio with 95% Cl |
p-value |
|
CAPITAL-RCT |
17/394 |
11 /400 |
—H--------- 1.21 [ 0.93, 1.57] |
0.163 |
|
REDUCE-AMI |
36 /2508 |
46/2512 |
—;----•------------1.41 [ 1.04, 1.92] |
0.027 |
|
REBOOT |
37 /4207 |
25/4231 - |
-----«-r-------- 1.17(0.88, 1.56] |
0.284 |
|
BETAMI-DANBLOCK |
45 /2783 |
35/2791 - |
—4------------ 1.24(0.87, 1.76] |
0.238 |
|
0.87 |
1.92 |
Random-effects REML model
F i g. 9. Leave-one-out sensitivity analysis of stroke
These meta-analyses reflect a growing concern about the universal prescription and benefits of BBs after MI without pre-specified patient-individualization strategies, their use is no longer justified in patientswith preserved EF, and only restricted to patients with mildly reduced EF (HFmrEF) and HFrEF [17; 18].
These modern data contrast with findings suggesting that the only benefit from BBs is in HFmrEF, where subgroup and IPD analyses have found a relative event reduction in this subgroup [21]. However, even in this specific subgroup, the magnitude of benefit appears to be smaller than historically suggested by earlier trials [22].
The reasons for this transition are diverse. Modern MI therapy, which includes rapid revascularization using PCI, antiplatelet therapy, statins, ACE/ARB/ARNI, and SGLT2 inhibitors, has significantly reduced the overall mortality and adverse event rates, limiting the marked contribution of particular medications, such as BBs in low-risk subgroups [23–25]. In patients with preserved EF, baseline cardiac function is already sufficient, so additional protective effect of blunting sympathetic drive may be less important than in patients with reduced systolic function or clinical HF, where neurohormonal modulation remains critical to prognosis [26].
Moreover, by excluding REDUCE-AMI, our sensitivity analysis revealed increased stroke risk after long-term use of BBs. Although this finding must be discussed with caution due to the low power and small number of trials, it should not be ignored. BBs may cause bradycardia or blood pressure drop in low-risk patients, thereby impairing cerebral perfusion reserve and predispose to ischemic events particularly in older patients with vascular stiffness [27; 28]. Importantly, none of the included trials were designed to evaluate stroke outcomes and reported stroke rates as an additional exploratory outcome. Therefore, while the overall analysis showed no significant increase in stoke risk, the sensitivity analysis highlight the need for individualized strategy rather than routine long-term therapy in post-MI patients with preserved EF.
T a b l e 2. GRADE assessment

Note: CI – confidence interval, HR – hazard ratio, RCT – randomized controlled trial, MACCE – major adverse cardiovascular and cerebrovascular event, a – included number of events not number of patients experienced an event, b – there was a moderate heterogeneity between the studies reporting reinfarction, c – sensitivity analysis changed stroke result when REDUCE-AMI was excluded.
Clinical Recommendations
For decades, BBs have been prescribed after AMI for life and often continued regardless the LV function. The consistent findings of the RCTs and pooled meta-analyses in patients with preserved EF and without HF suggest that this universal routine of BBs prescription should be reconsidered. The patients with LVEF > 50% who had successful revascularization and receiving secondary prevention medications, such as DAPT, statins, or ACE/ARB/ARNI, the addition of long-term BBs does not secure against mortality, reinfarction, heart failure, revascularization, or stroke rates. Therefore, BBs should be individualized rather than universalized and may to be considered only for patients with reduced EF (< 50%), clinical heart failure, persistent angina, significant ventricular arrhythmias, and other indications such as atrial fibrillation or hypertension.
For stable post-MI patients with preserved EF and no other indications for BBs, cardiologists should consider deprescribing after the stabilization phase, especially if patients exhibit adverse effects such as tiredness, bradycardia, hypotension, or decreased exercise tolerance, and shared discussion with the patients should be carried out. Limitations and Future Directions
Several limitation worth careful considerations. First, the number of included studies was small, which limits the power for detecting modest treatment effects and perform several analyses, such
as Funnel plot, Egger’s test, Galbraith plot, and subgroup analyses. Second, approximately 9% of the included patients had mildly reduced EF (40–49%), which might alter the overall findings and introduces a degree of clinical heterogeneity. Third, the differences in BBs type, dosing strategies and differential effects of specific agents were not explored in the included RCTs. Fourth, follow-up duration was mid-term (3–4 years), which limit the knowledge of the longterm effects of BBs. Future trials should focus on including only patients with HFpEF with longer follow-up durations and further stratification and multivariable analyses based on LVEF degree, infarct size, arrhythmic risk profile, and revascularization technique, providing more individualized approach regarding the prescription of BBs post-MI at discharge.
Conclusion
In patients with AMI without HF and preserved LV function, the most recent pooled evidence from RCTs suggest that routine BBs therapy after discharge from the hospital was not associated with reduction in MACCE rates or its individual components in the modern reperfusion era. These findings suggest that the universal prescription of BBs in all post-MI patients without absolute indications may warrant reconsideration, particularly among low-risk individuals with preserved systolic function.


