Omalizumab in Severe Asthma: A Case Report
Author: Begimova S., Abylkasymova A., Izmailovich M., Knaus A., Shaikhina Z., Muratkyzy A., Ykhlassova M.
Journal: Juvenis scientia @jscientia
Section: Клинические случаи
Article in issue: 6 т.11, 2025.
Free access
This article presents a clinical case of a 56-year-old female patient with severe uncontrolled persistent asthma of mixed phenotype, characterized by poor disease control despite optimized background therapy and biologic treatment. The patient had a long history of asthma with progressive respiratory symptoms, frequent exacerbations, dependence on systemic corticosteroids, and development of mild respiratory failure (grade I). Particular attention is given to the use of omalizumab, an anti-IgE monoclonal antibody, administered according to the patient’s total IgE level and body weight. Although initial improvement in clinical symptoms and asthma control questionnaire scores was observed, the therapeutic response was not sustained, and disease control subsequently deteriorated. The report summarizes objective findings, including pulmonary function test results, laboratory markers of allergic and eosinophilic inflammation, bronchoscopy findings, sputum cytology, and chest computed tomography demonstrating chronic airway inflammation and progressive fibrotic remodeling of lung tissue. This case highlights the heterogeneity of severe asthma and illustrates the limitations of anti-IgE therapy in patients with mixed inflammatory phenotypes or non-IgE-mediated mechanisms of disease. The findings emphasize the importance of individualized treatment strategies, continuous monitoring, and consideration of alternative biologic therapies in patients with severe uncontrolled asthma.
Asthma, severe asthma, asthma control, biologic therapy, omalizumab, case report
Short address: https://sciup.org/14134777
IDR: 14134777 | DOI: 10.32415/jscientia_2025_11_6_32-39
Опыт применения омализумаба при тяжёлой бронхиальной астме: клинический случай
В статье представлен клинический случай пациентки 56 лет с тяжёлой неконтролируемой персистирующей бронхиальной астмой смешанного типа, характеризующейся недостаточным контролем заболевания, несмотря на проведение оптимизированной базисной и биологической терапии. Описана длительная история заболевания с прогрессированием симптомов, частыми обострениями, необходимостью системных глюкокортикостероидов и развитием дыхательной недостаточности I степени. Особое внимание уделено применению омализумаба как анти-IgE биологической терапии, назначенной в соответствии с уровнем общего IgE и массой тела пациентки. Несмотря на временное улучшение клинических показателей и баллов по опросникам контроля астмы, в дальнейшем отмечалась утрата терапевтического эффекта и возврат симптомов к исходному уровню. В статье представлены объективные данные, включая показатели функции внешнего дыхания, лабораторные маркеры аллергического и эозинофильного воспаления, результаты бронхоскопии, микроскопии мокроты и компьютерной томографии органов грудной клетки, подтверждающие хроническое воспаление и прогрессирующее фиброзное ремоделирование лёгочной ткани. Представленный случай подчёркивает гетерогенность бронхиальной астмы и ограничения анти-IgE терапии при смешанных и не-IgE-опосредованных фенотипах заболевания, а также необходимость персонализированного, фенотип-ориентированного подхода и динамического пересмотра терапевтической стратегии у пациентов с тяжёлым течением астмы.
Text of the scientific article Omalizumab in Severe Asthma: A Case Report
Bronchial asthma (BA) is a chronic inflammatory airway disease characterized by variable airflow limitation and bronchial hyperresponsiveness. This condition affects approximately 5% of the global population, necessitating precise diagnosis based on objective functional parameters, regular symptom monitoring, and evaluation of exacerbation risks [1]. In Kazakhstan, the prevalence of asthma has shown a steady upward trend. While there were an average of 93.4 cases per 100,000 citizens in 2017, this figure increased to 97.2 in 2018, 102.8 in 2019, and 126.1 in 2020 [2].
Airway remodeling, defined as structural changes in the bronchial walls, including extensive epithelial damage, hypertrophy of airway smooth muscle, collagen deposition, and subepithelial fibrosis, represents a key pathological feature of asthma [3].
This clinical case highlights critical aspects of managing severe asthma using omalizumab, a monoclonal anti-IgE antibody. However, despite the integration of this therapy, the treatment outcomes were unsatisfactory, presenting a significant clinical challenge that warrants further investigation.
Omalizumab is a humanized monoclonal anti-IgE antibody that binds to circulating IgE, preventing its interaction with high-affinity receptors on mast cells and basophils, thereby reducing IgE-mediated inflammatory responses [4]. Despite these advantages, omalizumab therapy does not always yield the desired outcomes. Suboptimal response may be associated with individual immunological characteristics, comorbid conditions, persistent exposure to triggering factors, or predominance of non–IgE-mediated inflammatory mechanisms.
This case further illustrates the importance of a comprehensive, phenotype-oriented approach to asthma management, integrating pharmacologic therapy with control of environmental triggers and comorbid conditions.
CLINICAL CASE
In May 2023, a 56-year old female patient presented with persistent dyspnea at rest and on minimal exertion, expiratory wheezing, daily asthma attacks, nocturnal symptoms occurring 3–4 times per week, and frequent use of short-acting ₽2-agonists (up to 6–8 inhalations per day). Asthma Control Test (ACT) score was 6 points, and Asthma Control Questionnaire (ACQ) score was 5.0, indicating severe uncontrolled asthma. The patient reported reduced exercise tolerance and inability to discontinue systemic corticosteroids without worsening symptoms.
On physical examination, the patient was in moderate respiratory distress. Respiratory rate was 22 breaths per minute, oxygen saturation was 95% on room air. Auscultation revealed diffuse expiratory wheezing over both lung fields with prolonged expiration. No peripheral edema, cyanosis, or signs of cardiac decompensation were observed.
Anamnesis Morbi
Bronchial asthma was first diagnosed at the age of 38 by a pulmonologist based on characteristic clinical symptoms (recurrent wheezing, dyspnea, cough), spirometry demonstrating airflow limitation with partial reversibility, and a positive response to inhaled corticosteroid therapy. Initially, treatment with inhaled corticosteroids (ICS) led to clinical improvement. However, from 2019 onward, the patient experienced progressive worsening of symptoms with increased frequency of exacerbations. Therapy was escalated according to standard stepwise asthma management, including high-dose ICS combined with long-acting p2 -agonists (LABA).
In 2020, during a severe exacerbation, maintenance therapy included budesonide/formoterol (640/18 ^g daily) and short-acting bronchodilators as needed. Despite high-dose ICS/LABA therapy, asthma control remained inadequate. In June 2021, the patient developed a severe exacerbation requiring systemic glucocorticosteroids (prednisolone 30 mg daily with gradual tapering). Attempts to discontinue oral corticosteroids resulted in rapid clinical deterioration with recurrence of dyspnea and bronchial obstruction. In September 2021, the patient contracted COVID-19 infection without radiologically confirmed pneumonia. After this period, respiratory symptoms intensified and became more difficult to control. Between 2021 and 2023, the patient required repeated courses of systemic corticosteroids due to frequent exacerbations. She was hospitalized in the Pulmonology De- partment with a diagnosis of severe uncontrolled bronchial asthma and grade I respiratory failure (SpO2 94–95%).
Thus, despite optimized high-dose ICS/LABA therapy, addition of long-acting muscarinic antagonist (tiotropium), and repeated systemic corticosteroid courses, the disease remained uncontrolled, consistent with criteria for severe uncontrolled asthma according to international guidelines.
Allergological and Laboratory Evaluation
Allergological examination revealed persistently elevated total IgE levels: 279.5 IU/mL (December 2021), 485 IU/mL (September 2023), 426 IU/mL (February 2024), and 771.5 IU/mL (March 2024). Peripheral blood eosinophilia was documented, with a peak value of 25% (1.24 * 10 9 /L) in December 2023. Eosinophilic cationic protein (ECP) levels were elevated (80.6 ng/mL in August + 2023), indicating active eosinophilic inflammation.
Bronchial wash cytology (October 2023) demonstrated neutrophil predominance (60%), suggesting a mixed inflammatory phenotype with a significant neutrophilic component. The combination of elevated total IgE, peripheral eosinophilia, and neutrophil predominance in bronchial wash suggested a mixed inflammatory phenotype (type 2-high inflammation with a neutrophilic component), potentially influencing treatment response. The increase in total IgE observed during follow-up likely reflects the formation of IgE–omalizumab immune complexes and does not directly indicate treatment failure.
Pulmonary Function and Imaging Findings
Spirometry prior to biologic therapy showed persistent airflow limitation: FEV ! 38.8% predicted, FVC 64.4% predicted, FEV , /FVC 50.45%. These findings indicated severe obstructive ventilatory impairment.
Chest computed tomography (January 2020) revealed progressive fibrotic and interstitial changes consistent with airway remodeling and chronic inflammatory alterations. Follow-up CT (March 2024) demonstrated progression of interstitial and fibrotic remodeling, supporting the presence of structural airway changes contributing to persistent airflow limitation.
Initiation of Omalizumab Therapy
Given persistent severe uncontrolled asthma despite optimized Step 4–5 therapy, dependence on systemic corticosteroids, elevated total IgE levels within the therapeutic range for anti-IgE treatment, and frequent exacerbations, omalizumab therapy was initiated in May 2023. The dose (225 mg subcutaneously every two weeks) was determined according to the manufacturer’s dosing table based on body weight and baseline total IgE level.
Clinical Course After Initiation of Omalizumab
After initiation of omalizumab, a clinically significant improvement was observed within several months. ACT score increased from 6 to 22 (October 2023), and ACQ improved from 5.0 to 1.6, reflecting good asthma control. The frequency of exacerbations decreased, and systemic corticosteroids were temporarily discontinued. During follow-up, FEV t demonstrated only minimal improvement and remained below 50% predicted, indicating persistent severe airflow limitation despite clinical improvement. However, by April 2024, asthma control deteriorated again (ACT 8; ACQ 4.0), accompanied by worsening dyspnea and decline in lung function parameters. This loss of control occurred despite continued biologic therapy and stable maintenance treatment.
The subsequent loss of asthma control may reflect progressive airway remodeling and predominance of non–IgE-mediated inflammatory mechanisms, limiting the long-term effectiveness of anti-IgE therapy in this patient. No serious adverse events related to omali-zumab therapy were observed during the follow-up period. A summary of key clinical and laboratory parameters during follow-up is presented in Table 1.
A chronological summary of disease progression and therapeutic response is illustrated in Figure 1.
DISCUSSION
According to published data, omalizumab directly influences IgE-dependent reactions. Omalizumab is a humanized monoclonal antibody derived from a murine antibody. The final product consists of 95% human IgG protein and 5% of a fragment specifically targeting the Ce3 region of human Ig E. Through this mechanism, omalizumab exerts a multifaceted effect
Table 1
Dynamics of Key Clinical and Laboratory Parameters During Follow-up
|
Parameter |
Before omalizumab (May 2023) |
After 5 months (Oct 2023) |
Follow-up (Mar–Apr 2024) |
|
ACT score |
6 |
22 |
8 |
|
ACQ score |
5.0 |
1.6 |
4.0 |
|
FEV i (% predicted) |
38.8 |
— |
42.1 |
|
FVC (% predicted) |
64.4 |
— |
47.2 |
|
Total IgE (IU/mL) |
279.5 |
485 |
771.5 |
|
Eosinophils (%) |
— |
25% |
7.1% |
|
ECP (ng/mL) |
— |
80.6 |
40.1 |
Abbreviations: ACT — Asthma Control Test; ACQ — Asthma Control Questionnaire; ECP — eosinophilic cationic protein; FEV i — forced expiratory volume in 1 second; FVC — forced vital capacity.
2019 2021 Oct 2023
Diagnosis of Recurrent exacerbations Clinical Improvement bronchial asthma Systemic corticosteroids ACT 22
2020 Apr 2024
Escalation to May 2023 Loss of control high-dose ICS/LABA Omalizumab initiated ACT 8
Fig. 1. Clinical timeline illustrating disease progression and response to omalizumab therapy in a patient with severe asthma.
on the allergic inflammatory cascade.
The clinical case described in this article illustrates the challenges in managing severe asthma [5]. Due to the severity of the patient’s condition and its resistance to Step 4 therapy, omalizumab was incorporated into the treatment regimen at a dose of 225 mg every two weeks.
From the onset of treatment, improvements were observed: the frequency of asthma attacks decreased, dyspnea episodes were reduced, and coughing became more productive. These clinical changes were reflected in improved ACT and ACQ scores. However, over time, the patient’s symptoms and pulmonary function metrics regressed to baseline levels. This is evident from spirometry data, where FVC decreased from 64.4% on September 5, 2023, to 47.2% on March 21, 2024, while FEVi showed a slight improve ment from 38.8% to 42.1%. Similarly, the FEVi/FVC ratio declined from 50.45% to 47.39%.
This case does not dispute the overall efficacy of omalizumab but demonstrates its limitations in managing severe asthma. The drug’s mechanism, highly specific to IgE, may not fully address cases where mixed inflammatory phenotypes or non-IgE-mediated mechanisms predominate. Such cases highlight the complexity of asthma as a heterogeneous disease and underscore the necessity of tailoring treatments to individual patient profiles.
The limited and transient response to omalizumab observed in this patient may be explained by a combination of factors, including a mixed inflammatory phenotype, persistent neutrophilic airway inflammation, airway remodeling, and comorbid conditions, as summarized in Figure 2.
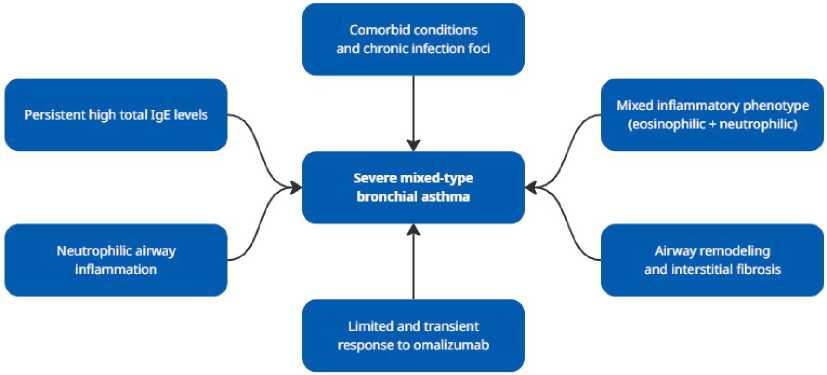
Fig. 2. Proposed mechanisms explaining the limited and transient response to omalizumab in severe asthma with a mixed inflammatory phenotype.
In conclusion, while omalizumab remains a cornerstone in the management of IgE-mediated asthma, this case emphasizes the importance of exploring alternative therapeutic options. These may include other biologics targeting IL-5 or IL-4/IL-13 pathways or novel treatments addressing mixed inflammatory mechanisms. Continuous monitoring, patient-specific adjustments to therapy and further research into asthma phenotypes are essential to optimize outcomes for patients with severe forms of the disease.
Accumulated evidence indicates that omalizumab improves asthma control, reduces exacerbation frequency, and lowers healthcare utilization in severe allergic asthma. However, treatment response is not uniform across all patients. Among the proposed predictors of response, elevated blood eosinophils and FeNO appear to be the most clinically informative, whereas baseline total IgE alone has shown limited predictive utility [6].
A multicenter retrospective analysis comparing du-pilumab, omalizumab, and mepolizumab demonstrated differences in exacerbation rates among these biologics, with dupilumab showing the most pronounced reduction in annual exacerbations. Improvements in FEV i were modest and comparable across treatment groups, highlighting that reduction in exacerbations may be a more sensitive indicator of therapeutic effectiveness than lung function parameters alone [7].
CONCLUSION
Patients with severe uncontrolled bronchial asthma represent a large and clinically challenging group requiring specialized treatment. These individuals often require higher doses of baseline therapy and frequent emergency medical interventions. Moreover, such patients frequently exhibit partially reversible or irreversible bronchial obstruction due to significant airway remodeling, which complicates diagnosis and differentiation from chronic obstructive pulmonary disease.
In the presented clinical case, despite the initially favorable clinical response to omalizumab, the therapeutic effect proved to be transient and was not sustained during long-term follow-up. Omalizumab has demonstrated efficacy even in patients with poorly reversible bronchial obstruction and reduced lung function (FEV t < 50%). Additionally, studies have shown that omalizumab reduces the need for oral corticosteroids in patients with moderate to severe atopic asthma, highlighting its value in managing complex cases. These findings emphasize the need for dynamic reassessment of asthma phenotype and therapeutic strategy in patients demonstrating transient response to biologic therapy.
This case underscores the importance of individualized, phenotype-oriented selection of biologic therapy and highlights the need for continuous evaluation of treatment effectiveness in patients with complex asthma phenotypes.
Funding: The authors declare that no funding was received.
Conflict of Interest: The authors declare no conflict of interest.
Ethics Statement: Written informed consent was obtained from the patient for the publication of this clinical case and any accompanying images. The patient’s anonymity and confidentiality were fully preserved in accordance with ethical standards.
Author Contributions: Conceptualization, M.I. and S.B.; methodology, M.I. and A.A.; validation, M.I. and A.M.; formal analysis, M.I., M.Y. and A.K.; investigation, A.A.; resources, S.B. and A.A.; data curation, S.B. and A.A.; writing – original draft preparation, S.B., A.A., M.I., A.K., Z.S., A.M. and M.Y.; writing – review and editing, S.B., A.A., M.I., A.K., Z.S., A.M. and M.Y.; supervision, M.I. and S.B.; project administration, S.B. and A.K. All authors have read and agreed to the published version of the manuscript.


