Variations in immunological stress responses in Heteropneustes fossilis to short term and long term hypoxia
Author: S. Srivastava, Sanjeev kumar Choudhary
Journal: Журнал стресс-физиологии и биохимии @jspb
Article in issue: 1 т.22, 2026.
Free access
The effects of stress on organisms are wide ranging and so are the responses. In this study, exposures to different durations and intensity of hypoxia stress and their effects were considered. The present investigation focusses on the effects of short (<3h) and long term (8-12h) exposure to moderate (30-50% O2 saturation) as well as acute (<30% O2 saturation) hypoxia conditions on the haematological and immunological parameters in an airbreathing fish, Heteropneustes fossilis (Bloch, 1872). A significant increase (p<0.05) in erythrocyte numbers was observed during exposure to both short and long term moderate hypoxia whereas WBC counts and haematocrit values showed significant (p<0.05) increase during exposure to short term acute hypoxia. The immunological parameters showed a significant decrease (p<0.05) in total WBC counts, lymhopenia during short term exposure to moderate hypoxia and significant increase in total WBC count and neutrophilia, upon exposure to short term acute hypoxia. N/L (neutrophil/lymphocyte ratio) ratio – an important stress biomarker, was found to be increased significantly in the fishes exposed to short term acute hypoxia condition. H. fossilis, an airbreathing fish is adapted to living in low oxygen environments. An immunological stress is observed in this fish upon exposures to short duration (<3h) of both – moderate and acute hypoxia but not to exposures to long duration (8-12h). The results of this study are interesting as to how the duration and intensity of a stressor may exert variable influences in fish. The findings also appear significant in the light of adaptive mechanisms for stress tolerance operating in organisms.
Hypoxia stress, leucocyte indicators, stress adaptive strategies, fish
Short address: https://sciup.org/143185423
IDR: 143185423
Text of the scientific article Variations in immunological stress responses in Heteropneustes fossilis to short term and long term hypoxia
Increasing water pollution, global warming and resulting climatic changes have drastically affected the aquatic environment and its ecology, which stand constantly challenged. Aquatic systems act as sinks for all the atmospheric physical and chemical changes which can have far reaching effects upon the aquatic organisms. Dissolved oxygen levels in water (DO) which is vital to the growth and survival of all aquatic organisms including fish (Kramer, 1987; Caldwell and Hinshaw, 1994), is greatly affected with fluctuations in temperature and dissolved CO 2 levels in water. Tropical water bodies encounter seasonal drop in oxygen levels seen as naturally occurring aquatic hypoxia but pollution and global warming induced changes have resulted in unprecedented occurrence of short or prolonged reductions in dissolved oxygen levels in water bodies becoming a regular feature and one of global concern. Aquatic hypoxia has become a potential environmental stressor which is a major threat to the survival of all aquatic organisms including fish.
Effects of hypoxia, hypoxemia on fish physiology have been studied extensively ( andall, 1982; Shelton, et al. , 1986). Both acute and chronic hypoxia, can severely affect the physiology and metabolism of fishes causing homeostatic imbalances. Acute hypoxia is known to elicit ventilatory and cardiorespiratory responses in fish ( andall, 1982) whereas chronic hypoxia can produce several physiological and metabolic adjustments in tissues for greater oxygen extraction and delivery of oxygen (Jensen et al. , 1993; Sollid et al. , 2003). These physiological and biochemical changes are a part of the secondary stress responses that are initiated through the corticosteroids and catecholamines- the primary stress hormones (Mazeaud and Mazeaud, 1981; Peters et al. , 1991). The catecholamines have a marked influence upon the cardiovascular system causing haematological changes in erythrocytes and leucopenia (reduction in numbers), osmotic imbalances as well as on the immune system.
Most of the fish studied are found to be sensitive to hypoxia, and display varied responses to hypoxia stress. The responses of fishes however, are subject to the individual’s /species’ ability to mitigate, adapt or counter stress. Hypoxia induces many physiological and biochemical changes in fishes such as alterations in blood carrying capacity (Urbina and Glover, 2012), cardiorespiratory function (Belao et al., 2011, 2015), haematological changes (Peterson and Gamperl, 2011;
ichards, 2011; Ni et al. , 2014; ValAl et al. , 2015) and immunological changes (Ellsaeasser and Clem 1986; Kvamane et al. , 2013; Tawwab et al. , 2019) besides affecting food intake and growth (Bernier et al. , 2012; Mitter and ose, 2014) and reproductive function (Shang and Wu, 2004; Lanry et al. , 2007; Zhao et al. , 2014). Stress induced immunomodulation is commonly reported in fishes (Pickering,1981, Pickering and Pottinger, 1987; Pulsford et al. , 1994; Bowden, 2008). Immunocompetence is affected by the action of stress hormones, in the form of changes in lymphocyte numbers and antibody production capacity (Pickering, 1984). Any dysregulation of the immune system can be deleterious to the fish as it can lead to a persistent inflammatory state of the fish (Weyts et al. , 1999). This can lead to reduced disease resistance affecting the overall health and welfare of fishes.
Both acute and chronic fluctuations in dissolved oxygen levels affect the health and wellbeing of fish and other aquatic organisms. This holds true for most fishes that are water breathers. Heteropneustes fossilis (Bloch, 1872) is an air breathing fish and is generally found thriving in low oxygen swamps and muddy habitats. This study aims to investigate the effects of short and long term exposures of moderate and acute hypoxia conditions on the haematological and immunological parameters in this fish which is a dweller of a naturally hypoxic habitat.
MATERIALS AND METHOD
Acclimatization
Adult specimens of Heteropneustesf ossilis of average body weight 35 – 45g and average size 16 – 25 cm were purchased from the local market and maintained in the laboratory under natural photoperiod 14L:10D. The fishes were acclimatized in aerated aquaria (dissolved O2 7.5 – 11.5 mgl-1; pH 7.03-8.5) and water temperature (25-30◦C) in disease free condition. They were fed with chopped goat liver on alternate days and were maintained for 2-3 months before they were put on experiment.
Experimental protocol
Five batches (for each n=5/6) of Heteropneustes fossilis (body weight 31.22 ± 4.57g ; length 19.45 ± 1.96 cm) were made for each set. In each set of experiment, one batch served as control. Out of the four batches, two were employed for short exposures (<3h) of aquatic hypoxia and two for long exposures (8-12h). Each exposure schedule (short and long) was of both moderate hypoxia (30-50% oxygen saturation level) and acute hypoxia (<30% oxygen saturation level) experimental conditions. Three replicates were performed for each experiment.
For creating aquatic hypoxia conditions in the laboratory, a simplified version (Srivastava and uhela, 2019) of the Bennet and Beitinger model (1995) was used.
Blood Sampling
For blood sampling, the fishes were gently netted from the experimental aquaria and were anaesthetised with 0.35gl-1 ethyl-p-aminobenzoate (Acros, Germany). For hypoxia exposed fishes, care was taken to anaesthetise the fishes in hypoxic water that was drawn from the specific experimental aquaria. Blood was drawn from the caudal vein of the anaesthetised fishes, which was then collected in heparinized tubes. Aliquots of this, were then used for blood cell counts and smear preparations.
Blood Cell Count (RBC,WBC and DLC)
Total erythrocyte count ( BC) and total leucocyte count (WBC) were optically done with a Neubaur’s haemocytometer. For differential leucocyte count (DLC), whole blood was used to prepare blood smears that was first air-dried and then stained with Wright’s and Leishman’s stain. 200 cells were counted and the different leucocytes – lymphocytes, thrombocytes and neutrophils were represented as a percentage. Identification of these cells was done according to Ellis (1977). All the counts were performed in triplicates which were then averaged for agreement within a 15% difference.
Haematocrit (% Hct)
Whole blood was centrifuged and haematocrit was read to obtain the values.
Statistical Analysis
The statistical analysis was done using SPSS software. The analysis of variance between the observations of the experimental schedules was carried out using ANOVA and Duncan’s Multiple tests. A difference of p<0.05 was considered significant in all the tests.
RESULTS
RBC Count
A significant increase (p<0.05) in BC counts were found in H. fossilis exposed to moderate hypoxia both for short (<3h) and long durations (8-12h) as compared to the control (Table 1). During exposures to acute hypoxia – either short or prolonged, BC counts in the fish remained elevated.
WBC Count
Total WBC counts (Table 2) were observed to be significantly decreased (p<0.05) in the experimental fishes exposed to moderate hypoxia for less than 3h as compared to control. Short exposure to acute hypoxia produced significantly increased (p<0.05) WBC counts whereas no significant response was seen during long exposure to both moderate hypoxia and acute hypoxia.
DLC Counts
Differential leucocyte counts in H. fossilis were observed to be affected by exposures of the fish to experimental hypoxia conditions (Table 2). The percentage of lymphocytes were found to be significantly decreased (p<0.05) in fishes exposed to short duration (<3h) of moderate hypoxia whereas the percentage of neutrophils were observed to be significantly increased (p<0.05) in fishes exposed to short duration of acute hypoxia condition. The percentage of thrombocytes increased in all the exposure conditions, though insignificantly.
N/L ratio
The ratio of neutrophils to lymphocytes (N/L) in H. fossilis were found to be significantly increased (p<0.05) in the fishes exposed to short duration of acute hypoxia whereas it was significantly decreased (p<0.05) in the fishes exposed to long duration (Table 2., Fig. 2).
Haematocrit values
Haematocrit values (Hct%) were found to be significantly (p<0.05) increased in the fishes exposed to short exposure to acute hypoxia condition (Table 1., Fig.1.).
60,0 □
^ 55,0 : | 50,0 z 1 45,0 4 £ 40,0 4
I 35,0 4
о 30,0 4 га
- 25,0 4
20,0
control
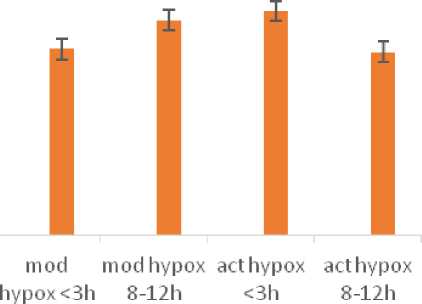
Figure 1. Haematocrit values (%) of H. fossilis under control and upon exposures to short (<3h) and long (8-12h) exposures of moderate and acute hypoxia conditions. Error bars indicate 95% confidence interval.
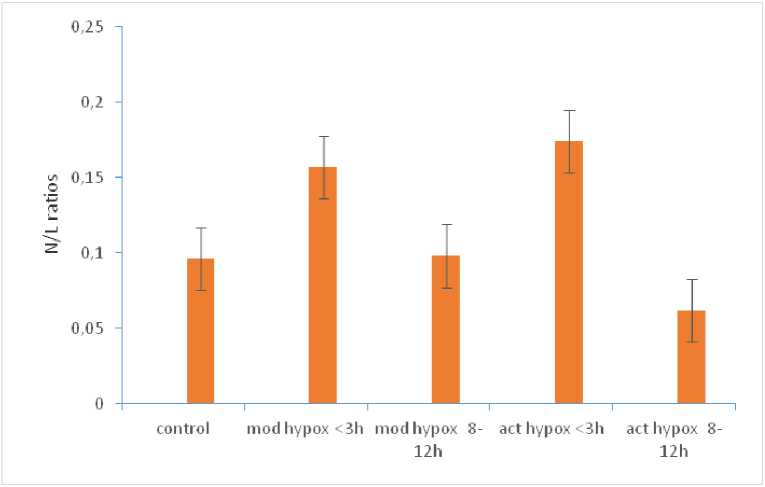
Figure 2. N/L ratios of H. fossilis under control and upon exposures to short (<3h) and long (8-12h) exposures of moderate and acute hypoxia conditions. Error bars indicate 95% confidence interval.
Table 1. Haematological parameters of H. fossilis exposed to short (<3h) and long (8-12h) exposures of moderate and acute hypoxia conditions. Values are expressed as Mean ± s.d. Different superscripts in lower case letters in columns represent significant differences (p<0.05).
|
Experimental conditions |
Experimental batches (n) |
BC count (x104 µL-1) |
WBC count (x103 µL-1) |
Haematocrit (Hct) (%) |
|
Control |
n=6 |
546.417±143a |
78.8287.3±7.46a |
43.867 ±1.46a |
|
Moderate Hypoxia (30-50% saturation) Short exposure (<3h) |
n=5 |
667.907± 102.10b |
60.705± 6.36b |
45.88± 3.14 |
|
Moderate Hypoxia (30-50% saturation) Long exposure (8-12h) |
n=5 |
730.760± 83.165 |
81.340±5.28 |
49.400 ±2.15 |
|
Acute Hypoxia (>30% saturation) Short exposure (<3h) |
n=6 |
566.133 ± 87.69 |
93.082 ±10.71c |
51.167 ± 8.11b |
|
Acute Hypoxia (>30% saturation) Long exposure (8-12h) |
n=6 |
600.57 ± 29.19 |
159.11 ± 16.28 |
45.520 ± 5.19 |
Table 2. Differential leucocyte counts and Neutrophil/lymphocyte (N/L) ratio in Heteropneustes fossilis exposed to short (<3h) and long (8-12h) exposures of moderate and acute hypoxia conditions. Values are expressed as Mean ± s.d. Different superscripts in lower case letters in columns represent significant differences (p<0.05).
|
Experimental conditions |
Experimental batches (n) |
Lymphocyte (%) |
Neutrophil (%) |
Thrombocyte (%) |
|
Control |
n=6 |
79.942 ± 4.0a |
7.44 ± 2.82a |
12.17 ± 2.08 |
|
Moderate Hypoxia (30- 50% O 2 saturation) Short Exposure(<3h) |
n=5 |
66.982 ± 4.55b |
10.502±3.59 |
22.437 ± 2.31 |
|
Moderate Hypoxia (30- 50% O 2 saturation) Long exposure (8-12h) |
n=5 |
80.875 ± 2.55 |
8.017 ± 1.53 |
10.857 ± 0.94 |
|
Acute Hypoxia (>30% O 2 saturation) Short exposure (<3h) |
n=6 |
73.962 ± 5.97 |
13.188±1.6b |
20.348±1.08 |
|
Acute Hypoxia (>30% O 2 saturation) Long exposure (812h) |
n=6 |
78.487±11.94 |
6.36±1.44 |
10.245± 2.40 |
DISCUSSION
Significant changes in haematological and immunological parameters were found to be induced in this study on Heteropneustes fossilis upon exposure to artificial aquatic hypoxia conditions.
In this study, both short (<3h) and long (8-12h) exposures of moderate hypoxia elicited a significant (p<0.05) increase in the erythrocyte numbers in H.
fossilis as compared to control, although the mean values of BC were fairly raised during both – long and short exposures of acute hypoxia as well. Fish erythrocytes are known to be sensitive to environmental changes. Their morphological evaluation are used as bioindicator of toxicity and their count values show considerable seasonal variability as well as variation with several environmental factors particularly water temperature and dissolved oxygen concentration (Swift, 1981, Moraes et al. , 1996, 1997, Affonso et al. , 2002,
Moraes et al. , 2002). The effects of hypoxia on fish respiratory physiology have been studied extensively (Shelton et al. , 1992) and low dissolved oxygen levels are known to affect respiratory efficiency and metabolic mechanisms. Increase in circulating erythrocyte numbers in response to hypoxia have been reported in the cod, Gadus morhua (Peterson and Gamperl, 2011), in intertidal fishes ( ichards, 2011), in sturgeon, Acipenser (Ni et al. , 2014) and are seen as adaptation to increase oxygen carrying capacity of blood and its delivery to the tissues during low availability of oxygen in the surrounding water. The response of the erythrocytes are also seen as part of the secondary stress response. The same seems to hold true for H. fossilis , which is adapted to living in waters of low dissolved oxygen levels.
A significant (p<0.05) increase in haematocrit values as compared to control, was observed, when H. fossilis was exposed to short duration (3h) of acute hypoxia whereas in the other exposure schedules, the values remained near to normal. An increase of haematocrit values in the blood of hypoxic fish are generally observed to be a fall out of changes in the erythrocytes. They may be due to erythrocyte swelling (Gallaugher and Farrell, 1998), decrease in plasma volume, increase in erythrocyte numbers and /or many such blood related responses, either in combination or singly (Peterson, 1990) and more recently has been ascribed to hypoxia inducing transcription factors-HIFs (Fang et al. , 2009). Tuvira, an electric fish Gymnotuscarapo presented a 50% increase in Hct values under exposures to hypoxia conditions (Moraes et al. , 2002). Increased values of haematocrit was also reported in the cod, (Petersen and Gamperl, 2011), in intertidal fishes ( ichards, 2011) and in sturgeons (Ni et al. , 2014). Haematocrit values are considered a key indicator of secondary stress response (Seibel et al. , 2021). Significantly increased values of haematocrit as observed for H. fossilis exposed to short exposure (<3h) of acute hypoxia indicate a stressed state of the fish.
Haematological responses are a part of the various adaptive mechanisms seen in fishes for coping with the oscillations in oxygen availability that often occur in the aquatic environment (reviewed by Val, 1993). However in fish, under stressful conditions such as hypoxia stress, catecholamines are suddenly released from chromaffin tissues ( andall and Perry, 1992). Increased levels of circulating catecholamines in the blood cause numerous adaptive changes, many of which are directed towards enhancing oxygen transfer and delivery to the tissues ( andall, 1990). The release of catecholamines has also been correlated with the release of red blood cells from the spleen (Nilsson and Grove, 1974; Perry and Kinkead, 1989), red cell swelling (Nikinmaa et al., 1990) and changing haematocrit values (Le Blanc et al., 2011).
It is notable that the effect of short exposures of both moderate and acute hypoxia was also observed on the circulating leucocytes in Heteropneustes fossilis. Total WBC counts were found to be significantly decreased (p<0.05) when the fishes were exposed to short duration of moderate hypoxia and the numbers increased significantly in the fishes exposed to short exposure to acute hypoxia (Table 2). The leucocytes are important components of the first line of defense and are involved in the regulation of immunological functions (Ellis, 1977; Tort, 2011). All environmental stressors, both natural and artificial, are considered to suppress immune functions in fish (Bly et al., 1997). Several environmental stress factors – pollutants (Dunvier et al., 1991, Zelikoff, 1993), handling and confinement (Elsaesser and Clem, 1993) and low temperatures (Abram et al., 2017) are reported to suppress components of both innate and adaptive immunity in fish. Hypoxia is a potential environmental stressor and has been reported to elicit both primary and secondary stress responses (Pollock et al., 2007). It is interesting that while short exposures of hypoxia conditions affect immune functions as observed in this fish, long exposures of both moderate and acute hypoxia do not elicit any such response. According to Barton and Peter, 1982, the initial period of exposure to a moderate stressor induces a transient increase in plasma cortisol and associated stress response in fish. The responses observed in H. fossilis can be seen as adaptive stress responses to mitigate short duration hypoxic conditions. Prolonged hypoxia conditions do not elicit any such response. Though cortisol was not measured in this study, it could well be assumed that the immune response induced by short exposures of hypoxic conditions could have been triggered by the transient release of the stress hormones.
Table 2 sums up the predominant stress effect of short exposures – both moderate and acute on the immunological responses in H. fossilis . While short exposure to moderate hypoxia produces lymphopenia in H. fossilis , a similar exposure to acute hypoxia produces neutrophilia (Table 2), long duration exposures to both moderate and acute hypoxia did not elicit any significant immunlogical response. Stress induced immunomodulation is not uncommon in fish (Ellsaesser and Clem, 1986, Pickering and Pottinger, 1987, Tort, 2011). Differential leucocyte counts in fish exposed to various stressors showed an increase in phagocytes and a decrease in thrombocytes (Pulsford et al. , 1994). A decrease in lymphocytes was also observed by Ellseasser and Clem (1986). Altered levels of leucocytes in blood are considered a reflection of the physiological responses generated in response to increased cortisol levels under stressful conditions and are considered key indicator of secondary stress response. Stress hormones are reported to inhibit the proliferation of lymphocytes (Barton and Iwama, 1991), apoptosis of granulocytes (Weyts et al. , 1998), as also the release of monocytes and neutrophilic granulocytes from the haematopoetic tissues of the head kidney into the peripheral circulation (Ortuno et al. , 2001). DLC have been used to assess the stressed state of the fish in several cases such as heavy metal exposure in common carp, Cyprinus carpio (Vinodhini and Narayanan, 2009), organochloride herbicide exposure in the African catfish, Clarias gariepinus (Oluah et al. , 2020), or crowding stress in the Atlantic salmon, Salmo salar (Delfosse et al. , 2020). A stress leukogram involving significant lymphopenia and neutrophilia was reported in the zebra fish, Danio reiro (Grzelak et al. , 2017) in both chronic and acute stress.
atios of leucocyte population in fish blood are considered as good indicators of the immunological state of the fish and their response to the environmental changes. Especially important are the neutrophil/lymphocyte (N/L) ratio which are considered as important indicator of distress across vertebrates (Davis et al., 2008). In this study, a significantly increased N/L ratio was observed in H. fossilis exposed to short duration (3h) exposure to acute hypoxia indicating a stressed state of the fish. This is certainly due to the increase in the circulating neutrophils. Short exposure to acute hypoxia induced significant neutrophilia as well as a significant increase in the total WBC count. This also explains the increased value of the total WBC count under short exposure to acute hypoxia stress, which can be ascribed to neutrophilia. Neutrophilia have been reported to proliferate in blood in response to infections, inflammation and stress (Campbell, 1995; upley, 1997; Thrall, 2004) and was also found in the zebra fish only during acute stress exposures (Grzelak et al., 2017). Srivastava and Choudhary (2010) observed altered N:L ratio in C.batrachus, following exposure to artificial photoperiods. However, under long duration (12h) exposure to acute hypoxia, the ratio was observed to be significantly decreased indicating a no-stress condition for the fish. In all probability, decreased numbers of neutrophils could have contributed to this. It appears to be a kind of an adaptive response of this fish to a chronic situation of acute hypoxia which is also in ecological adjustment with the habitat of the fish. The observations in this study on H. fossilis emphasize the role of neutrophils as an important stress indicator.
Stress responses are undoubtedly, indicative as well as measurable characteristics of a disturbed homeostasis in response to challenged environmental conditions, which are ultimately aimed to restore the internal equilibria. Much depends on the coping ability of the organism, its threshold for stress tolerance (Vaquer Sunyer and Duarte, 2008) and its ecological adaptation, that primarily will govern the mitigation of the stress responses and restoration of the homeostasis through stress responses. The stress responses induced in an organism may not necessarily always indicate a “distressed state”. They could also be a part of a coping / adaptive strategy to tide over a challenging condition. Barton (2002) considered stress responses as adaptive mechanisms to cope with perceived stressors. This holds particularly true for organisms that are continuously exposed to the challenging conditions as in case of intertidal fishes ( ichards, 2011) or chronic (prolonged) hypoxia as in the case of the fish under study, H. fossilis. The results emphasize that a sudden short exposure to hypoxia is temporarily stressful to such fishes to which they elicit adaptive stress responses. Prolonged (chronic) hypoxia is a condition to which they are adapted and so are not affected by them. The mechanism and evolution of the adaptive value of the stress responses is an area generating lot of interest ( ichards, 2011, Balasch and Tort, 2019, Tenji et al., 2020).
Heteropneustes fossilis is reported to show acclimation to moderate hypoxia and display morphometric and anatomical changes in the interrenal and chromaffin tissues upon exposures to acute hypoxia (Srivastava and uhela, 2017, 2019) wherein the fish was observed to have considerable plasticity for hypoxia stress. Significantly, the immunological responses of the fish to hypoxia stress as seen in this study reflect the same plasticity. It is primarily the short exposures – either to moderate or acute hypoxia, that has elicited significant changes in the haematological and immunological parameters. The N/L ratio, a significant stress indicator (Davis et al. , 2008; Davis and Maney, 2018) is significantly increased under short exposure (<3h) to acute hypoxia indicating a stressed state of the fish and significantly decreases during prolonged exposure to acute hypoxia in this fish, again indicating a ‘no stressed state’. It appears that prolonged hypoxic conditions which are in accordance with the ecological requirements of H. fossilis , do not induce stress in this fish, though sudden brief exposures of both moderate or acute hypoxia elicit stress signals. The results of this study can be important in understanding the adaptive nature of the stress responses to hypoxia in H. fossilis and may find application in dealing with hypoxia stress in fish culture systems.
CONCLUSION
The study highlights the importance of adaptive stress responses and their mechanisms. A stressor may not always elicit a stress response unequivocally; it is subject to the type, intensity, duration of a stressor as well as an organisms’ adaptive ability to tolerate stress. Short durations of both moderate and acute hypoxia exposures elicit haematological and immunological stress in H. fossilis whereas prolonged exposure to acute hypoxia conditions do not. Heteropneustes fossilis, an airbreathing fish adapted to hypoxic environment displays variable stress responses. The findings of the present study may help in our understanding of stress responses and find application in aquaculture and fish farming systems in the management of hypoxia stress and stress adaptation in fishes in general.
ACKNOWLEDGEMENT
Acknowledgements are due to the financial assistance provided by UGC, Delhi and to the Principal, MM(PG) College, Modinagar, for all the laboratory and financial facilities.
CONFLICTS OF INTEREST
The authors declares that they have no potential conflicts of interest.


