Synergistic effect of potassium humate and urea on wheat grain germination indicators: germination, root surface area and antioxidant response
Author: Yang Fan, Potoroko I.Yu.
Section: Биохимический и пищевой инжиниринг
Article in issue: 1 т.14, 2026.
Free access
Humic substances are widely used as plant biostimulants, yet their growthpromoting efficacy depends on nutrient background and growth stage. Here, we evaluated potassium humate (Khumate) alone and in combination with urea for wheat (Triticum aestivum L.) performance in a twophase assay. In a Petridish germination test (DI water vs 0.1 % Khumate), germination was scored at day 3 using a radicle length threshold of 2 mm. Subsequently, seedlings were transferred to hydroponics and subjected to three treatments: G1 (Khumate during germination followed by Khumate + urea), G2 (Khumate followed by Khumate), and G3 (DI water control). At day 12, shoot and root fresh mass, root surface area (SmartRoot), TTC reduction, ascorbate peroxidase (APX) activity, and chlorophyll content were quantified. Germination percentages at day 3 were similar between DI water (59.2 %) and 0.1 % Khumate (56.0 %). Root surface area differed significantly among treatments (oneway ANOVA, F = 8.35, p = 0.0015, n = 10), peaking in G1 (9.91 ± ± 3.51) and exceeding G3 (5.05 ± 1.42). Biomass accumulation was highest in G1, while APX activity increased ~4.8–4.9fold relative to G2 and G3, indicating a stronger antioxidant response under the combined Khumate + nitrogen regime. Overall, the results support that Khumate benefits wheat seedlings primarily when paired with an available nitrogen source, translating into larger absorptive root surface area and higher biomass.
Humic substances, potassium humate, urea, Triticum aestivum, root architecture, SmartRoot, APX, TTC
Short address: https://sciup.org/147253476
IDR: 147253476 | UDC: 633.13:631.82-027.236 | DOI: 10.14529/food260107
Синергетическое воздействие гумата калия и мочевины на показатели прорастания зерна пшеницы: всхожесть, площадь поверхности корней и антиоксидантная реакция
Гуминовые вещества широко используются в качестве биостимуляторов для растений, однако их эффективность в стимулировании роста зависит от питательного фона и стадии развития. В данном исследовании мы оценили эффективность гумата калия (K-гумата) как отдельно, так и в сочетании с мочевиной для пшеницы (Triticum aestivum L.) в двухфазном эксперименте. В чашке Петри (тест с дистиллированной водой против 0,1 % K-гумата) всхожесть оценивали на 3-й день, используя пороговое значение длины корешка 2 мм. Затем рассаду пересаживали в гидропонную систему и подвергали трем вариантам обработки: G1 (K-гумат во время прорастания, затем K-гумат + мочевина), G2 (K-гумат, за-тем K-гумат) и G3 (контроль с дистиллированной водой). На 12-й день определяли свежую массу побегов и корней, площадь поверхности корней (SmartRoot), восстановление TTC, активность аскорбатпероксидазы (APX) и содержание хлорофилла. Процент всхожести на 3-й день был схожим при использовании дистиллированной воды (59,2 %) и 0,1 % гумата калия (56,0 %). Площадь поверхности корней значительно различалась между вариантами обработки (однофакторный дисперсионный анализ, F = 8,35, p = 0,0015, n = 10), достигая пика в варианте G1 (9,91 ± 3,51) и превышая вариант G3 (5,05 ± 1,42). Накопление биомассы было самым высоким в варианте G1, в то время как активность APX увеличилась при-мерно в 4,8–4,9 раза по сравнению с вариантами G2 и G3, что указывает на более сильную антиоксидантную реакцию в условиях комбинированного режима гумата калия + азота. В целом результаты подтверждают, что гумат калия приносит пользу всходам пшеницы в первую очередь в сочетании с доступным источником азота, что приводит к увеличению площади поглощающей поверхности корней и повышению биомассы.
Text of the scientific article Synergistic effect of potassium humate and urea on wheat grain germination indicators: germination, root surface area and antioxidant response
Wheat ( Triticum aestivum L .) provides a major share of calories and protein for the global population, and improving its early growth remains a central objective for sustainable intensification. Seedling establishment and the early development of the root system are critical determinants of nutrient capture, stand uniformity, and subsequent yield potential, particularly under fluctuating resource availability. In parallel, the growing pressure to reduce fertilizer losses and environmental impacts has accelerated interest in plant biostimulants that can enhance nutrient use efficiency and stress tolerance while maintaining productivity.
Plant biostimulants have been broadly defined as substances or microorganisms applied to plants to improve nutrient efficiency, abiotic stress tolerance, and/or quality traits, irrespective of their direct nutrient contribution [13]. Among the major categories of biostimulants, humic substances (HS) and their soluble salts (humates) represent one of the most widely used inputs, owing to their abundance in natural organic matter and their long history in agricultural practice [1–3]. However, HS-based products exhibit variable performance across studies due to differences in source material, extraction method, mo- lecular characteristics, concentration, application route, and baseline fertility conditions [8, 14]. Disentangling these factors using controlled systems can help identify conditions under which HS inputs are most likely to deliver consistent benefits.
Humic substances are complex, heterogeneous assemblies formed during the decomposition and transformation of plant and microbial residues (humification). Beyond their indirect effects mediated through soil structure, pH buffering, and cation exchange capacity, HS can directly affect plant physiology [3, 4]. Multiple lines of evidence suggest that HS promote root system development by stimulating plasma membrane H+-ATPase activity, enhancing cell expansion, and triggering hormone-like signaling [4, 19, 20]. For example, humic acids have been shown to induce lateral root formation via pathways related to indole-3-acetic acid (IAA) and proton pump activation [20], and to interact with signaling mediators such as nitric oxide (NO) and abscisic acid (ABA), which modulate root hydraulics and root-to-shoot communication [10]. These processes can increase root length density and surface area, thereby enlarging the plant’s absorptive interface and potentially improving nutrient acquisition under limiting conditions.
Nitrogen (N) availability is a central context for interpreting HS responses. Nitrogen often limits shoot growth and chlorophyll biosynthesis, and it also shapes root allocation patterns. Biostimulant-induced changes in root architecture are more likely to translate into biomass accumulation when plants can access sufficient mineral nutrients to support new tissue construction. Accordingly, reviews and meta-analyses emphasize that HS effects are frequently conditional on baseline fertility and can be amplified when HS are integrated with mineral fertilization programs [5, 7, 14]. Despite this consensus, experimental datasets that explicitly compare HS alone versus HS combined with an accessible N source under identical conditions remain useful, especially for interpreting physiological indicators and root traits.
Hydroponic systems provide a convenient platform to examine direct plant responses to soluble inputs while reducing confounding soil variability. In wheat, hydroponic studies are commonly used to quantify early root traits and enzyme activities that are difficult to interpret in soil. In addition to morphological traits, antioxidant enzymes such as ascorbate peroxidase (APX) are often responsive to growth stimulation and redox signaling. Humic-induced growth enhancement has been linked to ROS-mediated signaling, where moderate ROS can function as
Germination Phase (Days 1-7)
Treatment 1: Control
Treatment 2: K-Humate
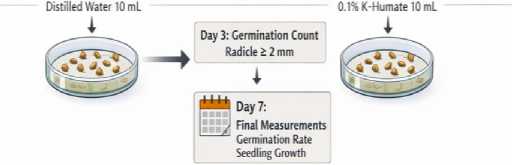
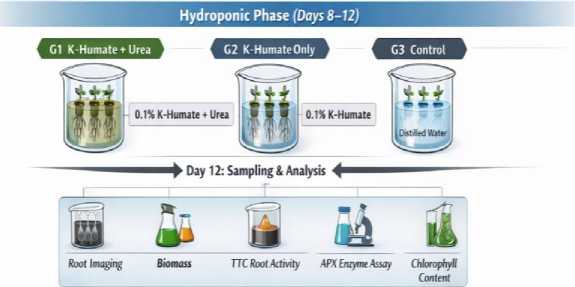
Fig. 1. Two-phase experimental workflow and sampling schedule
messengers while antioxidant systems protect cells from oxidative damage [4, 14, 16]. Therefore, measuring antioxidant responses together with root architecture provides a more integrated view of plant performance.
The present study used a two-phase workflow (germination followed by short-term hydroponics) to evaluate the effects of potassium humate (K-humate) and its combination with urea on wheat performance. We asked three questions: (i) does 0.1 % K-humate influence early germination (day 3)? (ii) does K-humate alter seedling root surface area and biomass at day 12, and are effects enhanced when urea is supplied? and (iii) do biochemical indicators (TTC reduction, APX activity, chlorophyll concentration) suggest physiological mechanisms associated with the observed growth patterns? We hypothesized that K-humate would have limited impact on early germination but would promote root surface area more strongly when paired with urea, and that accelerated growth would coincide with elevated antioxidant enzyme activity.
Materials and methods
Plant material and chemicals. Wheat seeds were selected for uniform size and absence of visible damage. Potassium humate (K-humate) was prepared as a 0.1 % (w/v) aqueous solution using deionized (DI) water. Urea was prepared as a 0.1 % (w/v) aqueous solution. For the combined solution used in G1 during hydroponics, 100 mL of 0.1 n% K-humate was added to 1 L of 0.1 % urea solution and mixed thoroughly. All solutions were prepared fresh or stored in the dark at 4 °C for short periods to minimize chemical alteration.
Two-phase experimental design and treatments. The workflow consisted of a Petri-dish germination phase (day 0-7) followed by a hydroponic seedling phase (day 8-12) (Figure 1). In Phase I, germination was compared between DI water and 0.1 % K-humate. In Phase II, three treatments were established: G1 (0.1 % K-humate in Phase I; 0.1 % K-humate + 0.1 % urea in Phase II), G2 (0.1 % K-humate in Phase I; 0.1 % K-humate in Phase II), and G3 (DI water throughout). A summary of treatment composition is provided in Table 1.
Table 1
Treatment composition used in the hydroponic phase (day 8–12)
|
Treatment |
Germination phase (day 0–7) |
Hydroponic phase (day 8–12) |
|
G1 |
0.1 % K-humate |
0.1 % K-humate + 0.1 % urea |
|
G2 |
0.1 % K-humate |
0.1 % K-humate |
|
G3 |
DI water |
DI water |
Seed sterilization and germination assay. Seeds were surface-sterilized in 75 % (v/v) ethanol for 1 min and rinsed three times with DI water. For each treatment, seeds were placed in 9cm Petri dishes lined with two layers of filter paper and supplied with 10 mL of the corresponding solution. Fifty seeds were arranged per dish and incubated at 25 °C. Germination was scored at day 3 using the criterion that the radicle emerged and reached a length of at least 2 mm. Germination percentage was calculated as (germinated seeds / total seeds) × 100.
Hydroponic cultivation and sampling. On day 8, seedlings of similar size were transferred to hydroponic containers (beakers) containing the assigned solutions. Seedlings were grown under laboratory conditions; solutions were replenished to maintain volume and to reduce concentration drift. At day 12, seedlings were harvested. Shoots and roots were separated and fresh mass was recorded. Fresh mass is presented as the sum of 10 seedlings per treatment. For root trait measurements, 10 individual seedlings per treatment served as biological units.
Root imaging and surface area quantification. Roots from each seedling were gently spread in a thin water layer to reduce overlap and photographed under consistent illumination. Root traits were quantified using SmartRoot, a semiautomated root image analysis toolbox implemented as an ImageJ plugin [6]. ImageJ is a widely used open platform for biological image processing [15]. In SmartRoot, primary and lateral roots were traced to generate vector representations, from which root surface area was calculated. Because all images were acquired under the same settings and analyzed using the same workflow, the surface area output is reported in consistent arbitrary units suitable for between-treatment comparisons.
TTC reduction assay (root metabolic activity). Root metabolic activity was estimated using the reduction of 2,3,5-triphenyltetrazolium chloride (TTC) to red triphenylformazan, which can be extracted and measured at 485 nm [17]. Absorbance values are presented as relative units. The available dataset represents technical replicates of assays performed on pooled root material; thus, results are interpreted descriptively.
APX activity assay. Ascorbate peroxidase (APX) activity was quantified as a proxy for antioxidant capacity. Absorbance was recorded at time 0 s (A0) and 100 s (A100s), and ΔA = A0 – – A100s. APX activity was calculated as APX = = ΔA × 144.05 (nmol s–1 g–1 DW). Given limited biological replication in the available dataset, APX values are interpreted as indicative trends.
Chlorophyll determination. Leaf chlorophyll was extracted with 95 % ethanol. Absorbance was measured at 665 and 649 nm. Chlorophyll a, chlorophyll b, and total chlorophyll were calculated using established spectrophotometric equations for ethanol extracts [18]. Total chlorophyll concentration is reported in mg L–1 for comparability among treatments under the same extraction conditions.
Statistical analysis. Root surface area was analyzed by one-way ANOVA with treatment as the fixed factor (n = 10 seedlings per treatment). Post-hoc Tukey HSD testing was performed at α = 0.05. Other indicators were not subjected to inferential statistics due to limited biological replication and are presented as descriptive comparisons.
Results and discussion
Germination at day 3. Day-3 germination percentages were 59.2 % in the DI water control and 56.0 % in 0.1 % K-humate (Table 2). Under the applied criterion (radicle length ≥ 2 mm), the difference was small, suggesting that K-humate had no clear promotive effect on early germination.
Table 2
Germination percentage scored at day 3 (radicle length ≥ 2 mm)
|
Treatment |
Germination (%) |
|
DI water |
59.2 |
|
0.1 % K-humate |
56.0 |
Seedling fresh biomass at day 12. Shoot and root fresh mass at day 12 (sum of 10 seedlings per treatment) indicated the highest biomass accumulation in G1 (Table 3; Figure 2). Compared with the DI water control (G3), G1 increased shoot fresh mass and markedly increased root fresh mass, consistent with enhanced root development under K-humate + urea.
Table 3
Shoot and root fresh mass at day 12 (sum of 10 seedlings)
|
Treatment |
Shoot fresh mass (g) |
Root fresh mass (g) |
|
G1 |
0.6168 |
0.3762 |
|
G2 |
0.5271 |
0.3243 |
|
G3 |
0.5644 |
0.2830 |
Root system architecture and surface area. Representative root images illustrate qualitative differences in root system size among treatments
(Figure 2). Quantitative analysis revealed significant treatment effects on root surface area (oneway ANOVA, df = 2,27; F = 8.35; p = 0.0015; n = 10). Mean root surface area was highest in G1 (9.91 ± 3.51), intermediate in G2 (7.18 ± ± 2.64), and lowest in G3 (5.05 ± 1.42) (Table 4; Figure 3 and 4). Tukey HSD indicated that G1 exceeded G3 at p < 0.05, while G2 was not significantly different from either group.
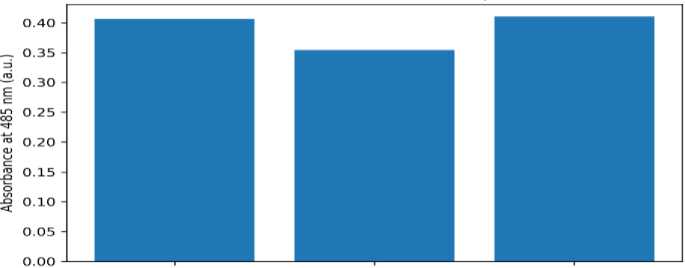
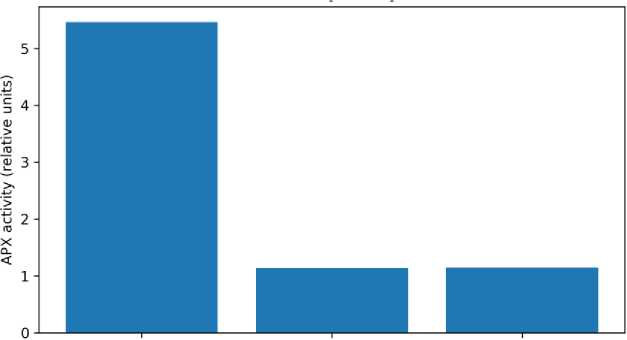
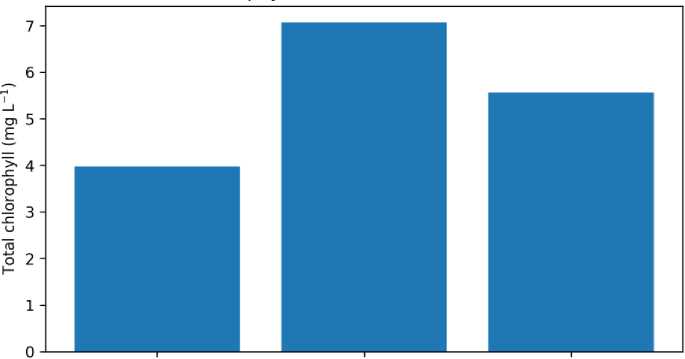
Physiological and biochemical indicators. TTC reduction, APX activity, and chlorophyll concentration are summarized in Table 5 and Figures 5–7. TTC absorbance at 485 nm was similar for G1 (0.4067) and G3 (0.4105) but lower for G2 (0.3545), suggesting reduced TTC reduction capacity in the continuous K-humate treatment. APX activity was strongly elevated in G1 (5.46) compared with G2 (1.14) and G3 (1.15), indicating a pronounced induction of antioxidant capacity under K-humate + urea. Total chlorophyll concentration (mg L–1) ranked
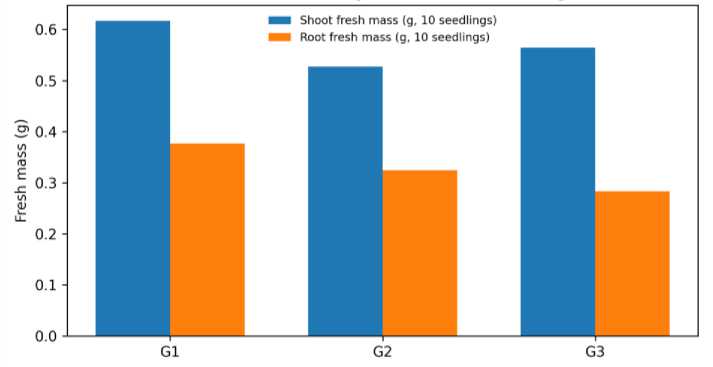
Fig. 2. Shoot and root fresh mass at day 12 (sum of 10 seedlings per treatment)
G3 Water
G2 Humate
G1 Humate+Urea

Fig. 3. Representative root system images for each treatment group at 12 day
Table 4
Root surface area at day 12 (mean ± SD; n = 10)
|
Treatment |
Mean |
SD |
Tukey group |
|
G1 |
9.91 |
3.51 |
a |
|
G2 |
7.18 |
2.64 |
ab |
|
G3 |
5.05 |
1.42 |
b |
Table 5
Physiological and biochemical indices at day 12
|
Treatment |
TTC absorbance (485 nm) |
APX (relative units) |
Total chlorophyll (mg L–1) |
|
G1 |
0.4067 |
5.46 |
3.98 |
|
G2 |
0.3545 |
1.14 |
7.07 |
|
G3 |
0.4105 |
1.15 |
5.57 |
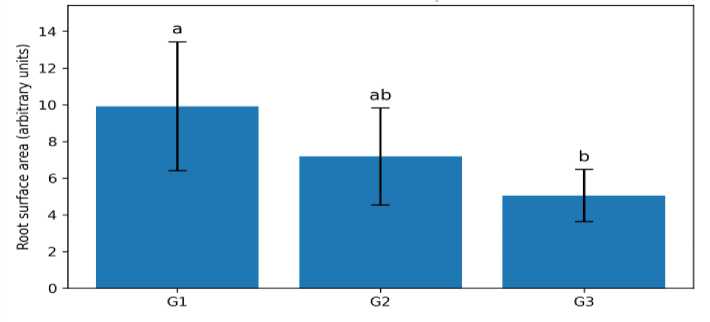
Fig. 4. Root surface area at day 12 (mean ± SD, n = 10). Different letters indicate significant differences by Tukey HSD (p < 0.05)
G1 G2 G3
Fig. 5. Root TTC reduction measured as absorbance at 485 nm (technical replicates)
G1 G2 G3
Fig. 6. APX activity at day 12 (relative units; limited replication)
G1 G2 G3
Fig. 7. Total chlorophyll concentration in ethanol extracts (mg L –1 )
G2 (7.07) > G3 (5.57) > G1 (3.98). Because these indices were obtained with limited biological replication, they are interpreted as descriptive trends rather than definitive statistical outcomes.
This study provides a compact dataset linking potassium humate application to wheat seedling root architecture and physiological indicators, with an explicit comparison of humate alone versus humate combined with a nitrogen source. The two-phase design separated early germination processes from subsequent seedling growth, enabling clearer interpretation of stage-specific responses.
Limited effects on early germination. K-humate did not improve day-3 germination percentage under the adopted scoring criterion (radicle ≥ 2 mm). This outcome is consistent with the general expectation that HS inputs often exert more pronounced effects after germination, during phases characterized by active root elongation and branching. At early stages, germination is largely driven by seed hydration, reserve mobilization, and intrinsic hormonal control; therefore, external HS may not readily alter the initiation threshold. Moreover, the direction and magnitude of HS effects can vary with concentration and product composition, and it is plausible that the 0.1 % concentration used here was not optimal for germination promotion in wheat.
Root surface area as a sensitive morphological endpoint. In contrast to germination, root surface area at day 12 responded strongly to treatments. The significant increase in G1 relative to G3 supports the view that HS can stimulate root system development, enhancing absorptive capacity [4, 9]. Mechanistically, HS-induced root proliferation has been associated with stimulation of plasma membrane H+-ATPase activity and auxin-like signaling, which together facilitate cell expansion and lateral root emergence [20]. Further, HS have been implicated in NO- and ABA-mediated pathways that regulate root hydraulics and root-to-shoot signaling [10], providing a plausible mechanistic bridge to enhanced shoot growth.
Synergy with nitrogen supply. A key observation is that the largest root surface area occurred when K-humate was combined with urea (G1). Meta-analyses and reviews highlight that HS responses depend strongly on environmental and nutritional context, and that synergistic effects with mineral fertilization are frequently reported [5, 14]. In the present dataset, urea likely provided readily available nitrogen that supported new tissue construction, allowing humate-induced architectural remodeling to translate into measurable increases in surface area and biomass. Because root surface area is closely related to nutrient uptake kinetics, increasing surface area under adequate N supply can generate a positive feedback loop: larger roots increase nutrient acquisition, which supports greater shoot growth and photosynthetic capacity, enabling further root development.
Antioxidant response and growth regulation. APX activity was markedly higher in G1 than in G2 and G3. This may reflect an elevated antioxidant requirement to sustain higher metabolic rates during rapid growth, or it may indicate that humate + urea triggered redox-related signaling. Humic-induced growth promotion has been linked to ROS and NO as signaling components.
Under such models, controlled ROS production can act as a growth signal, whereas antioxidant enzymes maintain redox homeostasis to prevent damage. Thus, elevated APX in G1 is consistent with a scenario where humate plus nitrogen enables vigorous growth accompanied by stronger redox regulation.
Interpretation of chlorophyll and TTC trends. Total chlorophyll concentration was lowest in G1, despite the apparent biomass advantage. Several explanations are possible. First, a dilution effect may occur if leaves expand faster or contain more water, reducing pigment concentration per extract volume even if total pigment per plant increases. Second, nitrogen partitioning and chlorophyll turnover may differ among treatments, particularly in short-term assays. Because chlorophyll was quantified using ethanol-based spectrophotometry [18], extending the analysis to chlorophyll per unit leaf area or per plant dry mass would clarify interpretation. TTC reduction suggested lower activity in G2 relative to G1 and G3; if confirmed, this could indicate constrained respiratory metabolism under continuous humate without supplemental nitrogen. However, TTC measurements are sensitive to sampling (e.g., root tips versus whole roots) and extraction conditions [17], so replicated assays standardized by root mass are recommended.
Limitations and future work. The main limitation of the current dataset is replication structure: root surface area was measured on 10 biological units per treatment and supported inferential testing, whereas other indices were measured with limited biological replication. Future experiments should implement independent replicate containers as experimental units, increase sample size for biochemical assays, and include additional endpoints such as total root length, root volume, nitrogen content, and nitrate reductase activity. Integrating ion analyses and transcriptlevel markers for nutrient transporters and H+-ATPase isoforms could further elucidate mechanisms proposed in HS literature [10, 20].
Taken together, the evidence supports a conditional model of K-humate bioactivity: humate alone showed modest or inconsistent effects, whereas humate plus urea produced stronger and more coherent improvements in root architecture and growth-related physiology. This finding aligns with the broader biostimulant framework emphasizing context dependence and integration with fertility management [3, 5, 13, 14].
Quantitative interpretation and effect sizes. To complement significance testing for root surface area, we calculated simple effect sizes relative to the DI water control (G3). Root surface area increased by approximately 96.2 % in G1 and 42.2 % in G2 compared with G3, highlighting a large morphological response when urea was included. Similarly, pooled root fresh mass increased by 32.9 % in G1 relative to G3, while shoot fresh mass increased by 9.3 %. Although pooled biomass values cannot be statistically tested in the present dataset, the directionality is consistent with root surface area differences, supporting the hypothesis that increased absorptive interface drives improved whole-seedling growth (Table 6).
Practical implications for integrating humate products with nitrogen management. From an agronomic perspective, the results suggest that potassium humate functions most effectively as a co-input rather than as a stand-alone growth promoter under low-nutrient conditions. In practical settings, humate products are often marketed for improving root growth and nutrient efficiency; this study supports that such claims are more likely to be realized when plants can access a mineral N source. For early wheat establishment, this implies that applying humate together with an initial N supply (e.g., starter fertilization, fertigation, or urea-amended irrigation) may better leverage humate-driven root branching and surface area expansion. Conversely, applying humate without adequate N may yield
Table 6
Effect size summary relative to the DI water control (G3)
|
Metric |
G1 vs G3 |
G2 vs G3 |
Notes |
|
Root surface area |
+96.2 % |
+42.2 % |
Mean of n=10 seedlings; ANOVA significant |
|
Root fresh mass (sum) |
+32.9 % |
+14.6 % |
Sum of 10 seedlings; descriptive |
|
Shoot fresh mass (sum) |
+9.3 % |
+-6.6 % |
Sum of 10 seedlings; descriptive |
|
APX activity |
4.75x |
0.99x |
Relative units; limited replication |
modest or inconsistent outcomes, as suggested by the intermediate and sometimes lower physiological indicators in G2. Because urea hydrolysis and N availability dynamics differ in soil compared with hydroponics, subsequent soil-based trials should evaluate timing (seed treatment, infurrow, early topdressing) and dose to identify robust field-relevant protocols.
Proposed mechanistic framework. Based on the observed patterns and prior mechanistic literature, we propose a conceptual framework in which K-humate acts as a signaling-active input that primes root developmental pathways, while accessible nitrogen supplies the resources needed for sustained tissue construction (Figure 8). At the root level, humate may trigger auxin/NO/ABA-related signaling and activate plasma membrane H+-ATPase, promoting cell expansion and lateral root emergence [10, 20]. When nitrogen is sufficient, these architectural changes expand the absorptive interface and improve nutrient uptake, supporting higher biomass and a more robust antioxidant system (e.g., APX).
Relationship to previous studies and broader evidence. Most mechanistic work on humic substances has been performed on horticultural species and model plants, while cereal-specific evidence is more dispersed. Nevertheless, the direction of the response observed here-enhanced root development under humate inputs, especially when nutrients are not limiting – is aligned with several general trends reported for HS-based biostimulants. Meta-analyses indicate that root traits often respond more strongly than shoot traits, reflecting the direct contact of HS with the rhizosphere and root epidermis. In addition, studies describing humate-induced stimulation of H+-ATPase and auxin-related pathways provide a mechanistic explanation for why branching and surface area are particularly sensitive endpoints.
When comparing the magnitude of our root surface area response (~96 % increase in G1 vs the DI control), it is important to consider that our assay was short-term and conducted in hydroponics. Hydroponic conditions can magnify root trait differences because nutrient delivery is homogeneous, root observation is direct, and confounding soil constraints are absent. In soil, physical impedance, heterogeneous nutrient patches, and microbial interactions can moderate – or in some cases enhance-biostimulant responses. For example, HS can act as chelators and redox buffers and can interact with microbial communities, which can amplify plant responses in natural systems [4, 11]. Therefore, while the present effect size provides evidence of potential, translating the magnitude to field expectations requires caution.
The strong APX induction in G1 is also consistent with a growing body of work describing HS as redox-active inputs. Humic fractions can contain quinone-like moieties and redox-active functional groups that may influence plant redox state directly or indirectly. In plants, redox regulation is tightly coupled to growth through modulation of cell wall loosening, hormone signaling, and defense readiness. Thus, the observed APX response could reflect both (i) a higher metabolic flux associated with faster growth and (ii) an HS-triggered signaling response in which antioxidant enzymes are upregulated as part of a controlled
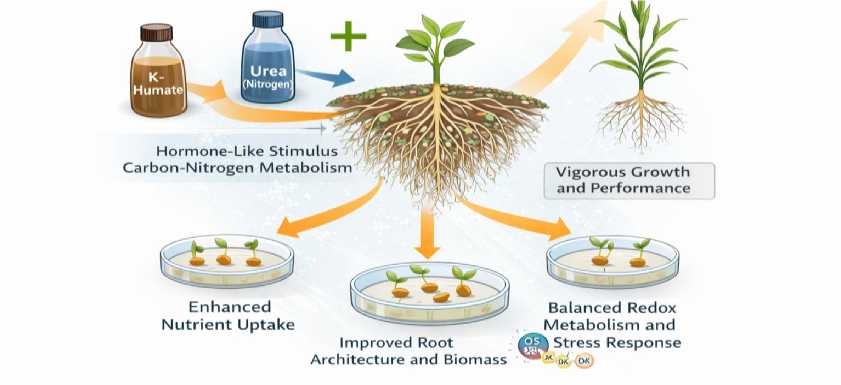
Fig. 8. Conceptual model of synergy between K-humate and nitrogen supply in promoting root development and seedling performance
redox program. Disentangling these alternatives would require replicated measurements of additional redox markers (e.g., H2O2, malondialdehyde, glutathione redox state) and growth rate over time.
Nitrogen form and transformation should also be considered. Urea is hydrolyzed to ammonium and subsequently nitrified to nitrate in soil, whereas in hydroponics the availability dynamics can differ depending on microbial activity and solution chemistry. HS have been reported to influence nitrate uptake and root-to-shoot nitrate translocation through signaling effects, which can interact with cytokinin distribution and shoot growth [8, 10]. Although we did not directly measure nitrate or ammonium uptake, the observed synergy between humate and urea is compatible with the hypothesis that nitrogen availability interacts with humate-driven signaling to promote both root architecture and whole-plant growth. Future work measuring tissue N concentration and expression of N transporter genes would help link the phenotypes to nitrogen assimilation mechanisms.
Finally, our study uses potassium humate, which provides both humic components and potassium ions. While the humic fraction is the focus, potassium is also an essential macronutrient involved in osmotic regulation and enzyme activation. Because the DI control did not contain added K, some fraction of the response could be influenced by potassium availability. However, the treatment ranking (G1 > G2 > G3 for root surface area) and the large incremental effect of adding urea to humate (G1 vs G2) suggest that nitrogen availability and humate–N interactions were major drivers. A useful control in follow-up experiments would be a potassium-matched treatment without humate to separate K effects from humate bioactivity.
Recommendations for replication, reporting, and data integration. To strengthen inference and facilitate comparison across studies, future experiments should adopt a replication structure in which the hydroponic container (or growth tray) is treated as the experimental unit and multiple independent containers are used per treatment. This avoids pseudoreplication that can occur when multiple seedlings share the same solution environment. In addition, reporting should include key environmental variables (temperature, photoperiod, light intensity, solution pH and elec- trical conductivity) because HS bioactivity and nutrient availability can be sensitive to these parameters. Root phenotyping can be expanded beyond surface area to include total root length, mean diameter, lateral root density, specific root length, and root hair traits, which together provide a more complete functional profile. SmartRoot enables the extraction of several of these traits [6], and consistent analysis workflows (including thresholds for tracing and handling of overlapping roots) should be documented to ensure reproducibility. Where possible, providing raw or minimally processed root images as supplementary material can further support transparent reporting.
For biochemical assays, increasing biological replication is essential. APX and TTC can vary with root age, sampling position, and diurnal rhythms. Standardizing sampling (e.g., using defined root segments, sampling at a consistent time of day) and reporting enzyme activity on both fresh and dry mass bases can improve interpretability. In addition to APX, measuring superoxide dismutase (SOD), catalase (CAT), and peroxidase (POD) activities would provide a broader view of antioxidant regulation, while lipid peroxidation and electrolyte leakage can help distinguish signaling-related responses from stress injury.
Because HS products differ in composition, describing the humate source and basic properties (e.g., pH, ash content, elemental composition, UV – Vis indices) is valuable for reproducibility and for linking structure to function. The supramolecular view of HS emphasizes that biological activity may depend on the assembly state and functional group exposure [12]. Therefore, even simple characterization (e.g., total acidity, carboxyl/phenolic content, or spectroscopic indices) can improve the scientific utility of HS studies.
Finally, translating hydroponic responses to agronomic recommendations requires multi-scale validation. A practical roadmap is to (i) confirm phenotypes in a replicated hydroponic experiment with full biochemical replication; (ii) test in a controlled substrate system (sand/vermiculite or soil columns) where urea transformation and microbial processes are more realistic; and (iii) conduct field trials focusing on establishment, early-season nutrient uptake, and yield components. Integrating isotopic tracing (e.g., 15N-urea) could directly quantify nitrogen uptake efficiency changes attributable to humate inputs.


