Современные представления о CAR T-клеточной терапии
Автор: Кувшинов А. Ю., Волошин С. В., Кузяева А. А., Шуваев В. А., Михалева М. А., Мартынкевич И. С., Чечеткин А. В., Бессмельцев С. С.
Журнал: Вестник гематологии @bulletin-of-hematology
Рубрика: Обзор литературы
Статья в выпуске: 2 т.15, 2019 года.
Бесплатный доступ
Адоптивная иммунотерапия является одним из признанных современныхметодов лечения онкологических заболеваний,основанном на достижениях генетики, молекулярной биологии и иммунологии, цельюкоторого является восстановление нормального функционирования противоопухолевогоответа. В основу данного вида терапии положено создание опухоль-специфичных цитотоксических лимфоцитов, экспрессирующиххимерный антигенный рецептор (chimericantigen receptor, CAR). Из экспериментальнойтехнологии лечение онкологических заболеваний CAR T-клетками превращается в быстроразвивающийся подход к терапии с более чем500 зарегистрированными клиническими исследованиями при гематологических новообразованиях и солидных опухолях. В статьепредставлено описание биотехнологии, результаты клинических исследований, обсуждениепобочных эффектов и проблем появления резистентности опухолей, а также рассмотреныперспективы дальнейшего развития и внедрения в рутинную клиническую практику CART-клеточной терапии.
Адоптивная иммунотерапия, химерный антигенный рецептор, cart-клетка, онкогематология
Короткий адрес: https://sciup.org/170172533
IDR: 170172533
Modern representation of CAR T-cell therapy
Adoptive immunotherapy is oneof the accepted modern methods of cancertreatment, based on achievements of genetics,molecular biology and immunology and its goalis to repair the normal functioning of antitumorresponse. The basis of this type of therapy is thecreation of tumor-specific cytotoxic lymphocytesexpressing the chimeric antigen receptor (CAR).The treatment of cancer with CAR T-cells istransformed from experimental technology into arapidly developing therapeutic approach with morethan 500 registered clinical trials in hematologicmalignancies and solid tumors. The article presentsa description of biotechnology, the results of clinicaltrials, a discussion of side effects and problems ofthe emergence of tumor resistance, as well as theprospects for further development and introductionof CAR T-cell therapy into routine clinical practice.
Текст обзорной статьи Современные представления о CAR T-клеточной терапии
Иммунная система. Иммунный ответ осуществляется посредством двух форм иммунитета: врожденного и приобретенного (адаптивного). Врожденный иммунитет распознает чужеродные антигены, не нанося вреда клеткам и тканям организма. Распознавание чужеродных антигенов вызывает ответную реакцию воспаления и фагоцитоза с участием врожденных иммунных клеток (нейтрофилы и макрофаги). Такой ответ происходит быстро. Адаптивный иммунитет требует больше времени для формирования ответа. В тимусе в течение первого года жизни человека развиваются Т-клетки, которые отражают ненаследуемые материнские аллоантигены, поскольку они вероятно, не подвергались иммуностимуляции патогеном [1]. Воздействие различных антигенов улучшает процесс адаптивного иммунитета. Т-лимфоциты, являясь ключевым звеном адаптивного иммунного ответа, обладают следующими эффекторными функциями: 1) уничтожение инфицированных или опухолевых клеток (цитотоксические Т-лимфоциты); 2) активация (Т-хелперы) макрофагов, В-клеток, секреция цитокинов; ослабление иммунных реакций.
Иммунная система и онкология. Критическим компонентом адаптивного иммунитета является способность Т-клеток распознавать патоген-ассоциированные иммуностимуляторы по топографии эпитопа чужеродного антигена. Процесс развития опухоли и изменение ее свойств при взаимодействии с иммунной системой, в том числе с участием Т-клеток, сформировался как концепция «иммуноредактирования рака». Основные фазы взаимодействия опухоли и иммунной системы: 1) элиминация; 2)равновесие и 3)ускользание [2]. Переориентирование иммунной системы опухолью возможно вследствие ее пластичности.
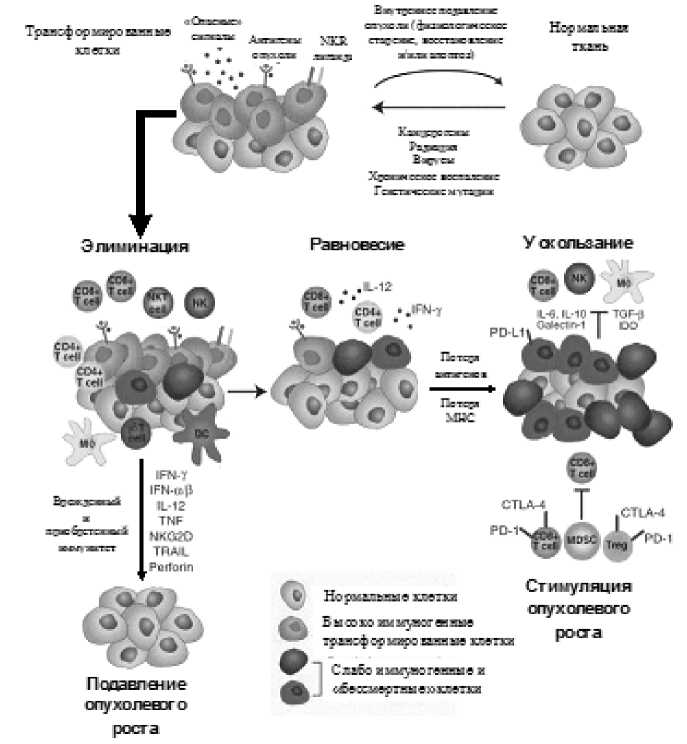
Рис. 1. Процесс взаимодействия иммунной системы и опухоли [2]
Во время фазы «элиминации» чужеродные эпитопы, продуцируемые раковыми клетками устраняются Т-клетками, что согласуется с теорией иммунного надзора, когда иммунная система активно ищет раковые клетки и устраняет их [3]. По мере роста опухоли включаются механизмы,связанные с ангиогенезом и локальным воспалением,что ведет к сильному иммунному ответу с вовлечением NKT, NK, γδ T-клеток, макрофагов и дендритных клеток продуцирующих IFN-γ [1, 3]. IFN-γ создает прямые и непрямые механизмы клеточной гибели и, как следствие, ведет к образованию клеточного детрита,который попадает в лимфатические узлы, где дендритные клетки косвенно активируют CD4 и CD8 Т-клетки. В конечном итоге, CD4 и CD8 Т-клетки мигрируют к опухолевому очагу, чтобы «убить» опухолевые клетки [4].
Фаза «равновесия» наступает тогда, когда раковые клетки погибают с той же скоростью с какой происходит их рост. Если оставшиеся после фазы «элиминации» опухолевые клетки мутируют и размножаются таким образом, что динамическое равновесие сдвигается в сторону роста опухоли, то, согласно Дарвиновской эволюции, те клетки, которые приспособились к иммунному ответу, могут размножаться бесконтрольно [3].
Таким образом, фаза «ускользания» из-под иммунного надзора происходит, когда раковые клетки становятся неузнаваемыми для иммунной системы,посредством мутаций в генах или под влиянием эпигенетических факторов. Процесс дальнейшего подавления иммунного контроля продолжается путем формирования микроокружения опухоли [3].
Мутации на клеточной мембране,которые не распознаются иммунной системой,пред-ставляют особый интерес для иммунотерапии. Необходимо понимать, что процесс распознавания лимфоцитами целого спектра антигенов которые могут быть различны по своей иммуногенности, не представляется возможным рассматривать в простой бинарной системе.
История. Концепция создания химерного антигенного рецептора принадлежит G. Gross и его коллегам, которые в 1989 г. обнаружили возможность перенаправлять Т-клеточный ответ с помощью сконструированного искусственного рецептора. Это исследование заложило основу для разработки CAR первого поколения клеток [5]. Годы последовательных и масштабных испытаний ознаменовались первым успешным клиническим применением CAR T-клеток 1-го поколения (α-GD2 CAR) о котором в 2008 г. сообщили M. Brenner et al.: у трех из 11 пациентов с нейробластомой была достигнута полная ремиссия заболевания [6 7]. С этого момента началось бурное развитие данной биотехнологии.
Химерный антигенный рецептор . Химерный антигенный рецептор состоит из внеклеточного домена — одноцепочечного вариабельного фрагмента антитела (single-chain variable fragment, scFv) и внутриклеточных сигнальных доменов,состоящих из ζ-цепи CD3 и дополнительных ко-стимулирующих молекул. Вне- и внутриклеточный домены связаны с помощью внеклеточного шарнира (spacer)и трансмембранного домена. Активация Т-клеток происходит в процессе передачи сигнала от внеклеточного домена,который распознает ассоциированный с опухолью антиген (tumor associated antigen, TAA), через трансмембранный домен на внутриклеточный [7]. Такая структура CAR заставляет Т-лимфоциты специфически реагировать на опухолевые клетки с избранными антигенами.
В настоящее время существует 4 поколения CAR, отличающихся между собой по строению внутриклеточного домена. Внутриклеточный домен химерных рецепторов 1-го поколения состоит только из ζ-цепи CD3, в то время как у химерных рецепторов 2-го и 3-го поколений он состоит из одной или двух костимулирующих молекул, таких как CD27, CD28, CD134 (OX-40) CD137 (4–1BB) [7]. Костимулирующие молекулы усиливают литическую и пролиферативную активность CAR T-клеток. Но, наличие второго костимулирующего домена в химерных рецепторах 3-го поколения увеличивает активность CAR T-клеток, что приводит к усилению выброса цитокинов [1]. Это ограничило применение рецепторов 3-го поколения,в то время как химерные рецепторы 2-го поколения находятся на различных этапах клинических испытаний. Последнее, 4-е поколение — Т-клетки перенаправленные для универсального цитокин-опос-редованного уничтожения (T cells redirected for universal cytokine-mediated killing, TRUCKs) которые активируют врожденную иммунную систему посредством депонирования интерлейкина-12 (ИЛ-12) в опухолевом очаге [8].
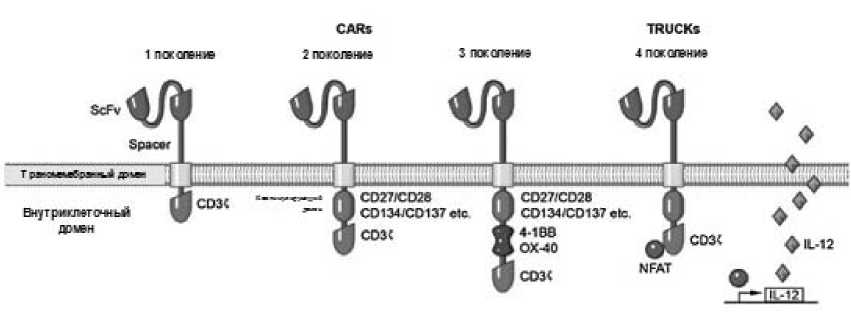
Рис. 2. Поколения химерных антигенных [1]
Приоритетом разработки внеклеточного домена является поиск подходящих мишеней которыми обычно являются поверхностные антигены, эпитоп которых уникален для раковых клеток.
Таблица 1
Мишени в CAR T-клеточной терапии (адаптировано из [1, 7])
|
Опухоль |
Антигены, на которые нацелены CAR |
|
Онкогематологические заболевания |
CD19 |
|
CD20 |
|
|
CD22 |
|
|
CD30 |
|
|
CD33 |
|
|
CD38 |
|
|
CD138 |
|
|
ROR1 |
|
|
Lewis(Le)-T (Ley) |
|
|
α-цепь рецептора интерлейкина-3 (ИЛ-3) |
|
|
Igκ легкие цепи |
|
|
Антиген созревания В-клеток (B cell matura^on an^gen, BCMA) [9, 10]. |
|
|
Лиганд NKG2D [11]. |
|
|
Немелкоклеточный рак легких |
Рецептор эпидермального фактора роста (Epidermal growth factor receptor, EGFR) |
|
Меланома |
HLA-A1/MAGE-A1 |
|
Ганглиозид GD2 |
|
|
Нейробластома |
L1-клеточная молекула адгезии (CD171) |
|
Глиома |
EGFRvIII |
|
Рак предстательной железы |
Человеческий простат-специфический мембранный антиген (Human prostate-specific membrane antigen, hPSMA) |
|
Мезотелиома |
Мезотелин |
|
Белок активации фибробластов (Fibroblast ac^va^on protein, FAP) |
|
|
Остеосаркома |
Человеческий рецептор эпидермального фактора роста (Human epidermal growth factor receptor, HER2) |
|
α-цепь рецептора интерлейкина-11 (ИЛ-11) |
|
|
Рак шейки матки |
CD44v7 и CD44v8 |
|
Желудочно-кишечные стромальные опухоли |
KIT |
|
Рак яичников |
Фолатный рецептор-α (Folate receptor-α, FRα) |
|
Лиганд NKG2D |
|
|
Опухоли брюшины |
MUC16 |
Продолжение таблицы
|
Опухоль |
Антигены, на которые нацелены CAR |
|
Глиобластома |
α2-цепь рецептора интерлейкина-13 (ИЛ-13) |
|
HER2 и α2-цепь рецептора ИЛ-13 |
|
|
Метастатический почечно-клеточный рак |
Карбокси-ангидраза-IX (carboxy-anhydrase-IX, CAIX) |
|
Аденокарцинома поджелудочной железы |
Her2/neu и CD24 |
|
Рак молочной железы |
ErbB2 и MUC1 |
|
Метастазирование в печень |
Раковый-эмбриональный антиген (Carcinoembryonic antigen, CEA) |
|
Универсальное воздействие |
Биотин |
|
Изотиоцианат флюоресцин (fluorescein isothiocyanate, FITC) |
|
|
Воздействие на опухолевую сосудистую сеть |
VEGFR-2 |
Ведутся разработки тандемных CAR, внеклеточный домен которых состоит из двух разных scFv. Например, биспецифический тандем CAR — HER2 и α2-цепь рецептора ИЛ-13 демонстрирует дополнительную активацию Т-клеток при встрече с обоими антигенами [12]. A. Bister et al. сообщают о потенциально эффективной комбинации CD5 и CD19 тандемного CAR у пациентов с мантийноклеточной лимфомой. Данная комбинация позволяет уменьшить воздействие на здоровые клетки несущие на себе лишь один из этих рецепторов (« on-target off-tumor » токсичность) [13].
С целью упрощения конструкции разработана концепция универсальных CAR, которые с помощью искусственных молекул-адапто- ров (например, изотиоцианат флюоресцин (fluorescein isothiocyanate, FITC)) распознают альтернативные ТАА, что позволяет «настраивать» активность CAR T-клеток и делает их применение более безопасным [7].
Одна из многообещающих и наиболее распространенных [14] мишеней (57 %) — рецептор CD19, который экспрессируется на большинстве как нормальных, так и опухолевых В-лимфоцитов.
Технологии производства. Существует два возможных способа получения CAR T-клеток: in vivo (in situ) (в кровь пациента вводятся наночастицы, превращающие Т-клетки пациента в CAR T-клетки) и ex vivo (производство состоит из нескольких этапов).
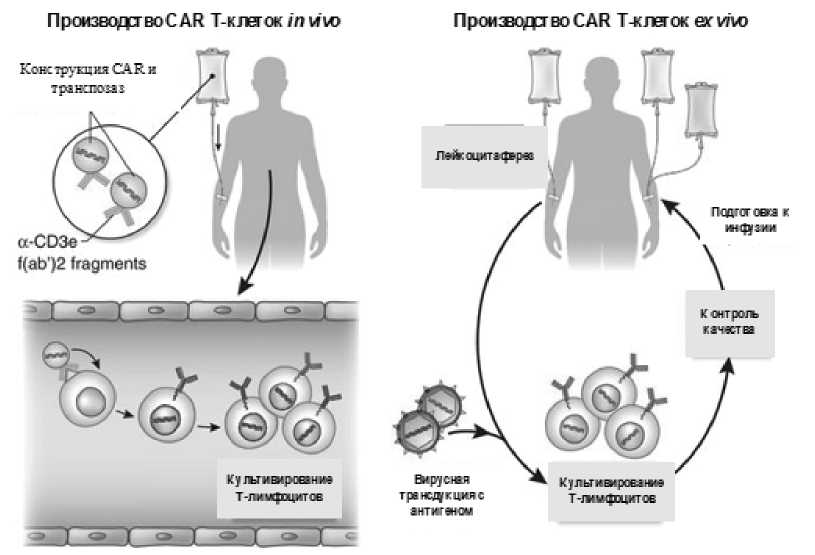
Рис. 3. Способы получения CAR T-клеток [15]
Этапы производства CAR T-клеток ex vivo [16]:
-
1) лейкоцитаферез;
-
2) вирусная /невирусная трансдукция с антигеном;
-
3) культивирование до идеальной дозы от 1 до 5 х 10 8 клеток;
-
4) контроль продукта (качество, стерильность);
-
5) подготовка к инфузии.
Клинические исследования . Из экспериментальной технологии лечение онкологических заболеваний CAR T-клетками превращается в быстро развивающийся подход к терапии с более чем 500 зарегистрированными клиническими исследованиями при гематологических новообразованиях и солидных опухолях. При этом большинство из них проводится в США и Китае [17].
Клинические исследования CD19 CAR T-клеток показали эффективность при онкоге-матологических заболеваниях, включая острый лимфобластный лейкоз (ОЛЛ), хронический лимфолейкоз (ХЛЛ) и неходжкинские лимфомы (НХЛ) [18].
Продемонстрирована активность BCMA-CAR T-клеток у пациентов с множественной миеломой (ММ) [19, 20], начаты испытания у пациентов с лимфомой Ходжкина (ЛХ) [21 22]. Продолжаются исследования в области применения CAR T-клеточной терапии при некоторых солидных опухолях (глиобластома, нейробластома, HER2-позитивная саркома) [23–25].
По данным систематизированного обзора и мета-анализа исследований в области CAR T-клеток за период 2008–2017 гг. [26] было опубликовано 60 исследований: 18 исследований (30 %) в области онкологии и 42 исследования в области гематологии (32 % — ОЛЛ 18 % — НХЛ, 3 % — ОМЛ, 2 % — ХЛЛ, 2 % — ЛХ 13 % — пациенты с двумя или более онкогема-тологическими диагнозами). В большинстве (57 %) — это исследования I фазы.
Аутологичные CAR T-клетки. В 2017 году Управление по контролю качества пищевых продуктов и лекарственных средств США (FDA) одобрило первые два препарата для CAR T-клеточной терапии: Kimriahтм (tisagenlecleucel CTL019, Novartis) [27] для лечения пациентов до 25 лет с рецидивирующими или рефрактерными (Р/Р) формами пре-В-клеточного острого лимфобластного лейкоза (пре-В-ОЛЛ) [28] и для лечения взрослых пациентов с Р/Р формами диффузной крупноклеточной В-клеточной лимфомы (ДКВКЛ) [29] и Yescartaтм (axicabtagene ciloleucel, Kite Pharma/Gilead) [30], для лечения взрослых пациентов с Р/Р формами ДКВКЛ [31]. Оба препарата являются CD19-CAR2-го поколения, с отличием в использованном векторе на этапе производства: лентивирусный вектор — Kimriahтм, ретровирусный — Yescartaтм. Другие CAR T-клетки являются исследовательскими продуктами в рамках международных или локальных клинических испытаний.
По данным многоцентрового исследования Yescartaтм II фазы у взрослых пациентов с рефрактерной формой ДКВКЛ (Zuma 1) частота общего ответа (ЧОО) составила 82 %, полной ремиссии (ПР) — 54 %. За период наблюдения (медиана — 15,4 месяца) у 40 % пациентов ПР сохраняется [32].
В исследовании Kimriahтм II фазы у пациентов с В-ОЛЛ ЧОО составила 81 %, ПР с отсутствием минимальной остаточной болезни (МОБ-негативная) — 60 %, МОБ-негативная ПР с не полностью восстановившимся кроветворением — 21 %. Бессобытийная выживаемость (БСВ) и общая выживаемость (ОВ) на период наблюдения 6 месяцев составили 73 % и 90 % соответственно, а на период 12 месяцев: БСВ — 50 %, а ОВ — 76 % [28].
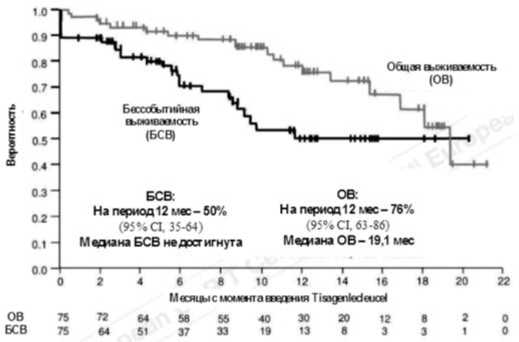
Рис. 4. Бессобытийная и общая выживаемости у пациентов с В-ОЛЛ при проведении терапии Kimriah тм [28]
Побочные эффекты терапии. Лечение онкологических заболеваний без возникновения токсичности и нежелательных явлений — редкость. Аутологичная CAR T-клеточная терапия не исключение,она имеет серьезные побочные эффекты и различный профиль токсичности.
Наиболее распространенными побочными эффектами являются синдром выброса цитокинов («цитокиновый шторм»);
нейротоксичность;
синдром лизиса опухоли;
аллергические реакции;
В-клеточная аплазия.
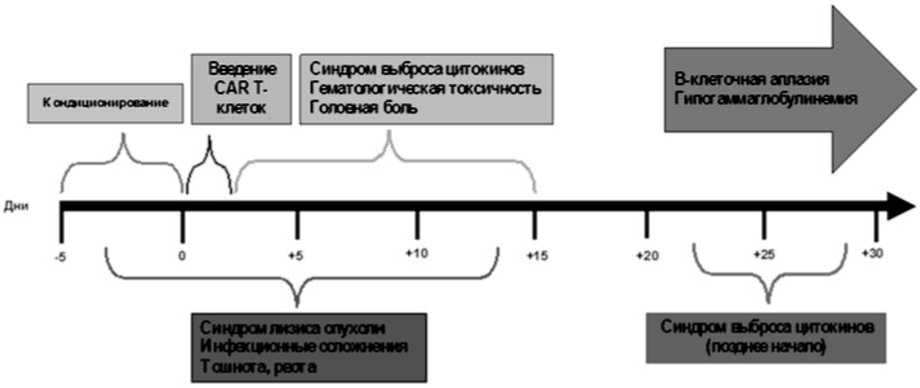
Рис. 5. Сроки возникновения токсичности [32, 33]
Таблица 2
Частота синдрома выброса цитокинов и нейротоксичности (адаптировано из [34])
|
Исследование |
Продукт |
Заболевание |
Синдром выброса цитокинов, степень 3/4 |
Нейро-токсичность, степень 3/4 |
|
ZUMA-1 |
Yescartaтм |
ДВККЛ и ПМВКЛ |
13 % |
28 % |
|
Transcend |
Tisangenlecleucel |
ДВККЛ |
1 % |
15 % |
|
Juliet |
Kimriahтм |
ДВККЛ |
22 % |
12 % |
|
CRB-401 |
BB2121 |
ММ |
2 % |
<1 % |
Синдром выброса цитокинов (СВЦ) — это жизнеугрожающее состояние, основными проявлениями которого являются гипотензия гипоксия, лихорадка и неврологические нарушения. Данное осложнение развивается в период от 1 до 10 дней (медиана — 3 дня) [34]. Факторами риска являются высокая опухолевая нагрузка, наличие активной инфекции на момент начала CAR T-клеточной терапии. Нейротоксичность развивается в течение 8 недель после введения препарата: медиана — 7 дней для пре-В-ОЛЛ и 12 дней для ДКВКЛ [34].
Основные проявления — изменение уровня сознания, галлюцинации, делирий, афазия атаксия, судороги, тремор. В-клеточная аплазия обусловлена деплецией пула нормальных В-лимфоцитов.
Проблемы CAR T-клеточной терапии и пути их преодоления. Возникновение рецидивов заболевания связано со снижением количества CD19 CAR T-клеток в крови пациента, либо обусловлено потерей экспрессии CD19 на поверхности опухолевых клеток.
Потеря эффективности
Потеря CD19 с поверхности клеток ОЛЛ

Потеря CAR-T клеток из крови

CD 19 рецидив
CD19+ рецидив
-
• CAR Т-клеткам требуется более высокая плотность таргетного антигена, чем естественным Т-клеткам.
Потеря активных CAR Т-клеток связана с их истощением
-
* Опухолевые клетки имеют низкую плотность CD 19
-
• Данный механизм резистентности «включается» у пациентов с высокой опухолевой нагрузкой
Е. \Л»л«, 1« European САЯ-I tai m<*1ift£ Рйга 5019
Рис. 6. Причины потери эффективности CAR T-клеточной терапии (адаптировано из [35])

Как улучшить CAR Т терапию?
Потеря CD19 с поверхности клеток ОЛЛ
Потеря CAR-T клеток из крови


CD19 рецидив

-
• -Двойное таргетирование»: CD19 и CD22
-
• Четвертое поколение CAR Т клеток
-
• Повышение эффективности CAR T клеток с помощью ингибиторов сигнального пути
• Повторная использование таргетирования* • Использование терапии, для истощения
CD19' рецидив

«двойного
комбинированной предотвращения
• Создание гуманизированного scFv против таргетных антигенов.
Е Wck-rg. Iм Етгермп С*ЯТ Рис. 7. Пути повышения эффективности CAR T-терапии (адаптировано из [35]) Заключение. Адоптивная иммунотерапия с использованием генно-модифицированных Т-лимфоцитов, безусловно, стала прорывным направлением в медицине и квинтэссенцией современной парадигмы онкологии и не только ведет к персонализированной медицине,но и дает надежду на значительное улучшение результатов лечения. Данные биотехнологии требуют высоких материальных (автоматизация технологического процесса), интеллектуальных и организационных ресурсов что предполагает их возможную реализацию (во всяком случае, на первоначальном этапе освоения) лишь в крупных научных центрах. Необходимо повышение как эффективности так и безопасности применения CAR T-клеток. В то же время, предстоит решить один из немаловажных вопросов: управляемость CAR T-клетками во временных, пространственных и функциональных измерениях. Нельзя отрицать, что адоптивная Т-клеточная иммунотерапия обладает потенциально высокой эффективностью. К сожалению, несмотря на то что появление CAR T-клеточной терапии открывает новые возможности лечения пациентов с онкологическими заболеваниями, частота достижения ответа ограничена,а появление рецидивов не редко. В терапии солидных опухолей успехи менее впечатляющие, чем в он-когематологии,что связано с особенностями биологии солидных опухолей и нерешенными вопросами стимулирования инфильтрации и персистирования CAR T-клеток в опухолевом микроокружении. Тем не менее, адоптивная иммунотерапия находится в начале своего пути и, возможно, пришло время для более тщательной оценки способов влияния на иммунную систему, которые позволят повысить эффек- тивность и минимизировать неблагоприятные явления связанные с CAR T-клеточной терапией в будущем. Вероятно, дальнейшие клинические исследования терапии CAR T-клетками будут направлены не столько на преодоление рефрактерности опухоли к стандартным программам лечения,сколько на решение проблемы эрадикации остаточного опухолевого клона на ранних этапах терапии, что откроет перспективы к излечению онкологических заболеваний в условиях реальной клинической практики.
Список литературы Современные представления о CAR T-клеточной терапии
- Smith AJ, Oertle J, Warren D, Prato D. Chimeric antigen receptor (CAR) T cell therapy for malignant cancers: Summary and perspective. Journal of cellular Immunotherapy. 2016;2:59-68.
- Schreiber RD, Old LJ, Smyth MJ. Cancer immunoediting: integrating immunity's roles in cancer suppression and promotion. Science. 2011;331:1565-70.
- Dunn GP, Bruce AT, Ikeda H, Old LJ, Schreiber RD. Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol. 2002; 3:991-8.
- ShankaranV, Ikeda H, Bruce AT, White JM, Swanson PE, Old LJ, et al. IFN-gamma and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature. 2011;410:1107-11.
- Gross G, Waks T, Eshhar Z. Expression of immunoglobulin-T-cell receptor chimeric molecules as functional receptors with antibody-type specificity. Proc Natl Acad Sci Unit States Am. 1989;86(24):10024-8.
- Pule MA, Savoldo B, Myers GD, Rossig C, Russell HV, Dotti G, et al. Virus-specific T cells engineered to coexpress tumor-specific receptors: persistence and antitumor activity in individuals with neuroblastoma. Nat Med. 2008;14(11):1264.
- Wen Si, Cheng Li, Ping Wei. Synthetic immunology: T-cell engeneering and adoptive immunotherapy. Synthetic and Systems Biotechnology.2018;3: 179-85.
- Chmielewski M, Abken H. Trucks: the fourth generation of CARs. Expert Opin Biol Ther. 2015;15: 1145-54.
- Antonana-Vidosola A, Perez-Amill L, Sune G, Castella M, Najjar A, Lozano E, Urbano-Ispizua A, Martin-Antonio B. BCMA CAR T-cell immunotherapy beyond myeloma: a novel target to fight B-cell non-Hodgkin lymphoma. 1st European CAR T-cell meeting, 14-16 February 2019, Paris. PI17/01043.
- Perez-Amill L, Sune G, Antonana-Vidosola A, Najjar A, Castella M, Lozano E, Urbano-Ispizua A, Martin-Antonio B. Pre-clinical evaluation of murine versus humanized BCMA-chimeric antigen receptor modified T cells (ARI2) for Multiple Myeloma treatment. 1st European CAR T-cell meeting, 14-16 February 2019, Paris. PI17/01043, PI14/00798.
- Leivas A, Valeri A, Rio P, Garcia-Ortiz A, Pacello ML, Perez-Martinez A, Lee D, Martinez-Lopez J. NKG2D-CAR transduced primary natural killer cells efficiently target multiple myeloma cells. 1st European CAR T-cell meeting, 14-16 February 2019, Paris.
- Hegde M, Mukherjee M, Grada Z, Pignata A, Landi D, Navai SA, et al. Tandem CAR T cells targeting HER2 and IL13Ralpha2 mitigate tumor antigen escape. J Clin Invest.2016;126(8):3036-52.
- Bister A, Haist C, Schulte E, Scheckenbach K, Hanenberg H, Gattermann N, Wiek C. Dual targeting to reduce on-target off-tumor toxicities in CAR T-cell therapy for mantle cell lymphoma. 1st European CAR T-cell meeting, 14-16 February 2019, Paris.
- Grigor EJM, Fergusson D, Kekre N, et al. Risks and Benefits of Chimeric Antigen Receptor T-Cell (CAR-T) Therapy in Cancer: A Systematic Review and Meta-Analysis, Transfusion Medicine Reviews. 2019. https://doi. org/10.1016/ j. tmrv.2019.01.005.
- Olweus J. Manufacture of CAR-T cells in the body. Nature Biotechnology.2017;35:520-1.
- Zhao Z, Chen Y, Francisco NM, Zhang Y, Wu M. The application of CAR T-cell therapy in hematological malignancies: advantages and challenges. Acta Pharmaceutica Sinica B. 2018;8(4):539-551.
- Li Y, Huo Y, Yu L et al. Quality Control and Nonclinical Research on CAR-T Cell Products: General Principles and Key Issues, Engineering. 2019. https://doi. org/10.1016/j. eng.2018.12.003.
- Brudno JN, Kochenderfer JN. Recent advances in CAR T-cell toxicity: Mechanisms, manifestations and management. Blood Reviews. 2019. https://doi. org/10.1016/j. blre.2018.11.002.
- Brudno JN, Maric I, Hartman SD, Rose JJ, Wang M, Lam N, et al. T cells genetically modified to express an anti-B-cell maturation antigen chimeric antigen receptor cause remissions of poor-prognosis relapsed multiple myeloma. J Clin Oncol Aug. 2018;36(22):2267-80.
- Mikkilineni L, Kochenderfer JN. Chimeric antigen receptor T-cell therapies for multiple myeloma. Blood. 2017;130:2594-602.
- Wang CM, Wu ZQ, Wang Y, Guo YL, Dai HR, Wang XH, et al. Autologous T cells expressing CD30 chimeric antigen receptors for relapsed or refractory hodgkin lymphoma: an open-Label phase I trial. Clin Cancer Res. 2017;23:1156-66.
- Ramos CA, Ballard B, Zhang H, Dakhova O, Gee AP, Mei Z, et al. Clinical and immunological responses after CD30-specific chimeric antigen receptor-redirected lymphocytes. J Clin Invest. 2017;127(9):3462-71.
- Brown CE, Alizadeh D, Starr R, Weng L, Wagner JR, Naranjo A, et al. Regression of glioblastoma after chimeric antigen receptor T-cell therapy. N Engl J Med. 2016;375:2561-9.
- Louis CU, Savoldo B, Dotti G, Pule M, Yvon E, Myers GD, et al. Antitumor activity and long-term fate of chimeric antigen receptor-positive T cells in patients with neuroblastoma. Blood. 2011;118:6050-6.
- Ahmed N, Brawley VS, Hegde M, Robertson C, Ghazi A, Gerken C, et al. Human epidermal growth factor receptor 2 (HER2) -specific chimeric antigen receptor- modified T cells for the immunotherapy of HER2-positive sarcoma. J Clin Oncol. 2015;33:1688-96.
- Grigor EJM, Fergusson D, Kekre N, et al. Risks and Benefits of Chimeric Antigen Receptor T-Cell (CAR-T) Therapy in Cancer: A Systematic Review and Meta-Analysis. Transfusion Medicine Reviews. 2019. https://doi. org/10.1016/ j. tmrv.2019.01.005.
- Novartis (2017) Prescribing Information (KymriahH). Available at: https://www. pharma. us. novartis. com/ sites/www. pharma. us. novartis. com/files/kymriah. pdf
- Maude SL, Laetsch TW, Buechner J, Rives S, Boyer M, Bittencourt H, et al. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N Engl J Med. 2018;378(5):439-48.
- Schuster SJ, Svoboda J, Chong EA, Nasta SD, Mato AR, Anak O, et al. Chimeric antigen receptor T cells in refractory B-cell lymphomas. N Engl J Med. 2017;377:2545-54.
- YescartaH (2017) Prescribing Information. Available at: https://www. fda. gov/downloads/ BiologicsBloodVaccines/ CellularGeneTherapyProducts/ApprovedProducts/ UCM581226. pdf
- Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med. 2017;377(26):2531-44.
- Neelapu SS, Tummala S, Kebriaei P, Wierda W, et al. Chimeric antigen receptor T-cell therapy — assessment and management of toxicities. Rev Clin Oncol. 2018; 15: 47-62.
- Shank BR. Management of CAR T-cell Adverse Effects in hematological malignancies. HOPA Annual meeting. 2018 March.
- Ellard R, 1st European CAR T-cell meeting, 14-16 February 2019, Paris.
- Vickers E. What is driving CARs into the spotlight?-the science simplified. 1st European CAR T-cell meeting, 14-16 February 2019, Paris.


