Цитокиновый статус сыворотки крови больных раком яичников с различным уровнем циркулирующих опухолевых клеток
Автор: Генинг Снежанна Олеговна, Абакумова Татьяна Владимировна, Антонеева Инна Ивановна, Гафурбаева Дина Урхановна, Мифтахова Регина Ривкатовна, Долгова Динара Ришатовна, Песков Андрей Борисович
Журнал: Ульяновский медико-биологический журнал @medbio-ulsu
Рубрика: Клиническая медицина
Статья в выпуске: 4, 2019 года.
Бесплатный доступ
Циркулирующие опухолевые клетки (ЦОК) представляют собой основу гематогенного метастазирования. В 2003 г. было продемонстрировано их существование в крови больных раком яичников (РЯ). Известно, что воспаление играет важную роль в прогрессировании опухолей. В крови ЦОК встречаются с большим количеством компонентов, в т.ч. с цитокинами, которые способны модулировать метастатический потенциал опухолевой клетки. Целью данного исследования была оценка цитокинового статуса сыворотки крови больных раком яичников с различным уровнем циркулирующих опухолевых клеток. Материалы и методы. Объектом исследования явились первичные больные с верифицированным (гистологически либо цитологически) раком яичников II-IV степеней по FIGO (n=24) до лечения. В крови пациенток определяли число циркулирующих опухолевых клеток методом проточной цитофлюориметрии и уровни IL-6, IL-17A, IL-1ft, TGF-a, IL-4, VEGF, TNF-a, HGF, IL-18, IL-10, IL-8. Обработку результатов проводили с использованием пакета программ Statistica Windows...
Рак яичников, цитокины, циркулирующие опухолевые клетки
Короткий адрес: https://sciup.org/14116402
IDR: 14116402 | УДК: 616-006.66:616-08-039.34 | DOI: 10.34014/2227-1848-2019-4-50-62
Blood serum cytokine status in ovarian cancer patients with different levels of circulating tumor cells
Circulating tumor cells (CTCs) are essential for hematogenous metastasis. In 2003, it was found out that such cells were present in the blood of patients diagnosed with ovarian cancer (OC). It is known that inflammation plays an important role in tumor progression. There are CSCs with a large number of components in the blood, e.g. cytokines that can modulate the metastatic potential of a tumor cell. The aim of the study is to assess the blood serum cytokine status in ovarian cancer patients with different levels of circulating tumor cells. Materials and Methods. Untreated primary patients (n=24) with histologically or cytologically verified ovarian cancer, stage II-IV according to FIGO classification, were the trial subjects. Flow cytometry was used to detect the number of circulating tumor cells in the blood from the patients; the authors also determined IL-6, IL-17A, IL-1ft, TGF-a, IL-4, VEGF, TNF-a, HGF, IL-18, IL-10, IL-8 levels. The results were processed using the Statistica Windows software package...
Текст научной статьи Цитокиновый статус сыворотки крови больных раком яичников с различным уровнем циркулирующих опухолевых клеток
Введение. Присутствие опухолевых клеток в крови онкологических больных впервые было открыто в 1869 г., но большой интерес к этой проблеме возник лишь в последние два десятилетия, что связано с развитием методик, позволяющих количественно и качественно оценить такие клетки. В 2003 г. P.L. Judson с соавт. продемонстрировали существование циркулирующих опухолевых клеток (ЦОК) у больных эпителиальным раком яичников (РЯ) [1]. В зависимости от метода детекции количество ЦОК-положительных пациенток с РЯ в исследованиях варьируется от 18 до 88 % от общего числа [2]. ЦОК представляют собой промежуточную ступень гематогенного метастазирования. Они могут проникать в кровеносное русло активно, расщепляя межклеточный матрикс при помощи матриксных металлопротеи- наз (MMP), либо пассивно за счет непрочности межклеточных контактов в опухоли [3]. В кровеносном русле ЦОК находятся в виде единичных клеток и в виде кластеров [4]. Большинство клеток погибает внутри сосудов под воздействием физических факторов, оксидатив-ного стресса, аноикиса, из-за недостатка факторов роста и цитокинов. Выжившие клетки, достигшие паренхимы органа-мишени, могут персистировать в нем в состоянии покоя или «спячки», не пролиферируя. Продолжительность «спячки» определяется внутриклеточными факторами и свойствами микроокружения. Лишь небольшая часть ЦОК дает начало метастатическому очагу [5]. ЦОК гетерогенны: они могут иметь эпителиальный, мезенхимальный или промежуточный фенотип; могут иметь свойства стволовых клеток опухоли. Предпола- гается, что именно стволовые ЦОК являются основой для развития метастазов [6]. ЦОК не только предшествуют отдаленным метастазам, но также имеют свойство возвращаться в первичный очаг (самообсеменение опухоли) и давать начало более агрессивному клону [7]; кроме того, они могут взаимодействовать с клетками крови (нейтрофилами, моноцитами, тромбоцитами), что в одних случаях приводит к уничтожению ЦОК, а в других – способствует метастазированию. Приобретение мезенхимального фенотипа в ходе эпителиально-мезенхимального перехода благоприятствует активной интравазации раковой клетки, а мезенхимально-эпителиальный переход – выходу из состояния «спячки» и началу пролиферации [8]. Кроме того, эпителиальный фенотип, в частности экспрессия Е-кадгерина, облегчает формирование кластеров опухолевых клеток. В составе кластеров, вероятно, более мезенхимальные клетки тянут за собой более эпителиальные (коллективная миграция) [9]. Феномен эпителиальной пластичности (существование промежуточных фенотипов опухолевых клеток) описан при различных локализациях опухолей, в т.ч. при РЯ [10], при этом, по-видимому, наиболее агрессивными являются именно промежуточные клетки: показано, что им присущи свойства стволовых клеток опухоли [11] и высокая устойчивость к аноикису [12].
В крови ЦОК встречаются с большим количеством компонентов, в т.ч. с цитокинами. Известно, что воспаление играет важную роль в прогрессии опухолей [13]. Цитокины способны активировать онкогены, подавлять гены-супрессоры, менять уровни транскрипции сигнальных белков, в т.ч. в условиях воздействия химиопрепаратов [14].
Цель исследования. Оценка цитокино-вого статуса сыворотки крови больных раком яичников с различным уровнем циркулирующих опухолевых клеток.
Материалы и методы. Объектом исследования являлись первичные больные с верифицированным (гистологически либо цитологически) раком яичников II–IV степеней по FIGO в возрасте от 36 до 78 лет (средний возраст – 63,50±10,63 года), поступавшие в стационар гинекологического отделения ГУЗ «Ульяновский областной клинический онко- логический диспансер» в 2018–2019 гг. (n=24). Перед поступлением все больные проходили обследование для оценки степени распространенности опухоли (осмотр гинеколога, рентгенография органов грудной клетки, УЗИ брюшной полости, забрюшинного пространства и малого таза, МРТ органов малого таза и определение маркера СА-125).
Забор крови осуществлялся после подписания информированного согласия и до начала специального лечения. Исследование проводилось согласно требованиям комиссии по этике ИМЭиФК ФГБОУ ВО «Ульяновский государственный университет» (протокол № 3 от 15.03.2015). Образцы крови пациенток анализировались на предмет наличия циркулирующих опухолевых клеток. При этом из 7,5 мл стабилизированной цитратом натрия крови выделялась мононуклеарная фракция клеток методом центрифугирования на градиенте фи-колла (плотность 1,077 см3). Выделенные клетки красились с помощью флюоресцентно меченных антител к цитокератинам (СК-pan), общелейкоцитарному антигену CD45 и молекуле адгезии эпителиальных клеток (EpCAM). Анализ проводился на проточном цитометре Cytoflex S (BECKMAN COULTER, USA). За циркулирующие опухолевые клетки была принята популяция CD45-/Epcam+/CK+-клеток. Цитокиновый профиль сыворотки крови: уровни IL-6, IL-17A, IL-1β, TGFα, IL-4, VEGF, TNF-α, HGF, IL-18, IL-10, IL-8 – оценивался при помощи мультиплексного магнитного набора для оценки циркулирующих опухолевых биомаркеров MILLIPLEX HCCBP1MAG-58K (Merck KGaA, Germany). Связь между клиническими и молекулярными параметрами анализировалась с использованием непараметрического критерия Манна– Уитни и One-way ANOVA. Статистически значимыми считались значения при р≤0,05.
Результаты. Нами было установлено, что уровень ряда цитокинов в сыворотке крови больных РЯ: TNF-α, HGF, IL-10, IL-18 и IL-8 – статистически значимо изменялся в соответствии с количеством ЦОК. Так, концентрация TNF-α возрастала с увеличением числа ЦОК от 6,69 до 15,14 нг/мл. Содержание фактора роста гепатоцитов (HGF) возрастало с 383,19 до 555,61 нг/мл (рис. 1).
TNFα
HGF
Ё 15 ад с; 10
S х 5
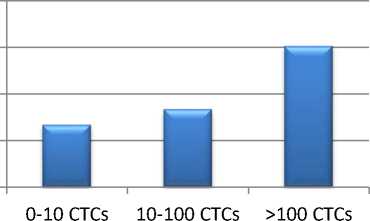
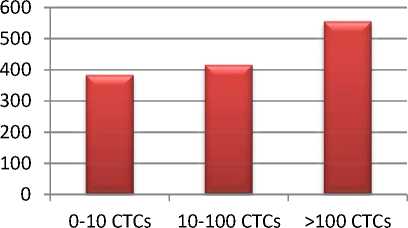
Рис. 1. Уровни TNF-α и HGF в сыворотке крови больных РЯ с различным количеством ЦОК
Fig. 1. TNF-α and HGF levels in the blood serum from OC patients with dissimilar number of CTCs
Уровень IL-18 возрастал с 168,47 до 304,52 нг/мл, IL-10 – с 12,43 до 231,43 нг/мл (рис. 2).
Содержание IL-8, напротив, снижалось с 77,09 до 28,93 нг/мл (рис. 3).
Уровни цитокинов IL-6, IL-17A, IL-1β, TGFα, IL-4, VEGF и лептина статистически значимо не коррелировали с числом ЦОК (табл. 1).
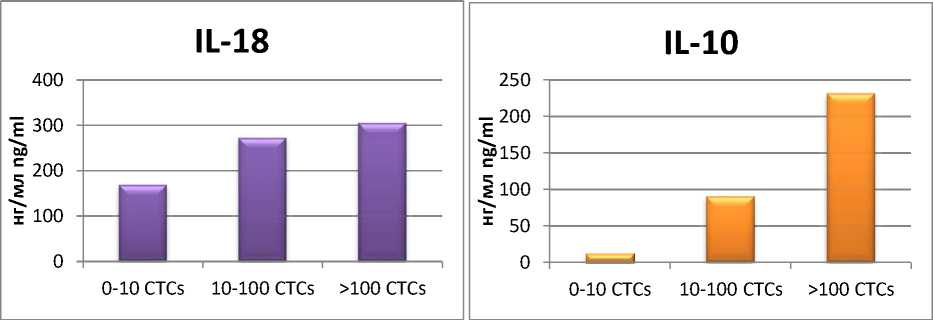
Рис. 2. Уровни IL-18 и IL-10 в сыворотке крови больных РЯ с различным количеством ЦОК
Fig. 2. IL-18 and IL-10 levels in the blood serum from OC patients with dissimilar number of CTCs
IL-8
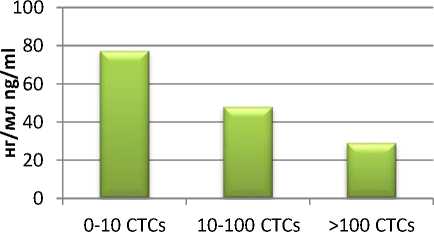
Рис. 3. Уровни IL-8 в сыворотке крови больных РЯ с различным количеством ЦОК Fig. 3. IL-8 level in the blood serum from OC patients with dissimilar number of CTCs
Таблица 1
Table 1
Уровни цитокинов в периферической крови больных РЯ с различным количеством ЦОК
Cytokine levels in the peripheral blood from OC patients with dissimilar number of CTCs
|
Группа Group |
IL-6, нг/мл IL-6, ng/ml |
IL-17A, нг/мл IL-17A, ng/ml |
IL-1β, нг/мл IL-1β, ng/ml |
TGFα, нг/мл TGFα, ng/ml |
IL-4, нг/м IL-4, ng/ml |
VEGF, нг/мл VEGF, ng/ml |
Лептин, нг/мл Leptin, ng/ml |
|
0–10 ЦОК, n=11 0–10 CTCs, n=11 |
7,18 (2,92– |
39,61 (29,6– |
5,00 (3,84– |
25,29 (9,54– |
3,09 (1,48– |
432,4 (146,0– |
3878,2 (2387,7– |
|
10,38) |
75,6) |
9,81) |
51,39) |
9,40) |
924,6) |
192928,8) |
|
|
10–100 ЦОК, |
16,61 |
246,9 |
4,33 |
31,05 |
2,73 |
392,4 |
19442,8 |
|
n=5 10–100 CNCs, n=5 |
(1,1– |
(30,4– |
(4,04– |
(2,96– |
(1,91– |
(146,0– |
(15,95– |
|
35,6) |
891,7) |
4,53) |
43,60) |
4,17) |
776,6) |
74271,90) |
|
|
>100 ЦОК, |
9,22 |
36,06 |
3,97 |
19,93 |
1,95 |
583,46 |
20228,8 |
|
n=8 >100 CTCs, n=8 |
(3,08– |
(26,9– |
(3,11– |
(4,39– |
(1,40– |
(146,0– |
(1247,3– |
|
21,20) |
46,2) |
6,60) |
42,70) |
2,34) |
890,3) |
59268,4) |
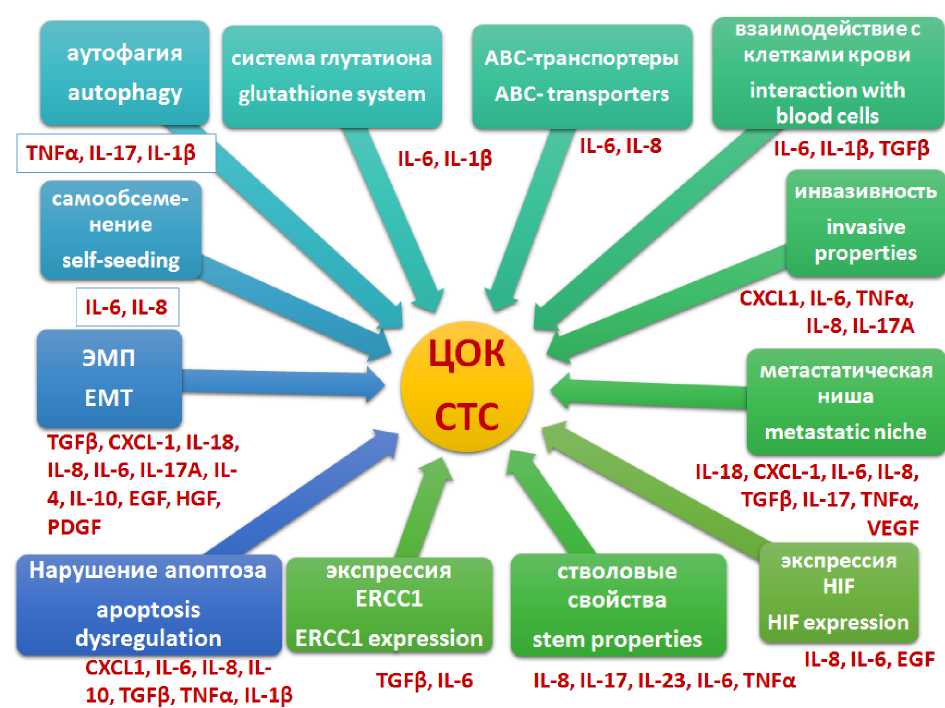
Обсуждение. Возможные прометастати-ческие эффекты цитокинов при их взаимодействии с ЦОК могут быть представлены в виде схемы (рис. 4).
Источником цитокинов в периферической крови могут служить как нормальные клетки организма больного, так и ЦОК. Показано, что ЦОК способны индуцировать системное воспаление, увеличивая выработку провоспалительных цитокинов [15].
IL-8 индуцирует образование сфероидных колоний и повышение экспрессии CD133 и CD44 в клетках РЯ [16]; он также активирует NFκB-сигналинг, поддерживающий стволо-вость клеток РЯ [17, 18], и повышает экспрессию ABCB1-транспортера [19]. Цитокины способны активировать антиапоптотические сигнальные пути: IL-8 стимулирует пролиферацию клеток РЯ, повышая уровни циклинов D1 и B1 и активируя такие пути, как PI3K/Akt, Raf/MEK/ERK [20]. Блокада IL-8 повышает чувствительность клеток серозного РЯ к цисплатину, снижает экспрессию Bcl-2 и повышает активность Bad [21]. Высокий сывороточный уровень IL-8 при РЯ связан со снижением выживаемости [22]. Продукция IL-8 циркулирующими клетками меланомы в капиллярах легких привлекает нейтрофилы и способствует метастазированию [23]. В отношении
его связи с ЦОК данные неоднозначны: в одной работе [24] корреляция уровня IL-8 с числом ЦОК рака молочной железы (РМЖ) была положительной, в другом исследовании [25] уровень IL-8 в присутствии ЦОК был снижен по сравнению с ЦОК-отрицательной группой больных РМЖ. В нашей работе также наблюдалось снижение концентрации IL-8 в крови с повышением числа ЦОК. Возможно, это связано с повышением его утилизации; данный факт требует дальнейшего изучения.
Известно, что HGF-сигналинг высокоактивен в опухолевой ткани РЯ и способствует пролиферации, инвазии и миграции клеток [26]. HGF входит в состав экзосом, секретируемых клетками РЯ [27]; он также высвобождается при расщеплении внеклеточного матрикса металлопротеиназами [28], что происходит в ходе интравазации ЦОК. IL-18 является плейотропным цитокином, способным регулировать такие сигнальные пути, как MAPK и PI3K/Akt [29]. При РЯ наблюдается повышение его концентрации как в микроокружении опухоли [30], так и в сыворотке крови больных [31]. IL-10 – противовоспалительный цитокин. Наличие полиморфизма промотора IL-10 ассоциировано с повышением общей и безрецидивной выживаемости при РЯ [32]. IL-10, продуцируемый макро-
фагами, активирует в опухолевых клетках Stat3 и выработку циклина D1 [33].
Таким образом, вышеперечисленные цитокины активируют ЦОК, повышают их тумо-рогенность, и представляется логичной тенденция к увеличению их уровней при возрастании числа ЦОК.
При помощи цитокинов ЦОК взаимодействуют и с клетками крови. ЦОК и нейтрофилы при помощи цитокиновой сигнализации способны объединяться в кластеры (нейтрофилы секретируют TNF-α, IL-1β, IL-6, ЦОК экспрессируют рецепторы к ним; в то же время ЦОК секретируют G-CSF, TGF-β3 и IL-15, рецепторы к которым имеются на нейтрофилах). В подобном кластере ЦОК приобретают более агрессивные свойства (дисрегуляция клеточного цикла и высокая пролиферативная активность), что способствует метастазированию [34]. Известно, что TNF-α, секретируемый клетками РЯ, индуцирует секрецию фибробла-
стами TGF-α, способствующего метастазированию [35]. При этом в клетках РЯ отмечено повышенное содержание TNF-α [36, 37]. TNF-α способен стимулировать выживание и метастазирование клеток РЯ, а также ангиогенез [38]. Высокая концентрация TNF-α в крови способна стимулировать привлечение циркулирующих клеток карциномы молочной железы в воспаленный эндотелий [39]. Трансэндотелиальная миграция ЦОК в ткань осуществляется при помощи EGF и TNF-α, секретируемых макрофагами и моноцитами [40, 41].
Как следует из представленного выше анализа данных литературы, роль TNF-α в опухолевой прогрессии крайне разнообразна. При этом данные об уровне TNF-α в сыворотке крови при РЯ противоречивы [42, 43]. Используя метод One-way ANOVA, мы установили, что в интервале ЦОК от 120 до 175 резко и значимо возрастает уровень TNF-α (рис. 5).
------------------------- IL-8,1L-6, E G F IL-8, IL-17, IL-23, IL-6, TN Fa
TGFp, CXCL-1, IL-18, IL-8, IL-6, IL-17A, IL-4, IL-10, EGF, HGF, PDGF
CTC
IL-18, CXCL-1, IL-6, IL-8,
TGFp, IL-17, TNFa, \ VEGF
CXCL1, IL-6, IL-8, IL-10, TGFp,TNFa, IL-lp
TGFP, IL-6
IL-6, IL-lp, TGFp
TNFa, IL-17, IL-ip
IL-6,IL-8
ЦОК
CXCL1, IL-6, TNFa, IL-8, IL-17A взаимодействие c клетками крови interaction with blood cells аутофагия autophagy
АВС-транспортеры ABC-transporters система глутатиона glutathione system самообсеме-нение self-seeding метастатическая ниша metastatic niche экспрессия HIF
HIF expression
Нарушение апоптоза apoptosis dysregulation
ERCC1 expression экспрессия ERCC1
ЭМП
EMT инвазивность invasive properties стволовые свойства stem properties
Рис. 4. Схематическое изображение участия цитокинов в регуляции биологических характеристик ЦОК при РЯ
Fig. 4. Schematic representation of cytokine effect on the regulation of CTC biological characteristics in OC
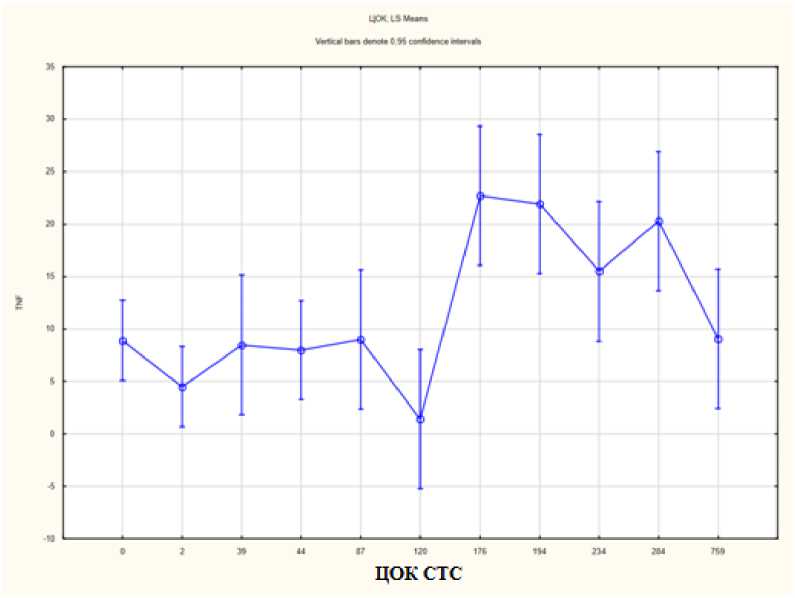
Рис. 5. Связь количества ЦОК с уровнем сывороточного TNF-α при РЯ
Fig. 5. Correlation of CTC number with serum TNF-α level in OC
Изменяется функция, описывающая связь этих двух параметров. Основными продуцентами TNF-α при РЯ являются опухолевые клетки и нейтрофилы. Сегодня является общепризнанным, что на определенном этапе канцерогенеза нейтрофилы начинают играть про-опухолевую роль [44]. Возможно, количество ЦОК при РЯ – один из определяющих факторов смены фенотипа нейтрофилов с противо-на проопухолевый.
Гипотеза метастатических «почвы и семени», предложенная Стефаном Педжетом в 1889 г., состоит в том, что опухолевая клетка способна дать начало метастатическому очагу только в подходящих условиях микроокружения [45]. Микроокружение включает в себя клеточные (фибробласты, макрофаги и др.) и внеклеточные компоненты (матрикс, цитокины, молекулы адгезии и др.), вступающие в сложное двустороннее взаимодействие с опу-
холевыми клетками. Считается, что микроокружение может провоцировать приобретение клеткой стволовых свойств [46], а привлекательность благоприятного микроокружения объясняет феномен самообсеменения опухоли, когда ЦОКи возвращаются в первичный опухолевый очаг и начинают пролиферировать в нем [47]. При этом цитокины оказывают влияние как на агрессивность «семени», так и на благоприятность «почвы» [48–50].
Заключение. Полученные результаты позволяют предполагать корреляцию уровня ЦОК с сывороточным уровнем цитокинов TNF-α, HGF, IL-10, IL-18, IL-8 у больных распространенным РЯ. При этом резкое и значимое возрастание уровня TNF-α при увеличении числа ЦОК может свидетельствовать о смене фенотипов TNF-продуцирующих клеток при РЯ.
Работа выполнена в рамках гранта РФФИ 19-315-50012.
Список литературы Цитокиновый статус сыворотки крови больных раком яичников с различным уровнем циркулирующих опухолевых клеток
- Judson P.L., Geller M.A., Bliss R.L., Boente M.P., Downs L.S., Argenta P.A. Preoperative detection of peripherally circulating cancer cells and its prognostic significance in ovarian cancer. Gynecologic oncology. 2003; 91 (2): 389-394.
- Pearl M.L., Zhao Q., Yang J., Dong H., Tulley S., Zhang Q. Prognostic analysis of invasive circulating tumor cells (iCTCs) in epithelial ovarian cancer. Gynecologic oncology. 2014; 134 (3): 581-590.
- Micalizzi D.S., Maheswaran S., Haber D.A. A conduit to metastasis: circulating tumor cell biology. Genes Dev. 2017; 31 (18): 1827-1840.
- Aceto N., Bardia A., Miyamoto D.T. Circulating tumor cell clusters are oligoclonal precursors of breast cancer metastasis. cell. 2014; 158 (5): 1110-1122.
- Klein C.A. Framework models of tumor dormancy from patient-derived observations. Current Opinion in Genetics & Development. 2011; 21 (1): 42-49.
- Bonnomet A., Syne L., Brysse A. A dynamic in vivo model of epithelial-to-mesenchymal transitions in circulating tumor cells and metastases of breast cancer. Oncogene. 2012; 31 (33): 3741-3753.
- Kim M.Y., Oskarsson T., Acharyya S., Nguyen D.X., Zhang X.H., Norton L., Massague J. Tumor self-seeding by circulating cancer cells. cell. 2009; 139 (7): 1315-1326.
- Skovierova H., Okajcekova T., Strnadel J., Vidomanova E., Halasova E. Molecular regulation of epithelial-to-mesenchymal transition in tumorigenesis (Review). Int. J. Mol. Med. 2018; 41 (3): 1187-1200.
- Moffitt L., Karimnia N., Stephens A., Bilandzic M. Therapeutic Targeting of Collective Invasion in Ovarian Cancer. Int. J. Mol. Sci. 2019; 20 (6): 1466.
- Hudson L.G., Zeineldin R., Stack M.S. Phenotypic plasticity of neoplastic ovarian epithelium: unique cadherin profiles in tumor progression. Clin. Exp. Metastasis. 2008; 25 (6): 643-655.
- Blassl C., Kuhlmann J.D., Webers A., Wimberger P., Fehm T., Neubauer H. Gene expression profiling of single circulating tumor cells in ovarian cancer - Establishment of a multi-marker gene panel. Mol. Oncol. 2016; 10 (7): 1030-1042.
- Strauss R., Li Z.Y., Liu Y. Analysis of epithelial and mesenchymal markers in ovarian cancer reveals phenotypic heterogeneity and plasticity [published correction appears in PLoS One. 2011; 6 (2)]. PLoS One. 2011; 6 (1): e16186.
- Trinchieri G. Cancer and inflammation: an old intuition with rapidly evolving new concepts. Ann. Rev. Immunol. 2012; 30: 677-706.
- Klymenko Y., Nephew K.P. Epigenetic Crosstalk between the Tumor Microenvironment and Ovarian Cancer Cells: A Therapeutic Road Less Traveled. Cancers (Basel). 2018; 10 (9): 295.
- Li Y.C., Zou J.M., Luo C., Shu Y., Luo J., Qin J., Wang Y., Li D., Wang S.S., Chi G., Guo F., Zhang G.M., Feng Z.H. Circulating tumor cells promote the metastatic colonization of disseminated carcinoma cells by inducing systemic inflammation. Oncotarget. 2017; 8 (17): 28418-28430.
- Ning Y., Cui Y., Li X., Cao X., Chen A., Xu C., Cao J., Luo X. Co-culture of ovarian cancer stem-like cells with macrophages induced SKOV3 cells stemness via IL-8/STAT3 signaling. Biomed. Pharmacother. 2018; 103: 262-271.
- Yung M.M., Tang H. W., Cai P.C. GRO-a and IL-8 enhance ovarian cancer metastatic potential via the CXCR2-mediated TAK1/NFkB signaling cascade. Theranostics. 2018; 8 (5): 1270-1285.
- House C.D., Jordan E., Hernandez L. NFkB Promotes Ovarian Tumorigenesis via Classical Pathways That Support Proliferative Cancer Cells and Alternative Pathways That Support ALDH+ Cancer Stemlike Cells. Cancer Res. 2017; 77 (24): 6927-6940.
- Zhai J., Shen J., Xie G., Wu J., He M., Gao L., Zhang Y., Yao X., Shen L. Cancer-associated fibroblasts-derived IL-8 mediates resistance to cisplatin in human gastric cancer. Cancer Lett. 2019; 454: 37-43.
- Wang Y., Li L., Guo X., Jin X., Sun W., Zhang X., Xu R.C. Interleukin-6 signaling regulates anchorage-independent growth, proliferation, adhesion and invasion in human ovarian cancer cells. Cytokine. 2012; 59 (2): 228-36.
- Stronach E.A., Cunnea P., Turner C. The role of interleukin-8 (IL-8) and IL-8 receptors in platinum response in high grade serous ovarian carcinoma. Oncotarget. 2015; 6 (31): 31593-31603.
- Sanguinete M.M.M., Oliveira P.H., Martins-Filho A., Micheli D.C., Tavares-Murta B.M., Murta E.F.C., Nomelini R.S. Serum IL-6 and IL-8 Correlate with Prognostic Factors in Ovarian Cancer. Immunol. Invest. 2017; 46 (7): 677-688.
- Huh S.J., Liang S., Sharma A., Dong C., Robertson G.P. Transiently entrapped circulating tumor cells interact with neutrophils to facilitate lung metastasis development. Cancer Res. 2010; 70 (14): 6071-6082.
- Lohmann A.E., Dowling R.J.O., Ennis M., Amir E., Elser C., Brezden-Masley C., Vandenberg T., Lee E., Fazaee K., Stambolic V., Goodwin P.J., Chang M.C. Association of Metabolic, Inflammatory, and Tumor Markers With Circulating Tumor Cells in Metastatic Breast Cancer. JNCI Cancer Spectr. 2018; 2 (2): pky028.
- Konig A., Vilsmaier T., Rack B., Friese K., Janni W., Jeschke U., Andergassen U., Trapp E., Juckstock J., Jdger B., Alunni-Fabbroni M., Friedl T., Weissenbacher T., Success Study Group. Determination of Interleukin-4, -5, -6, -8 and -13 in Serum of Patients with Breast Cancer Before Treatment and its Correlation to Circulating Tumor Cells. Anticancer Res. 2016; 36 (6): 3123-3130.
- Kwon Y., Godwin A.K. Regulation of HGF and c-MET Interaction in Normal Ovary and Ovarian Cancer. Reprod. Sci. 2017; 24 (4): 494-501.
- Dorayappan K.D.P., Gardner M.L., Hisey C.L., Zingarelli R.A., Smith B.Q., Lightfoot M.D.S., Gogna R., Flannery M.M., Hays J., Hansford D.J., Freitas M.A., Yu L., Cohn D.E., Selvendiran K. A microfluidic chip enables isolation of exosomes and establishment of their protein profiles and associated signaling pathways in ovarian cancer. Cancer Res. 2019; 79 (13): 3503-3513.
- Спирина Л.В., Кондакова И.В., Клишо Е.В., Какурина Г.В., Шишкин Д.А. Металлопротеиназы как регуляторы неоангиогенеза в злокачественных новообразованиях. Сибирский онкологический журнал. 2007; 1: 67-71.
- Yasuda K., Nakanishi K., Tsutsui H. Interleukin-18 in Health and Disease. Int. J. Mol. Sci. 2019; 20 (3): 649.
- Carbotti G., Barisione G., Orengo A.M., Brizzolara A., Airoldi I., Bagnoli M., Pinciroli P., Mezzanzanica D., Centurioni M.G., Fabbi M., Ferrini S. The IL-18 antagonist IL-18-binding protein is produced in the human ovarian cancer microenvironment. Clin. Cancer Res. 2013; 19 (17): 4611-4620.
- Orengo A.M., Fabbi M., Miglietta L., Andreani C., Bruzzone M., Puppo A., Cristoforoni P., Centurioni M.G., Gualco M., Salvi S., Boccardo S., Truini M., Piazza T., Canevari S., Mezzanzanica D., Ferrini S. Interleukin (IL)-18, a biomarker of human ovarian carcinoma, is predominantly released as biologically inactive precursor. Int. J. Cancer. 2011; 129 (5): 1116-1125.
- loana Braicu E., Mustea A., Toliat M.R., Pirvulescu C., Konsgen D., Sun P., Nurnberg P., Lichtenegger W., Sehouli J. Polymorphism of IL-1alpha, IL-1beta and IL-10 in patients with advanced ovarian cancer: results of a prospective study with 147 patients. Gynecol. Oncol. 2007; 104 (3): 680-685.
- Takaishi K., Komohara Y., Tashiro H., Ohtake H., Nakagawa T., Katabuchi H., Takeya M. Involvement of M2-polarized macrophages in the ascites from advanced epithelial ovarian carcinoma in tumor progression via Stat3 activation. Cancer Sci. 2010; 101 (10): 2128-2136.
- Szczerba B.M., Castro-Giner F., Vetter M., Krol I., Gkountela S., Landin J., Scheidmann M.C., Donato C., Scherrer R., Singer J., Beisel C., Kurzeder C., Heinzelmann-Schwarz V., Rochlitz C., Weber W.P., Beerenwinkel N., Aceto N. Neutrophils escort circulating tumour cells to enable cell cycle progression. Nature. 2019; 566 (7745): 553-557.
- Lau T.S., Chan L.K., Wong E.C., Hui C. W., Sneddon K., Cheung T.H., Yim S.F., Lee J.H., Yeung C.S., Chung T.K., Kwong J. A loop of cancer-stroma-cancer interaction promotes peritoneal metastasis of ovarian cancer via TNFa-TGFa-EGFR. Oncogene. 2017; 36 (25): 3576-3587.
- Szlosarek P.W., Grimshaw M.J., Kulbe H., Wilson J.L., Wilbanks G.D., Burke F., Balkwill F.R. Expression and regulation of tumor necrosis factor alpha in normal and malignant ovarian epithelium. Mol. Cancer Ther. 2006; 5: 382-390.
- Piura B., Medina L., Rabinovich A., Dyomin V., Levy R.S., Huleihel M. Distinct expression and localization of TNF system in ovarian carcinoma tissues: possible involvement of TNF-alpha in morphological changes of ovarian cancerous cells. Anticancer Res. 2014; 34: 745-752.
- Kulbe H., Thompson R., Wilson J.L. The inflammatory cytokine tumor necrosis factor-alpha generates an autocrine tumor-promoting network in epithelial ovarian cancer cells. Cancer Res. 2007; 67 (2): 585-592.
- Geng Y., Chandrasekaran S., Hsu J. W., Gidwani M., Hughes A.D., King M.R. Phenotypic switch in blood: effects of pro-inflammatory cytokines on breast cancer cell aggregation and adhesion. PLoS One. 2013; 8 (1): e54959.
- Reymond N., d'Agua B.B., Ridley A.J. Crossing the endothelial barrier during metastasis. Nature Reviews Cancer. 2013; 13 (12): 858-870.
- Evani S.J., Prabhu R.G., Gnanaruban V., Finol E.A., Ramasubramanian A.K. Monocytes mediate metastatic breast tumor cell adhesion to endothelium under flow. FASEB J. 2013; 27 (8): 3017-3029.
- Trabert B., Pinto L., Hartge P., Kemp T., Black A., Sherman M.E., Brinton LA., Pfeiffer R.M., Shiels M.S., Chaturvedi A.K., Hildesheim A., Wentzensen N. Pre-diagnostic serum levels of inflammation markers and risk of ovarian cancer in the prostate, lung, colorectal and ovarian cancer (PLCO) screening trial. Gynecol. Oncol. 2014; 135 (2): 297-304.
- Elizabeth M. Poole, I-Min Lee, Paul M. Ridker, Julie E. Buring, Susan E. Hankinson, Shelley S. Tworoger. A Prospective Study of Circulating C-Reactive Protein, Interleukin-6, and Tumor Necrosis Factor a Receptor 2 Levels and Risk of Ovarian Cancer. Am. J. Epidemiol. 2013; 178 (8): 1256-1264.
- Treffers L.W., Hiemstra I.H., Kuijpers T.W., van den Berg T.K., Matlung H.L. Neutrophils in cancer. Immunol. Rev. 2016; 273 (1): 312-328.
- Fidler I.J., Poste G. The "seed and soil" hypothesis revisited. Lancet Oncol. 2008; 9 (8): 808.
- Виноградова Т.В., Чернов И.П., Монастырская Г.С., Кондратьева Л.Г., Свердлов Е.Д. Раковые стволовые клетки: пластичность против терапии. Acta Naturae (русскоязычная версия). 2015; 4 (27): 52-63.
- Liu Q., Zhang H., Jiang X., Qian C., Liu Z., Luo D. Factors involved in cancer metastasis: a better understanding to "seed and soil" hypothesis. Mol. Cancer. 2017; 16 (1): 176.
- Xu Z., Jiang Y., Steed H., Davidge S., Fu Y. TGFp and EGF synergistically induce a more invasive phenotype of epithelial ovarian cancer cells. Biochem. Biophys. Res. Commun. 2010; 401 (3): 376-381.
- Yeung T.L., Leung C.S., Wong K.K. TGF-P modulates ovarian cancer invasion by upregulating CAF-derived versican in the tumor microenvironment. Cancer Res. 2013; 73 (16): 5016-5028.
- Lo C. W., Chen M.W., Hsiao M., Wang S., Chen C.A., Hsiao S.M., Chang J.S., Lai T.C., Rose-John S., Kuo M.L., Wei L.H. IL-6 trans-signaling in formation and progression of malignant ascites in ovarian cancer. Cancer Res. 2011; 71 (2): 424-434.


