Трансплантация генетически модифицированных стволовых клеток, повышенно экспрессирующих терапевтические ростовые факторы, при лечении экспериментального инфаркта миокарда крыс
Автор: Коноплянников Михаил Анатольевич, Хайдер Х.Х., Ашраф М.
Журнал: Клиническая практика @clinpractice
Рубрика: Оригинальные исследования
Статья в выпуске: 3 (15), 2013 года.
Бесплатный доступ
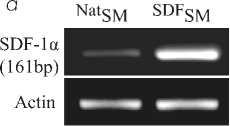
Мы проанализировали гипотезу, что одновременная трансгенная модификация трансплантируемых стволовых клеток 4-мя ростовыми факторами активирует разнообразные сигнальные механизмы для выживания введенных клеток, а также мобилизацию различных стволовых/прогениторных клеток для кардиогенеза при терапии инфаркта миокарда. Плазмиды, несущие гены, кодирующие IGF-1, VEGF, SDF-1α и HGF, были сконструированы и трансфицированы в скелетные миобласты (SM) крыс. Повышенная экспрессия ряда факторов в трансфицированных SM (TransSM) была подтверждена методами ПЦР, Вестерн-блоттинга (ВБ) и иммунофлюоресцентного окрашивания. Кондиционная среда от TransSM обладала цитопротекторными свойствами для кардиомиоцитов в условиях окислительного стресса, стимулировала миграцию клеток HUVEC, и усиливала ангиогенез клеток HUVEC in vitro. Интрамиокардиальная трансплантация 1,5×106 TransSM продемонстрировала обширную мобилизацию стволовых/прогениторных клеток в инфарктном миокарде и улучшала интегрирование TransSM в сердце. Экстенсивный неомиогенез и ангиогенез наблюдался через8 недель после трансплантации. Итак, одновременная активация разнообразных сигнальных механизмов благодаря повышенной экспрессии нескольких ростовых факторов вызывает массовую мобилизацию и хоминг как стволовых/прогениторных клеток из костного мозга и циркулирующих в крови, так и резидентных кардиальных стволовых клеток, что приводит к ускоренной репарации инфарктного миокарда.
Стволовые клетки, инфаркт миокарда, трансгенная модификация, трансплантация клеток, сердечно-сосудистая регенерация, паракринные факторы
Короткий адрес: https://sciup.org/14338459
IDR: 14338459
Transplantation of genetic modified stem cells over-expressing therapeutic growth factors for the treatment of experimental rat myocardial infarction
We tested the hypothesis that simultaneous transgenic overexpression of a select quartet of growth factors activates diverse signaling pathways for mobilization and participation of various stem/progenitor cells for cardiogenesis in the infarcted heart. IGF-1, VEGF, SDF-1α, and HGF plasmids were synthesized and transfected into rat skeletal myoblasts (SM). Overexpression of growth factors in transfected SM (TransSM) was confirmed by reverse transcription PCR, western blotting, and fluorescence immunostaining. The conditioned medium (CM) from TransSM was cytoprotective for cardiomyocytes following H 2O 2 treatment, promoted a higher transwell migration of HUVEC cells and in vitro tube formation. Intramyocardial transplantation of 1.5×10 6 TransSM in a rat model of acute myocardial infarction induced extensive mobilization of stem/progenitor cells into the infarcted heart on day 7 and improved integration of TransSM in the heart compared to NatSM. Extensive neomyogenesis and angiogenesis with resultant attenuation of infarct size and improvement in global heart function was observed at 8 weeks. In conclusion, simultaneous activation of diverse signaling pathways by overexpression of multiple growth factors caused massive mobilization and homing of stem/progenitor cells from peripheral circulation, the bone marrow, and the heart for accelerated repair of the infarcted myocardium.
Текст научной статьи Трансплантация генетически модифицированных стволовых клеток, повышенно экспрессирующих терапевтические ростовые факторы, при лечении экспериментального инфаркта миокарда крыс
Стратегия репарации миокарда с использованием трансплантации стволовых клеток является очень привлекательной из-за недостаточно эффективных собственных механизмов восстановления сердца после инфаркта [1]. Со времени первых трансплантаций с такими целями [2] достаточно часто использовались как скелетные миобласты (SM), так и стволовые клетки костного мозга (BMSC), в рамках I и II фаз клинических исследований [3, 4]. В свете полученных результатов и из-за сложности протекания патологического процесса в ишемизированном сердце, в настоящее время не существует общепринятого типа стволовых клеток, или общепринятой вспомогательной терапии стволовыми клетками, адекватно подходящих для репарации поврежденного миокарда. Такие типы клеток как BMSC, SM, эндотелиальные прогениторные клетки периферической крови (EPC) и резидентные кардиальные стволовые/прогениторные клетки продемонстрировали неплохой миогенный и васку-логенный потенциал в экспериментальных и клинических испытаниях. Кроме того, все эти типы клеток обладают характерными функциональными преимуществами. SM направленно дифференцируют в клетки скелетных мышц, волокна которых могут служить каркасом, поддерживающим ослабленный инфарктом миокард, предотвращают ремоделирование левого желудочка (ЛЖ), и улучшают диастолическую функцию сердца [5, 6], к тому же продуцируя ряд цитокинов [7]. BMSC секретируют различные ростовые факторы, осуществляющие паракринное действие, предотвращая расширение зоны инфаркта благодаря цитопротектор-ному эффекту и усилению ангиогенеза [8, 9]. Эндотелиальные прогениторные клетки периферической крови (EPC) демонстрируют вас- кулогенную активность и, кроме того, способны к адаптации кардиального фенотипа [10]. Наконец, резидентные кардиальные стволо-вые/прогениторные клетки вообще могут принимать фенотип всех типов клеток, необходимых для регенерации миокарда [11, 12].
Главной целью нашего исследования являлась разработка методологии и исследование механизмов терапевтического эффекта трансплантации стволовых клеток, повышенно экспрессирующих различные ростовые факторы (TransSM), что приводит к одновременной активации различных сигнальных путей в организме реципиента, влияющих на множественные функции регенерации миокарда, а именно – на выживание и пролиферацию клеток, индукцию щелевых контактов, секрецию паракринных факторов, а также мобилизацию BMSC, EPC и резидентных кардиальных стволовых клеток. С этой целью мы трансфицировали SM избранной группой ростовых факторов (IGF-1, VEGF, SDF-1a и HGF) и трансплантировали генетически модифицированные SM в инфарктное сердце. Несмотря на спорную проаритмогенную природу, мы выбрали SM в качестве клеток-носителей для доставки квартета указанных факторов в поврежденный миокард. Это связано, во-первых, с их доказанной способностью быть отличными носителями терапевтических генов [13]. Во-вторых, благодаря миогенной дифференцировке после приживления они служат в качестве каркаса, ослабляющего ремоделирование ЛЖ [5]. Мы предположили также, что IGF-1 усилит экспрессию Cx-43 в TransSM, что, в свою очередь, должно улучшить их приживление в зоне инфаркта миокарда.
Материалы и методы
Все экспериментальные процедуры выполнялись в соответствии с Руководством по ухо- ду и использованию лабораторных животных Национальных Институтов Здоровья, Бетезда, США (Guide for the Care and Use of Laboratory Animals, US National Institutes of Health, NIH Publication No. 85-23, 1996) и в соответствии с протоколом No. 06-03-13-03, одобренным Комитетом по уходу и использованию животных Университета Цинциннати, Огайо, США (Institutional Animal Care and Use Committee, University of Cincinnati). Все эксперименты in vitro были повторены 3 раза для обеспечения воспроизводимости данных.
Выделение и характеризация SM
Нативные SM (NatSM) были выделены из образцов биопсии скелетных мышц из нижних конечностей молодых крыс-самцов Fisher-344 и охарактеризованы проточной цитофлуори-метрией и иммунофлуоресцентным окрашиванием, как описывалось нами ранее [14].
Приготовление плазмид и трансфекция клеток
Плазмиды, кодирующие SDF-1a, HGF, IGF-1, и VEGF, были приготовлены, как описано в нашей работе [15]. Для трансфекции клетки были культивированы до 80%-90% конфлю-энтности, и по отдельности в каждой из 4 чашек Петри трансфицированы каждой из 4 плазмид (IGF-1, VEGF, SDF-1a, или HGF), используя катионный вектор Lipofectamine-2000 (Invitrogen). Комплексы липид-ДНК были приготовлены, используя соотношение ДНК (в мг) и Lipofectamine-2000 (в мл), равное 1:2,5; в базовой среде DMEM (Invitrogen). После 20минутной инкубации при комнатной температуре, трансфекционные комплексы были добавлены к клеткам в присутствии 10% фетальной телячьей сыворотки (FBS), аккуратно перемешаны и инкубированы в течение 8 часов при 37°C в CO2 инкубаторе. После этого удаляли трансфекционные комплексы, а клетки культивировали в среде DMEM, содержащей 10% FBS, в течение 72 часов. Затем объединяли все клетки из 4 чашек Петри в одной чашке Петри. Полученный пул клеток (TransSM) культивировали еще 24 часа при 37°C в CO2 инкубаторе, перед их использованием в in vitro и in vivo исследованиях.
ПЦР в реальном времени
Метод ПЦР в реальном времени был использован для количественного тестирования возможной экспрессии ряда факторов, усиливающих ангиогенез и выживаемость клеток, используя специально созданную нами ПЦР- панель генов таких факторов, на образцах, выделенных из сердечной ткани крыс через 7 дней после трансплантации.
Методы анализа миграции клеток и способности клеток к ангиогенезу in vitro
Кондиционная среда (CM) от TransSM и NatSM была использована для анализа миграции клеток HUVEC, методом транслуночной миграции ( in vitro transwell migration system), как было описано нами ранее [15]. Для анализа способности к ангиогенезу клеток HUVEC in vitro использовался метод формирования микротрубок (tube formation assay) [15].
Методы анализа гибели клеток
Для определения активности лактат-дегид-рогеназы (LDH) был использован набор LDH Assay kit (Sigma), согласно инструкции производителя. Программированная гибель клеток (апоптоз) была исследована методом TUNEL (Terminal dUTP nick-end labeling assay) с использованием набора In-Situ Cell Death Detection kit (Roche Diagnostics), согласно инструкции производителя. Количество апо-птозных клеток было рассчитано при микроскопическом анализе окрашенных клеток (TUNEL-положительных ядер клеток) в поле наблюдения (x400). Активность каспазы-3 (сaspase-3), индикатора апоптоза клеток, была исследована с использованием набора Caspase-3 Activity Assay Kit (Calbiochem), согласно инструкции производителя.
Образование щелевых контактов in vitro между TransSM и кардиомиоцитами и их функциональная оценка
Неонатальные кардиомиоциты были выделены из новорожденных (1-3 дневных) крысят, используя набор Neonatal Cardiomyocyte Isolation System (Worthington Biochemical) согласно инструкции производителя. Функциональная оценка щелевых контактов между TransSM или NatSM и кардиомиоцитами (оценки функции Cx-43) была осуществлена методом переноса красителя Lucifer Yellow (0,5 нг/мл; Sigma-Aldrich) [16].
Функциональный статус действия Cx-43 между соседними взаимодействующими клетками был охарактеризован степенью проникновения красителя от нагруженных красителем клеток в каждой клеточной популяции. Для оценки электрофизиологического сопряжения между TransSM и кардиомиоцитами в совместных культурах клетки были загружены Fluo-4 ацетоксиметиловым эфиром (зеленым флуоресцентным индикатором кальция, 6мM; Molecular Probes) и измерения были произведены согласно инструкции производителя, с использованием конфокального микроскопа (Carl Zeiss LSM-510), в сканирующем режиме, с длиной волны возбуждения – 488 нм, длиной волны эмиссии –510 нм.
Исследования in vivo
Модель инфаркта миокарда крыс и трансплантация SM
Экспериментальную модель острого инфаркта миокарда воспроизводили на молодых (6-8 недельных) самках крыс (Fisher-344) лигированием левой коронарной артерии, как описывалось ранее [17]. Животные были разделены по группам для проведения интрамио-кардиальных инъекций (4-5 уколов в сердце) в объеме 70 мкл физиологического раствора (бесклеточный контроль, группа-1); 1,5x10 6 NatSM (группа-2); 1,5x10 6 TransSM (группа-3). Для снижения болевых ощущений был использован Buprenex (0,1 мг/кг), в течение 24 часов после операции.
Для постмортемных исследований животные были эвтаназированы фенобарбиталом натрия, и тканевые образцы (сердце животного) были извлечены для дальнейшего анализа. Метод ПЦР был использован для количественного анализа гена sry для оценки степени выживаемости клеток через 7 дней после трансплантации (по 3 в каждой группе) [18].
Гистологические исследования
Для измерения размеров инфарктной зоны сердце было остановлено внутривенной инъекцией хлорида кадмия и фиксировано в 10% за-буференном формалине. Гистологические образцы толщиной 6 мкм были окрашены трихромом по Массону и проанализированы с использованием компьютерной программы ImageJ (version-1.6065; NIH).
Для анализа трансплантированных стволовых клеток было произведено иммуногистохимическое окрашивание образцов, с использованием первичных антител к зеленому флуоресцентному белку (GFP) (Abcam), и вторичных антител (Alexa Fluor-488, Molecular Probes). Плотность кровеносных сосудов была проанализирована иммуногистохимическим (ИГХ) окрашиванием с использованием первичных антител на von-Willebrand Factor-VIII (vWFactor-VIII) и гладкомышечный актин, далее подсчитывали число сосудов в поле зрения микроскопа (0,74 мм2) при увеличении x200. Другие первичные антитела, используемые нами в ИГХ исследованиях, включали Ki-67 (для анализа пролиферации клеток), десмин, кардиальный актинин, тяжелоцепочечный миозин, Cx-43 (для анализа миогенеза и интеграции новых миофибрилл с кардиомиоцитами хозяина), а также CD31, c-kit, CD44, CD45, и CXCR4 (для анализа стволо-вых/прогениторных клеток, мобилизуемых в инфарктный миокард). Список использованных антител представлен в таблице.
Таблица
Список использованных антител для Вестерн-блоттинга и ИГХ
|
Антитело |
Разведение |
Производитель |
|
Cardiac actin |
1:50 |
Sigma |
|
CD44 |
1:100 |
BD BioScience |
|
c-kit |
1:40 |
Chemicon |
|
Connexin-43 |
1:40 |
Santa Cruz |
|
Desmin |
1:50 |
Sigma |
|
HGF |
1:100 |
R&D Systems |
|
IGF-1 |
1:50 |
Santa Cruz |
|
pAkt |
1:50 |
Cell Signaling |
|
SDF-1a |
1:100 |
R&D Systems |
|
Smooth muscle actin |
1:50 |
Sigma |
|
vWillebrand Factor |
1:100 |
Dako |
|
VEGF |
1:50 |
Santa Cruz |
Исследование функции сердца
Исследование функции сердца было произведено через 8 недель после трансплантации клеток (6 животных в группе-1, по 8 животных в группе-2 и в группе-3) [17]. Функция сердца была определена трансторакальной эхокардиографией, используя ультразвуковой диагностический аппарат HDI/5000 SONOS CT (Philips). Показатели сократительной функции, включая фракцию выброса ЛЖ (LVEF) и фракцию укорочения ЛЖ (LVFS), были рассчитаны, используя следующие формулы:
LVFS = {(LVEDD - LVESD)/LVEDD} x 100
LVEF = (LVEDD 3 - LVESD 3 )/LVEDD 3 x 100
(где LVESD – конечно-систолический диаметр ЛЖ; LVEDD – конечно-диастолический диаметр ЛЖ; конечные результаты были выражены в процентах).
Статистический анализ
Статистический анализ был выполнен при помощи программы Statview-5.0. Все результаты были представлены как среднее значение ± стандартная ошибка, для сравнения значений использовался критерий Стьюдента и дисперсионный анализ (ANOVA). Статистическая достоверность P < 0,05 рассматривалась как значимая.
Результаты
Исследования in vitro
Выделение и трансфекция SM
SM были успешно выделены из образцов скелетной мышцы, полученных из крыс-самцов Fisher-344, используя стандартный протокол [14]. Анализ методом проточной цитометрии (FACS) продемонстрировал, что полученная культура SM более чем на 90% состояла из клеток, экспрессирующих десмин (Рис. 1a), и на 62% – из клеток, экспрессирующих CD56.
Плазмиды, несущие гены, кодирующие IGF-1, VEGF, SDF-1α и HGF, были сконструированы и
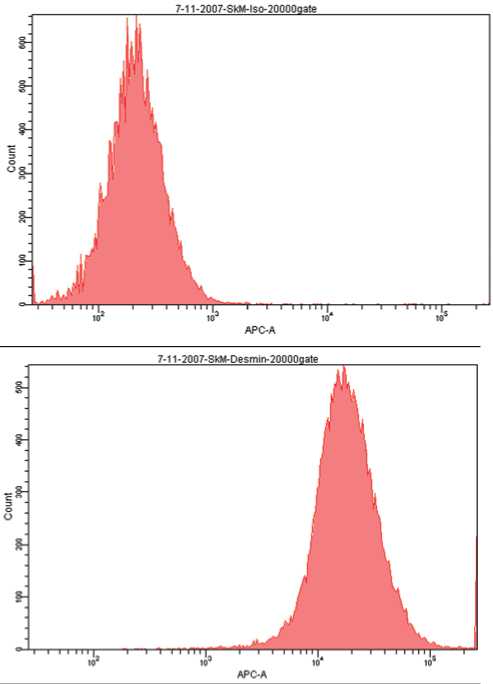
Рис. 1a. Проточная цитофлуориметрия продемонстрировала, что очищенная культура SM была положительна на маркер десмин более чем на 90%.
Верхняя диаграмма – изотипический контроль, нижняя диаграмма – окрашивание на десмин.
трансфицированы в скелетные миобласты (SM). Экспрессия ростовых факторов в трансфицированных SM (TransSM) была повышена, что подтверждено методами иммунофлуоресцентного окрашивания (Рис.1b), ПЦР с обратной транскрипцией и Вестерн-блоттинга (Рис. 2 a, b).
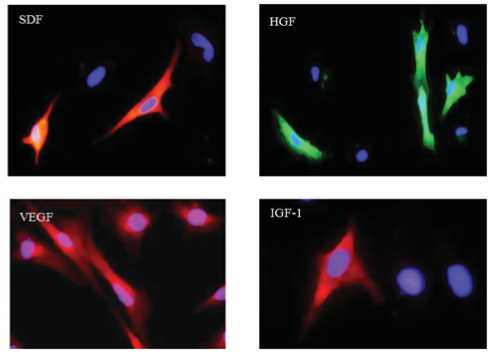
Рис. 1b. Флуоресцентное иммуноокрашивание, подтверждающее трансгенную экспресию VEGF, SDF-1a, IGF-1, и HGF, через 72 часа после трансфекции (увеличение х400). NatSM использовали в качестве контроля.
VEGF (226bp)
Actin
b
SDF-la (lOKd)
Actin
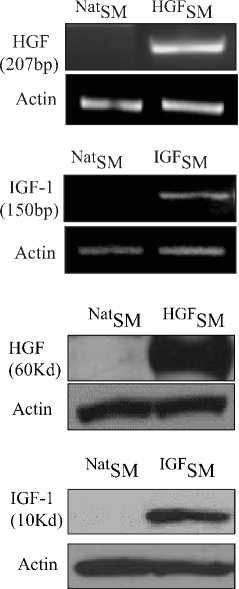
Рис. 2. Экспрессия трансфицированных ростовых факторов в TransSM in vitro, охарактеризованная методами: a) ПЦР с обратной транскрипцией (RT-PCR) и b) Вестерн-иммуно-блоттинга (Western blots), подтвердивших трансгенную экспрессию VEGF, SDF-1a, IGF-1, и HGF, через 72 часа после трансфекции (в сравнении с NatSM), NatSM – нативные скелетные миобласты; TransSM – трансфицированные SM.
NatSM VEGFSM

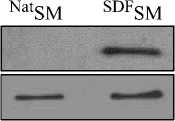
NatSM VEGFSM
VEGF (21Kda)
Actin

Профиль генной экспрессии TransSM
Кроме многократного повышения в экспрессии генов 4 ростовых факторов, метод ПЦР по- казал достоверное увеличение экспрессии ряда факторов, усиливающих ангиогенез и выживаемость клеток, в TransSM (Рис. 3).
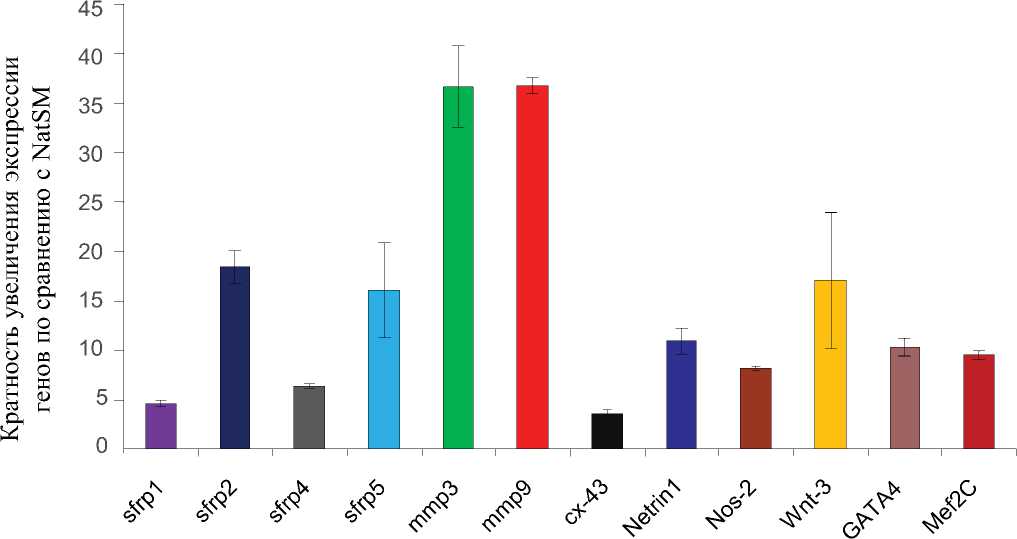
Рис. 3. Активация множественных сигнальных путей в TransSM, продемонстрированная методoм ПЦР с обратной транскрипцией (RT-PCR, данные 3-x экспериментов), показавших активацию различных сигнальных молекул, транскрипционных факторов, секретируемых и несекретируемых факторов, выделение которых повышено в TransSM по сравнению с контрольными NatSM.
Для детектирования транскрипционных факторов образцы клеточного лизата были фракционированы на цитоплазматические и ядерные фракции с использованием набора реагентов для ядерной и цитоплазматической экстракции (NE-PER; ThermoFisher Scientific), следуя инструкциям производителя. NatSM – нативные скелетные миобласты; TransSM – трансфицированные SM.
Значительное увеличение было продемонстрировано для Sfrp-1, Sfrp-2, Sfrp-4, Sfrp-5 (secreted frizzled related proteins), netrin-1, iNOS (индуцибельная синтаза оксида азота), MMP-3 и MMP-9 (матриксные металлопротеиназы). Мы также наблюдали повышенную экспрессию сердечных транскрипционных факторов, GATA-4 и Mef-2c, а также значительно лучшую экспрессию Cx-43.
Вестерн-блоттинг показал активацию сигнальных молекул, регулирующих выживание клеток, а также важнейших транскрипционных факторов: pAkt, p44/42MAPK, pStat3, HIF-1α, pPKC (pan), в TransSM, по сравнению с NatSM. Другой важнейшей особенностью TransSM было значительное повышение уровня индукции Cx-43, ключевого фактора электромеханического сопряжения прижившихся трансплантированных клеток с кардиомиоцитами хозяина, продемонстрированное на генном (рис. 3) и белковом уровне.
Цитопротекторный и ангиогенный эффекты кондиционной среды от TransSM
Кондиционная среда от TransSM (TransSM Condition Medium, TransCM), богатая цитокинами и ростовыми факторами, продемонстрировала цитопротекторное действие, предотвратив гибель культивируемых кардиомиоцитов человека H9C2 вследствие окислительного стресса. Выброс лактатдегидрогеназы (LDH), маркера клеточного повреждения, был значительно понижен в H9C2 клетках, предварительно инкубированных с TransCM (P < 0,01 vs. интактных H9C2), c последующим воздействием 100 мМ H2O2, индуцирующей окислительный стресс. Эти результаты были подтверждены методом TUNEL, который продемонстрировал, в тех же условиях окислительного стресса, при предварительной инкубации с TransCM, пониженное количество апоптотических (TUNEL+) H9C2 клеток (P < 0,01 vs. интактных H9C2 и NatCM-обработанных клеток (Рис. 4а).
0 —=—
Контроль
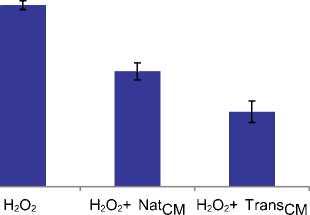
Рис. 4а. Цитопротекторные и ангиогенные эффекты TransCM in vitro и экспрессия белков щелевых контактов в TransSM. TUNEL – анализ показал уменьшение TUNEL + (апоптотических) клеток, обработанных TransCM, по сравнению с клетками, обработанными NatCM (P < 0,01 по сравнению с NatCM).
Кроме того, в аналогичных условиях пред-инкубация с TransCM приводила к понижению активности каспазы-3 (P < 0,01 vs. NatSM-об-работанных клеток.
TransCM также способствовала миграции эндотелиальных клеток HUVEC, анализируемой методом транслуночной миграции (P < 0,01 vs. NatCM; Рис. 4b).
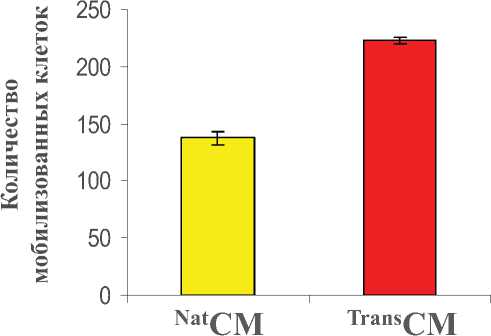
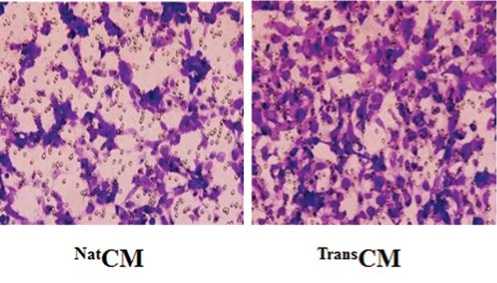
Рис. 4b. Анализ клеточной миграции с применением камер Transwell показал большее количество клеток HUVEC, мигрирующих в ответ на введение TransCM через 6 часов инкубации по сравнению с NatCM (P<0,01 по сравнению с NatCM).
Метод формирования микротрубок in vitro продемонстрировал заметное усиление ангиогенеза клеток HUVEC через 6 часов инкубации с TransCM (P < 0,01 vs. NatCM; Рис. 4c).
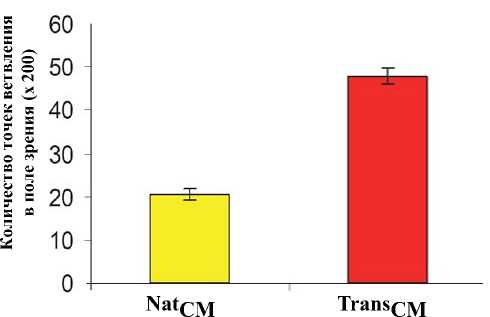
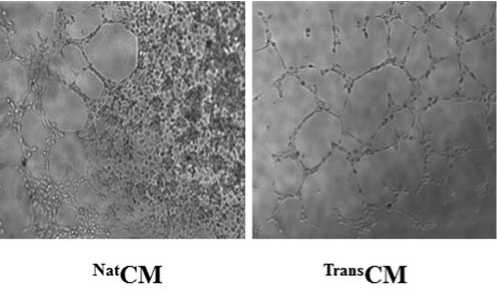
Рис. 4c. Анализ ангиогенеза показал большее количество точек ветвления после 6 часов инкубации клеток HUVEC с TransSM, по сравнению с NatCM (P<0,01).
Образование щелевых контактов in vitro между TransSM и их функциональная оценка
Мы сокультивировали неонатальные кардиомиоциты с NatSM или TransSM. Было продемонстрировано, что количество установленных щелевых контактов между TransSM и кардиомиоцитами (по иммуноокрашиванию на Cx-43, ключевой белок щелевых контактов), было значительно выше, чем между NatSM и кардиомиоцитами (P < 0,05).
Похожий эффект был выявлен и после иммуноокрашивания кардиомиоцитов, сокульти-вированных с NatSM и TransSM на N-cadherin. Мы показали, что количество контактов (позитивных на N-cadherin) между TransSM и кардиомиоцитами было значительно выше, чем количество контактов, установленное между NatSM и кардиомиоцитами (P < 0,01).
Метод переноса красителя был осуществлен с использованием красителя Lucifer Yellow, специфически переносимого функционирующим
Cx-43. Мы наблюдали заметно более широкое распространение и большую протяженность переноса красителя, переносимого от базовой точки, у TransSM по сравнению с NatSM.
Учитывая, что перенос красителя был опосредован через Cx-43, мы пришли к выводу, что экспрессия Cx-43 была выше у TransSM, по сравнению с NatSM. На Рис. 4d показаны микроскопические фотографии транзиентного переноса Ca++, для идентификации электрофизиологического сопряжения между SM (NatSM или TransSM) и неонатальными кардиомиоцитами на 3-й день после их совместного культивирования.
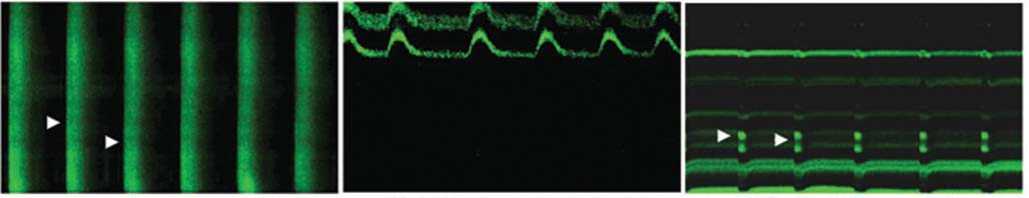
Рис. 4d. Характерные микроскопические изображения кардиомиоцита с кальциевым током, отрицательно перемещающегося NatSM (демонстрирующего двигательную активность благодаря сокращаемости соседних кардиомиоцитов) без кальциевого тока и бьющийся (сокращающийся) TransSM с частичным кальциевым током.
Следует заметить, что только одна треть сокращающихся TransSM имеют кальциевый ток, и кальциевый транзиент не реализовался полностью в целом TransSM, в то же время в NatSM кальциевый транзиент вообще не наблюдался.
Белые стрелки обозначают фазы Ca++ в клетках, которые отсутствовали в несокращающихся NatSM и частично наблюдались в TransSM.
Исследования in vivo
Все крысы (16 в каждой группе) выживали на протяжении всего периода исследования, ни одно животное не умерло вследствие процедуры трансплантации клеток. Каждая из групп крыс состояла из 2 подгрупп, которые анализировались через 7 дней и 8 недель после соответствующего воздействия. Кроме того, дополнительная подгруппа из 3 крыс была использована на 7-й день для молекулярного и гистологического исследования.
Метод ПЦР в реальном времени показал, что пересаженные TransSM продолжали демонстрировать повышенную экспрессию трансгенных факторов роста, включая SDF-1α, HGF, VEGF, и IGF-1, через 7 дней после пересадки. Трансплантированные мужские TransSM показали лучшую выживаемость в сердце, перенесшем инфаркт.
ПЦР-анализ гена sry в сердце самок крыс на 7 день показал обширную выживаемость донорских мужских TransSM в группе 3 (P < 0,05 по сравнению с NatSM в группе-2). В то же время, наличие гена sry не наблюдалось в сердцах самок животных, служивших отрицательным контролем, которым вводилась среда DMEM.
Данные результаты были подтверждены им-муногистологическими исследованиями, в которых визуализировалось выраженное присутствие GFP-положительных TransSM, в месте трансплантации в группе-3 (Рис. 5), по сравнению с GFP-положительными NatSM в группе-2. Кроме того, оказалось, что трансплантирован-
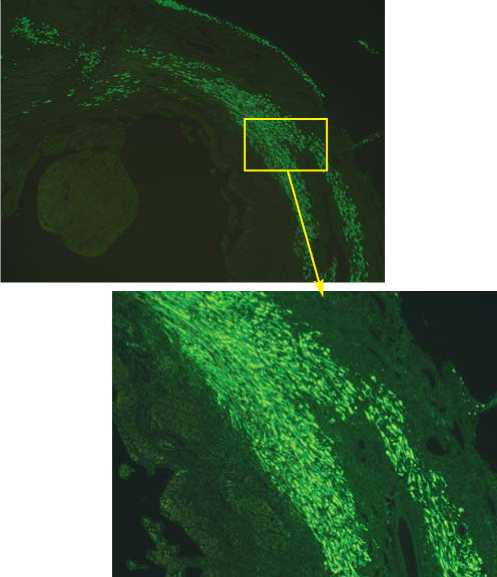
Рис. 5. Флуоресцентное иммуноокрашивание на GFP (зеленый цвет) показало повсеместное присутствие TransSM в группе-3 в инфарктной и периинфарктной зоне через 7 дней после трансплантации, которое было видимо выше, чем присутствие NatSM в группе-2 (х100 , х200).
ные GFP-положительные SM имели продолговатую форму, подобно новым волокнам, и были выстроены параллельно мышечной архитектуре сердца реципиента в обеих группах животных, подвергшихся клеточной трансплантации.
Однако, двойное иммунофлуоресцентное окрашивание на GFP и Cx-43 показало, что только TransSM экспрессировали Cx-43 после трансплантации в инфарктное сердце (красная флуоресценция; рис. 6), в то время как Cx-43 вообще не наблюдался в NatSM после пересадки.
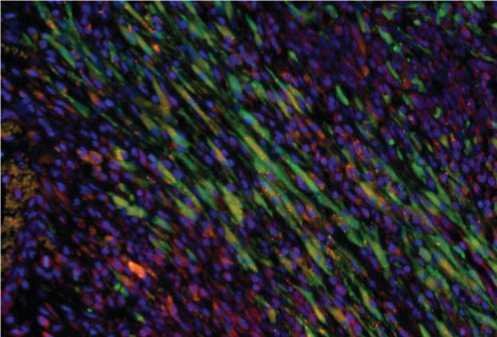
Рис. 6. Двойное флуоресцентное иммуноокрашивание гистологических срезов на GFP (зеленый цвет) и Cx-43 (красный цвет) обнаружило экспрессию Cx-43 клетками TransSM, которые выглядели вытянутыми, как новые волокна, через 7 дней после трансплантации. Для визуализации ядер использовали DAPI (синий цвет).
Эти наблюдения подтверждаются данными, полученными in vitro , о том, что TransSM экспрессируют высокий уровень Cx-43.
Флуоресцентное иммуноокрашивание на Ki67 (белок, экспрессируемый только в ядрах пролиферирующих клеток) показало, что TransSM пролиферируют в инфарктном миокарде в большей степени, чем NatSM (P < 0,01).
Идентификация пролиферирующих SM и их сепарация от делящихся воспалительных клеток в их окружении проводилась путем наложения окрашивания ядер красителями GFP (зеленый цвет), Ki67 (красный цвет) и DAPI (синий цвет).
Хоминг стволовых и прогениторных клеток в миокарде .
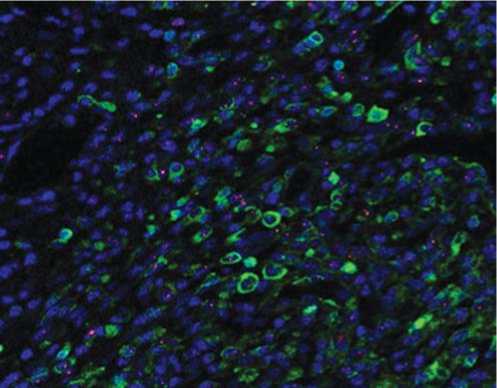
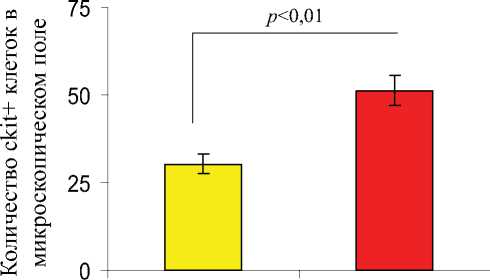
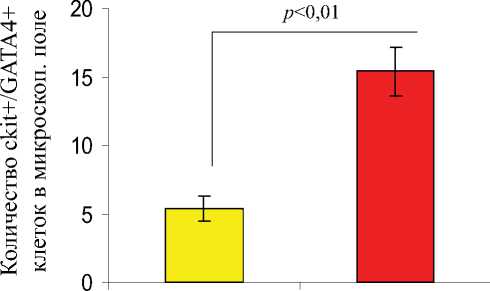
Гистологические срезы, полученные на 7-е сутки из сердечной ткани животных из разных групп лечения были подвергнуты иммуноокрашиванию на присутствие различных линий стволовых и прогениторных клеток, которые были мобилизованы в инфарктное сердце. Число клеток, демонстрирующих присутствие ckit+, ckit+/Gata-4+ (рис. 7.), CD31+, CXCR4+, и CD44+, было значительно выше в группе-3 по сравнению с группой-2. Повышенное количество этих прижившихся клеток наблюдалось в инфарктной и периинфарктной зоне, по сравнению с противоположными областями сердца, как в группе-2, так и в группе-3.
NatSM TranssM
NatSM TranssM
Рис. 7. Флуоресцентное иммуноокрашивание гистологических срезов и определение количества мобилизованных клеток, локализующихся в инфарктном сердце, через 7 дней после соответствующего лечения в группе-2 и группе-3. Число мобилизованных ckit + and ckit +/GATA4+ клеток было значительно выше в группе-3 по сравнению с группой-2.
Помимо мобилизации стволовых/ прогени-торных клеток, метод ПЦР в реальном времени показал более высокую экспрессию CD59 (известного также как пронектин, который широко экспрессируется в гематопоэтических и негематопоэтических клетках и известен своими ингибиторными эффектами на цитолитическую активность комплемента) в сердцах крыс группы-3 через 7 дней после трансплантации (P < 0,01 по сравнению с группой-2).
Размер зоны инфаркта и исследование сердечной функции
Гистологические срезы, полученные на уровне средней области папиллярной мышцы и ок- рашенные трихромом по Массону, показали трансмуральный инфаркт во всех животных через 8 недель после лигирования коронарной артерии (Рис. 8a). Заметное истончение стенки левого желудочка наблюдалось через 8 недель в группе-1, обработанной DMEM, в то же время у животных группы-3 толщина стенки левого желудочка была в значительной степени сохранена. Окрашивание гематоксилином/эозином показало обширную миогенную активность в группе-2 и группе-3 сердец животных, в отличие от группы-1, обработанной DMEM, в которой наблюдали значительно больше рубцовой ткани (Рис. 8b).
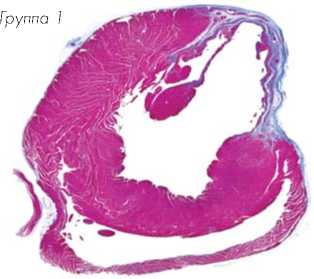
Группа-1 Группа-2 Группа-3
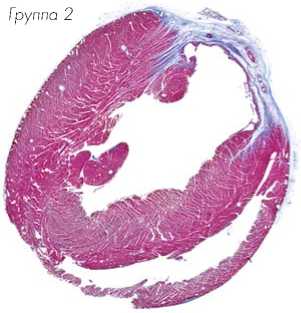
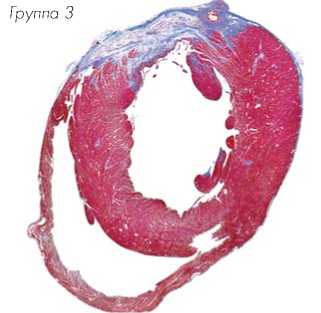
Рис. 8a.
Трансплантация TransSM привела к уменьшению размеров инфаркта и улучшению общей функции сердца.
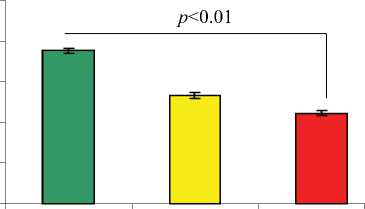
Размер инфаркта, измеренный после окраски трихромом по Массону, был значительно меньшим в группе TransSM (группа-3) по сравнению с группой NatSM (группа-2) и группой DMEM (группа-1) (P < 0,01 по сравнению с обеими группами).
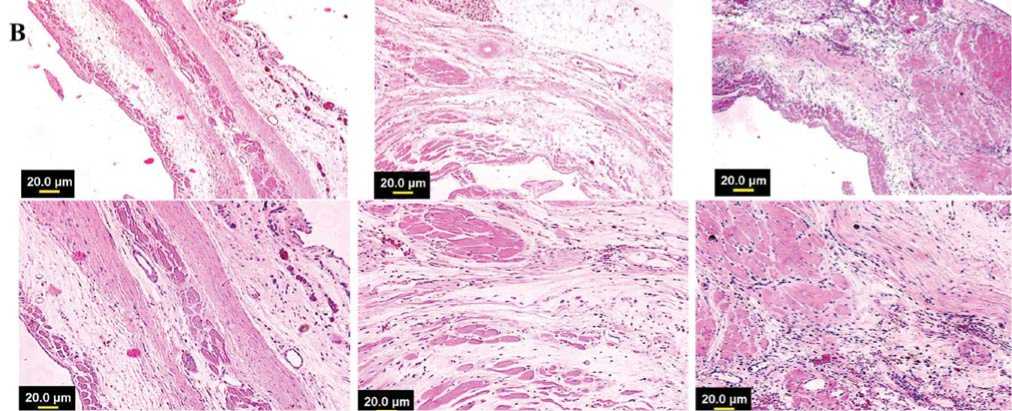
Рис. 8b. Окрашивание гематоксилином/эозином гистологических срезов проводилось для визуализации мышечной архитектоники, которая показала очевидное присутствие островков мышечных волокон в инфарктном миокарде в группе-3.
Расширение размеров инфаркта было значительно выше в группе-1, обработанной DMEM (37,6% ± 0,7%) по сравнению с группой-2 (26,6% ± 0,8%) и группой-3 (22,2% ± 0,6%; P < 0,01 группа-3 по сравнению с группой-2; Рис. 8a). Трансторакальная эхокардиография животных через 8 недель после каждой обработки показала существенные отличия в показателях функции левого желудочка между группами с различным лечением. LVEF (49,0% ± 1,2%) и LVFS (20,7% ± 1,2%) оказались значительно улучшенными в группе-3, обработанной TransSM, по сравнению с контрольной группой-2 (42,9% ± 3,1% и 17,3% ± 0,7% соответственно) и группой-1 (39,6% ± 2,8% и 14,3% ± 2,4% соответственно).
Трансплантация TransSM и неомиокар-диогенез сердца
Двойное флуоресцентное иммуноокрашивание гистологических срезов ткани на экспрессию GFP и десмина обнаружило повышенную миогенную дифференциацию трансплантированных SM в инфарктных и периинфарктных зонах, как в группе-2, так и в группе-3.
Плотность кровеносных сосудов измеряли в центральной и периинфарктной зонах после иммуноокрашивания гистологических срезов с использованием антител на von Willebrand Factor-VIII и гладкомышечный актин (Рис. 9 a, b).
Большое количество зрелых кровеносных сосудов было найдено в областях, окружающих область трансплантации клеток в группе-2 и группе-3.
Число кровеносных сосудов на площадь участка (0,74 мм2) было наибольшим как в пе-риинфарктной (55,2 ± 2,8), так и в инфарктной (31,5 ± 1,9) зоне в группе-3 TransSM-транc-плантации.
Это число было большим (P < 0,01) в пери-инфарктной и инфарктной зонах, чем в группе DMEM и в группе NatSM. Хотя число кровеносных сосудов, наблюдаемое в группе-2, было выше по сравнению с группой-1 (DMEM), мы не наблюдали значительной разницы (P > 0,05) между этими группами как в инфарктной, так и в периинфарктной зонах.
Контроль
TransSM
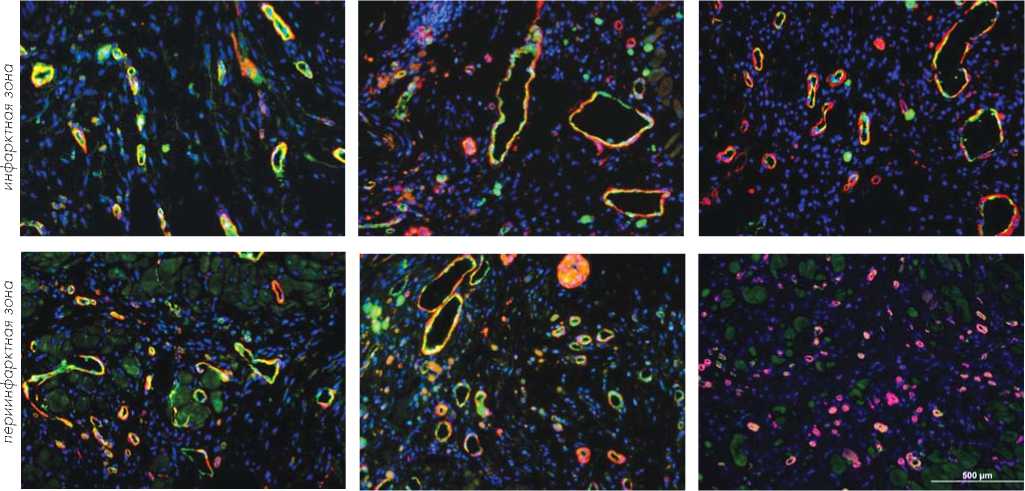
Рис. 9b. TransSM увеличивают плотность кровеносных сосудов. Гистологические срезы окрашивали с использованием с использованием антител на von Willebrand Factor – VIII (зеленый цвет) и гладкомышечный актин (красный цвет) и производили подсчет сосудов.
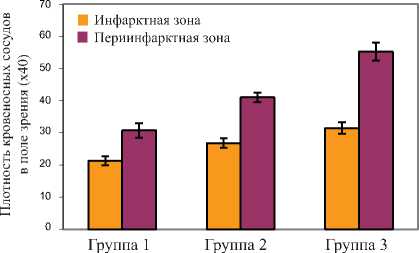
Рис. 9a. TransSM увеличивают плотность кровеносных сосудов. Плотность сосудов была значительно выше в группе-3, как в инфарктной, так и в периинфарктной зонах, по сравнению с группой-1 и группой-2 (P < 0,05).
Обсуждение результатов
Сложность процессов, происходящих в сердце, перенесшем инфаркт после ишемического повреждения, требует терапевтической стратегии, которая могла бы охватить различные аспекты заболевания и минимизировать тяжесть ишемического повреждения. Несмотря на обширные исследования в области терапии стволовыми клетками для восстановления миокарда, поиск подходящих донорских клеток с оптимальными характеристиками все еще не окончен. Полагают, что эффективной стратегией является перепрограммирование стволовых клеток для адаптации в жестких условиях микроокружения для лучшей выживаемости и миогенной дифференциации, а также для улучшения местного кровотока в ишемизированном миокарде [18-20]. Наша многоцелевая стратегия использует потенциал стволовых/ прогениторных клеток в мобилизации, выживаемости и ангиомиогенной дифференциации в ответ на градиент концентрации определенной группы цитокинов со специфическими функциями. Помимо одновременного рекрутинга внутренне доступных стволовых/проге-ниторных клеток в данном исследовании, высвобождение ростовых факторов из генетически перепрограммированных SM изменяло микроокружение ишемизированного сердца посредством паракринной активности. Данные молекулярные события в ответ на вызванную SM сверхпродукцию множественных ростовых факторов привели к дополнительным терапевтическим положительным эффектам, выявленным в изменении размера инфаркта и улучшении общей сердечной функции, в большей степени, чем трансплантация NatSM. Учитывая низкую эффективность невирусного метода трансфекции в целом и множественной трансфекции в частности, мы выбрали трансфекцию SM с использованием индивидуальных трансгенов. За этим следовало создание пула трансфицированных клеток, c выделением паракринных факторов перед трансплантацией. Наш мультимодальный подход с применением невирусной генной терапии осуществлял активацию и участие внутренних систем ли-ганд/рецептор – VEGF/VEGFR, HGF/cMet, IGF-1/IGF-1R, и SDF-1α/CXCR4 – в процессах регенерации миокарда путем создания градиента соотвествующих лигандов (ростовых факторов) в инфарктном сердце, вызванного трансплантацией SM, обеспечивающих сверx- продукцию этих ростовых факторов. Ранее мы показали эффективность каждого из выбранных ростовых факторов в лечении экспериментального инфаркта миокарда [14, 15, 17]. Хотя сравнение результатов этих исследований с результатами данного исследования затруднительно из-за различий в организации эксперимента, равно как и различий в стратегии доставки ростовых факторов и типе клеток, используемых для трансгенной доставки, эти данные послужили основой для нашей многоцелевой стратегии. Трансгенная доставка индивидуальных ростовых факторов продемонстрировала эффекты каждого отдельного ростового фактора, и в ней отсутствовали разнообразные сигнальные механизмы, достигнутые в данном исследовании за счет одновременной доставки нескольких ростовых факторов. Таким образом, активация нескольких сигнальных механизмов привела к уменьшению клеточного апоптоза, увеличению клеточной пролиферации, массивной мобилизации стволо-вых/прогениторных клеток и хоумингу в месте ишемического повреждения для участия в процессе восстановления путем ангиомиогенеза. Логическим обоснованием выбора различных ростовых факторов было наличие их определенной функциональной роли в процессе восстановления миокарда.
Выбор основывался на следующих критериях:
-
1. IGF-1 является плейотропным фактором, который взаимодействует с широко распространенными рецепторами IGF-1R, приводя к ци-топротекторному эффекту, а также эффектам улучшения пролиферации и дифференцировки клеток.
-
2. Взаимодействие лиганда VEGF с рецепторами тирозинкиназы, VEGFR1 и VEGFR2, является проангиогенным и эффективно про-мотирует выживание эндотелиальных клеток, пролиферацию и рост с активацией Akt-сиг-
- нального пути [23, 24]. Хотя в работе Lee RJ [25] было отмечено образование гемангиомы после доставки гена VEGF в сердце, данное исследование использовало неконтролируемую ретровирусную непрерывную экспрессию VEGF, в отличие от невирусной кратковременной экспрессии VEGF в нашем исследовании.
-
3. HGF известен как хемоаттрактант, который индуцирует миграцию стволовых клеток в ответ на концентрационный градиент, развивающийся при инфаркте. HGF действует путем формирования сигнала для фактора мезенхи-мального-эпителиального (Met) порождающего популяцию сердечных и прогениторных клеток [26]. Взаимодействие лиганда/рецепто-ра HGF/Met регулирует клеточный рост, подвижность и морфогенез путем активирования сигнального каскада тирозинкиназы [27]. Введение HGF улучшало эффективность клеточной трансплантации [28] и промотировало пролиферацию кардиомиоцитов в экспериментальных моделях на животных [29]. Более того, трансплантация внутрь миокарда SM, невирус-но модифицированных с целью экспрессии HGF, дает возможность лимитировать расширение размеров инфаркта и нарушение сердечной функции, и эти эффекты были еще более выражены в доставке этих клеток при помощи клеточных каркасов [30].
-
4. SDF-1α известен как лиганд для рецепторов CXCR4, экспрессированных на BMSC. Временная повышающая регуляция SDF-1α в инфарктном сердце в острой фазе после инфаркта создает сигнал для мобилизации и хоуминга BMSC в инфарктном сердце [31], помимо действия в качестве удерживающего фактора для обеспечения их участия в процессе восстановления [32]. Принимая во внимание их определенную функциональную роль, мы предположили, что совместный эффект 4-х ростовых факторов состоит в рекрутинге и вовлечении в процессы репарации собственных стволовых/ прогениторных клеток.
Недавно было показано, что IGF-1 также промотирует индукцию Cx-43 и его перемещение в митохондрии для исполнения его двоякой роли интеграции стволовых клеток и посттрансплантационной защиты [18]. Стимуляция IGF-1 также приводит к индукции HIF-1α через ERK-путь, в то время как гипоксия, вызванная экспрессией HIF-1α, не зависит от ERK [21]. Однако, высокие уровни экспрессии белка HIF-1α, как известно, возникают при стимуляции стволовыми клетками в условиях нор-моксии по Akt- и ERK-зависимым путям [22].
Способность SM к доставке терапевтических трансгенов в сердце интенсивно изучалась ранее [33-35]. Главным образом, эти исследования включали использование общепринятой стратегии генетической модификации SM одним или двумя трансгенами, обеспечивающими проангиогенный и/или способствующий выживанию эффект и демонстрирующие улучшение ангиогенеза в инфарктном сердце и уменьшение размеров зоны инфаркта.
Наша стратегия доставки трансгенов множественных ростовых факторов была направлена на то, чтобы использовать совокупное благотворное воздействие многих факторов при лечении инфаркта.
В отличие от предыдущих работ [33-35], мы демонстрируем четкое доказательство, что выбранная четверка ростовых факторов создает градиент ростового фактора для мобилизации собственного пула BMSC, EPC и резидентных сердечных стволовых клеток реципиента для участия в процессе репарации. Помимо градиента ростовых факторов, мобилизации и рекрутингу стволовых/прогениторных клеток также способствует паракринное высвобождение MMP из TransSM [36]. MMP3 и MMP9 задействованы в биогенезе внеклеточного матрикса и ремоделировании ткани в различных физиологических и патологических процессах [37]. VEGF и SDF-1α повышают экспрессию MMP-9, а также вызывают выброс KitL и рекрутинг c-Kit+ BMSC [36]. Высвобождение sKitL, осуществляемое MMP-9, позволяет вновь интегрирующим клеткам костного мозга перемещаться в сосудистую нишу, способствующую дифференцировке и восстановлению пула стволовых/прогениторных клеток.
Мы также наблюдали значительно большую экспрессию семейства Sfrp – Sfrp1, Sfrp2, Sfrp4, and Sfrp5. Sfrps принадлежит к семейству разнообразных гликопротеинов, которые структурно близки к поверхностным рецепторам и участвуют в активации сигнального Wnt, клеточной дифференцировки и клеточной гибели [38]. Повышенное образование белка из семейства Wnt (такого как Wnt3a) наблюдается при гипоксии и вызывает апоптоз кардиомиоцитов путем активации его рецептора [39].
В настоящее время полагают, что белки группы Sfrp, такие как Sfrp2, конкурируют с рецептором Wnt лигандов из-за их структурного сходства и таким образом противодействуют активации сигнального пути Wnt и, следовательно, приводят в действие антиапоптотичес-кие эффекты в кардиомиоцитах и уменьшают фиброз в тканях сердца [40, 41]. Как было показано недавно Laeremans и др. [42], пептид UM206 блокирует рецепторы, приводя к уменьшению размеров инфаркта и улучшению функционального восстановления сердца. Эти результаты доказывают, что UM206 обладает эффектом, близким к эффекту Sfrps в показате- лях восстановления сердца, что далее подтверждает логическое обоснование того, что блокирование рецептора (как показано в нашем исследовании, повышением белков Sfrp) является эффективным для Wnt3a и его нежелательного влияния на миокард.
Еще более важно, что мы наблюдали более высокое содержание Cx-43 в TransSM, что помогало клеткам приживаться после трансплантации. Исследования in vitro выявили, что NatSM, культивированные совместно с неонатальными кардимиоцитами, выделяли N-cad-herin и Cx-43 в межклеточных стыках [43]. Однако, большинство исследований показали недостаток экспрессии N-cadherin и Cx-43 клетками NatSM после трансплантации и невозможность их электромеханической синхронизации с кардиомиоцитами хозяина, несмотря на более высокую экспрессию обоих белков недифференцированными [44]. По нашим наблюдениям, TransSM экспрессировали значительные количества Cx-43 до 7-х суток после трансплантации. Однако, мы не наблюдали экспрессию Cx-43 в 8-недельных гистологических срезах. Несмотря на многообещающие результаты, наше исследование имело определенные ограничения. Хотя мы не наблюдали существенного увеличения плотности крове- носных сосудов, роль и судьба мобилизованных клеток в ангиомиогенезе в инфарктном сердце во временной точке 8 недель не была определена из-за отсутствия специфического маркера на клетках. Проблема еще сильнее подчеркивалась сложностью идентификации метки GFP на трансплантированных SM через 8 недель, затрудняя отделение новых волокон, происходящих из трансплантированных SM, от тех, которые были образованы мобилизованными клетками. Неспособность SM подвергаться кардиомиогенной дифференцировке и электромеханической синхронизации с миоцитами хозяина может послужить основанием того, что они в будущем будут заменены на другие клетки, имеющие потенциал кардиомио-генной дифференцировки.
Более того, будут проводиться исследования in vivo , с целью оценки эффективности кардиомиоцитов, полученных из SM. В заключение заметим, что использование цитокинов совместно со SM для ускоренной мобилизации стволовых/прогениторных клеток из системы периферической кровеносной системы, костного мозга и сердца, так же, как и улучшение выживаемости клеток за счет выделения паракринных факторов, является новым подходом к регенерации инфарктного миокарда.
Список литературы Трансплантация генетически модифицированных стволовых клеток, повышенно экспрессирующих терапевтические ростовые факторы, при лечении экспериментального инфаркта миокарда крыс
- Singla DK. Stem cells in the infarcted heart. J Cardiovasc Transl Res 2010; 3:73-78.
- P Menasche, A A Hagege, M Scorsin, B Pouzet, M Desnos, et al. Myoblast transplantation for heart failure. Lancet 2001;357: 279-280.
- P Menasche, O Alfieri, S Janssens, W McKenna, H Reichenspurner, L Trinquart, et al. The Myoblast Autologous Grafting in Ischemic Cardiomyopathy (MAGIC) trial: first randomized placebo-controlled study of myoblast transplantation. Circulation 2008; 117:1189-1200.
- KC Wollert, GP Meyer, J Lotz, S Ringes-Lichtenberg, P Lippolt, C Breidenbach, S Fichtner, et al. Intracoronary autologous bone-marrow cell transfer after myocardial infarction: the BOOST randomised controlled clinical trial. Lancet 2004;364:141-148.
- BZ Atkins, MT Hueman, J Meuchel, KA Hutcheson, et al. Cellular cardiomyoplasty improves diastolic properties of injured heart. J Surg Res 1999; 85:234-242.
- DA Taylor, BZ Atkins, P Hungspreugs, TR Jones, MC Reedy, KA Hutcheson, et al. Regenerating functional myocardium: improved performance after skeletal myoblast transplantation. Nat Med 1998; 4: 929-933.
- M Perez-Ilzarbe, O Agbulut, B Pelacho, C Ciorba, E San JoseEneriz, M Desnos, AA Hagege, P Aranda, EJ Andreu, P Menasche and F Prosper. Characterization of the paracrine effects of human skeletal myoblasts transplanted in infarcted myocardium. Eur J Heart Fail 2008;10:1065-1072.
- J Kajstura, M Rota, B Whang, S Cascapera, T Hosoda, C Bearzi, D Nurzynska, H Kasahara, E Zias, et al. Bone marrow cells differentiate in cardiac cell lineages after infarction independently of cell fusion. Circ Res 2005;96:127-137.
- R Uemura, M Xu, N Ahmad and M Ashraf. Bone marrow stem cells prevent left ventricular remodeling of ischemic heart through paracrine signaling. Circ Res 2006;98: 1414-1421.
- K Jujo, M Ii and DW Losordo. EPC in neovascularization of infarcted myocardium. J Mol Cell Cardiol 2008;45: 530-544.
- B Dawn, AB Stein, K Urbanek, M Rota, B Whang, R Rastaldo, D Torella, XL Tang, A Rezazadeh, et al. Cardiac stem cells delivered intravascularly traverse the vessel barrier, regenerate infarcted myocardium, and improve cardiac function. Proc Natl Acad Sci USA 2005;102:3766-3771.
- M Rota, ME Padin-Iruegas, Y Misao, A De Angelis, S Maestroni, J Ferreira-Martins, E Fiumana, R Rastaldo, ML Arcarese, et al. Local activation or implantation of cardiac progenitor cells rescues scarred infarcted myocardium improving cardiac function. Circ Res 2008;103:107-116.
- A Banfi, ML Springer,HM Blau. Myoblast-mediated gene transfer for therapeutic angiogenesis. Methods Enzymol 2002;346:145-157.
- MI Niagara, H Haider, S Jiang, M Ashraf. Pharmacologically preconditioned skeletal myoblasts are resistant to oxidative stress and promote angiomyogenesis via release of paracrine factors in the infarcted heart. Circ Res 2007;100:545-555.
- M Konoplyannikov, Haider KH, Lai VK, Ahmed RP, Jiang S, Ashraf M. Activation of diverse signaling pathways by ex-vivo delivery of multiple cytokines for myocardial repair. Stem Cells Dev 2013. Jan 15; 22(2): 204-15.
- MA Stagg, SR Coppen, K Suzuki, A Varela-Carver, J Lee, NJ Brand, S Fukushima, MH Yacoub, CM Terracciano. Evaluation of frequency, type, and function of gap junctions between skeletal myoblasts overexpressing connexin43 and cardiomyocytes: relevance to cell transplantation. Faseb J 2006; 20:744-746.
- Jiang S, H Haider, NM Idris, A Salim, M Ashraf. Supportive interaction between cell survival signaling and angiocompetent factors enhances donor cell survival and promotes angiomyogenesis for cardiac repair. Circ Res 2006; 99:776-784.
- G Lu, HK Haider, S Jiang and M Ashraf. Sca-1+ stem cell survival and engraftment in the infarcted heart: dual role for preconditioning-induced connexin. Circulation 2009;119: 2587-96.
- HW Kim, HK Haider, S Jiang and M Ashraf. Ischemic preconditioning augments survival of stem cells via miR-210 expression by targeting caspase-8-associated protein 2. J Biol Chem 2009; 284: 33161-168.
- JY Hahn, HJ Cho, HJ Kang, TS Kim, MH Kim, JH Chung, JW Bae, BH Oh, YB Park and HS Kim. Pre-treatment of mesenchymal stem cells with a combination of growth factors enhances gap-junction formation, cytoprotective effect on cardiomyocytes, and therapeutic efficacy for myocardial infarction. J Am Coll Cardiol 2008; 51: 933-43.
- KM Sutton, S Hayat, NM Chau, S Cook, J Pouyssegur, A Ahmed, N Perusinghe, et al. Selective inhibition of MEK1/2 reveals a differential requirement for ERK1/2 signalling in the regulation of HIF-1 in response to hypoxia and IGF-1. Oncogene 2007; 26: 3920-29.
- M Pedersen, T Lofstedt, J Sun, L Holmquist-Mengelbier, S Pahlman, L Ronnstrand. Stem cell factor induces HIF-1α at normoxia in hematopoietic cells. Biochem Biophys Res Commun 2008; 377: 98-103.
- N Ferrara, HP Gerber, J LeCouter. The biology of VEGF and its receptors. Nat Med 2003; 9: 669-76.
- HP Gerber, AK Malik, GP Solar, D Sherman, XH Xiang, G Meng, K Hong, JC Marsters, N Ferrara. VEGF regulates haematopoietic stem cell survival by an internal autocrine loop mechanism. Nature 2002; 417: 954-58.
- RJ Lee, ML Springer, WE Blanco-Bose, R Shaw, PC Ursell, HM Blau. VEGF gene delivery to myocardium: deleterious effects of unregulated expression. Circulation 2000;102: 898-901.
- A Linke, P Muller, D Nurzynska, C Casarsa, D Torella, A Nascimbene, et al. Stem cells in the dog heart are self-renewing, clonogenic, and multipotent and regenerate infarcted myocardium, improving cardiac function. Proc Natl Acad Sci USA 2005; 102: 8966-71.
- R Madonna, G Rokosh, R De Caterina, R Bolli. Hepatocyte growth factor/Met gene transfer in cardiac stem cells-potential for cardiac repair. Basic Res Cardiol 2010; 105: 443-52.
- K Tambara, GU Premaratne, G Sakaguchi, N Kanemitsu, X Lin, H Nakajima, et al. Administration of control-released hepatocyte growth factor enhances the efficacy of skeletal myoblast transplantation in rat infarcted hearts by greatly increasing both quantity and quality of the graft. Circulation 2005;112: I129-34.
- M Iwasaki, Y Adachi, T Nishiue, K Minamino, Y Suzuki, Y Zhang, et al. Hepatocyte growth factor delivered by ultrasound-mediated destruction of microbubbles induces proliferation of cardiomyocytes and amelioration of left ventricular contractile function in Doxorubicin-induced cardiomyopathy. Stem Cells 2005; 23: 1589-97.
- A Poppe, P Golsong, B Blumenthal, R von Wattenwyl, P Blanke, F Beyersdorf, C Schlensak, M Siepe. Hepatocyte growth factor-transfected skeletal myoblasts to limit the development of post-infarction heart failure. Artif Organs 2011; 36: 238-46.
- AT Askari, S Unzek, ZB Popovic, CK Goldman, F Forudi, M Kiedrowski, A Rovner, SG Ellis, et al. Effect of stromal-cell-derived factor 1 on stem-cell homing and tissue regeneration in ischaemic cardiomyopathy. Lancet 2003; 362: 697-703.
- M Grunewald, I Avraham, Y Dor, E Bachar-Lustig, A Itin, S Jung, S Chimenti, et al. VEGF-induced adult neovascularization: recruitment, retention, and role of accessory Cells. Cell 2006; 124:175-189.
- S Aharinejad, D Abraham, P Paulus, K Zins, M Hofmann, W Michlits, et al. Colony-stimulating factor-1 transfection of myoblasts improves the repair of failing myocardium following autologous myoblast transplantation. Cardiovasc Res 2008; 79: 395-404.
- M Bialas, Krupka M,Janeczek A, Rozwadowska N, et al. Transient and stable transfections of mouse myoblasts with genes coding for pro-angiogenic factors. J Physiol Pharmacol 2011; 6: 219-28.
- B Blumenthal, P Golsong, A Poppe, C Heilmann, C Schlensak, F Beyersdorf, M Siepe. Polyurethane scaf-folds seeded with genetically engineered skeletal myoblasts: a promising tool to regenerate myocardial function. Artif Organs 2011; 34: E46-E54.
- B Heissig, K Hattori, S Dias, M Friedrich, B Ferris, NR Hackett, RG Crystal, et al. Recruitment of stem and progenitor Cells from the bone marrow niche requires MMP-9 mediated release of kit-ligand. Cell 2002; 109: 625-37.
- Sang QX. Complex role of matrix metalloproteinases in angiogenesis. Cell Res 1998; 8: 171-77.
- Steve E, Jones CJ. Secreted frizzled-related proteins: searching for relationships and patterns. Bio Essays 2002; 24: 811-20.
- M Mirotsou, Z Zhang, A Deb, L Zhang, M Gnecchi, N Noiseux, H Mu, A Pachori, V Dzau. Secreted frizzled related protein 2 (Sfrp2) is the key Akt-mesenchymal stem cell-released paracrine factor mediating myocardial survival and repair. Proc Natl Acad Sci USA 2007; 104: 1643-48.
- Z Zhang, A Deb, A Pachori, W He, J Guo, et al. Secreted frizzled related protein 2 protects cells from apoptosis by blocking the effect of canonical Wnt3a. J Mol Cell Cardiol 2009; 46: 370-77.
- W He, L Zhang, A Ni, Z Zhang, M Mirotsou, et al. Exogenously administered secreted frizzled related protein 2 (Sfrp2) reduces fibrosis and improves cardiac function in a rat model of myocardial infarction. Proc Natl Acad Sci USA 2010; 107: 21110-15.
- H Laeremans, TM Hackeng, MA van Zandvoort, VL Thijssen, BJ Janssen, HC Ottenheijm, et al. Blocking of frizzled signaling with a homologous peptide fragment of Wnt3a/Wnt5a reduces infarct expansion and prevents the development of heart failure after myocardial infarction. Circulation 2011;124:1626-35.
- L Formigli, F Francini, A Tani, R Squecco, D Nosi, L Polidori, S Nistri, L Chiappini, V Cesati, et al. Morphofunctional integration between skeletal myoblasts and adult cardiomyocytes in coculture is favored by direct cell-cell contacts and relaxin treatment. Am J Physiol Cell Physiol 2005; 288: C795-C804.
- H Reinecke, E Minami, V Poppa and CE Murry. Evidence for fusion between cardiac and skeletal muscle cells. Circ Res 2004; 94: e56-e60.


